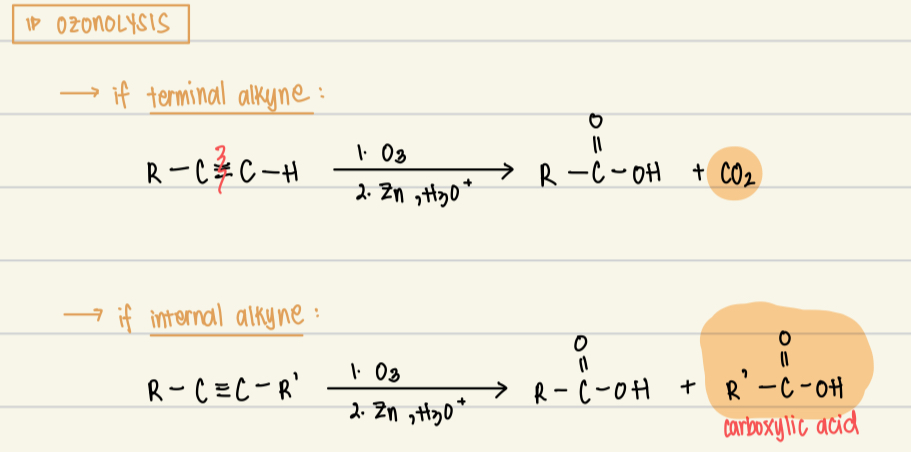Organic Chemistry - Alkynes
1/14
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
Saturation
Unsaturated
General formula of linear alkynes
C (n) H (2n-2)
General formula of cycloalkynes
C (n) H (2n-4)
Polarity
Non-polar
What increases its boiling point?
Increasing number of carbons
What lowers its boiling point?
More substitution along carbon chain
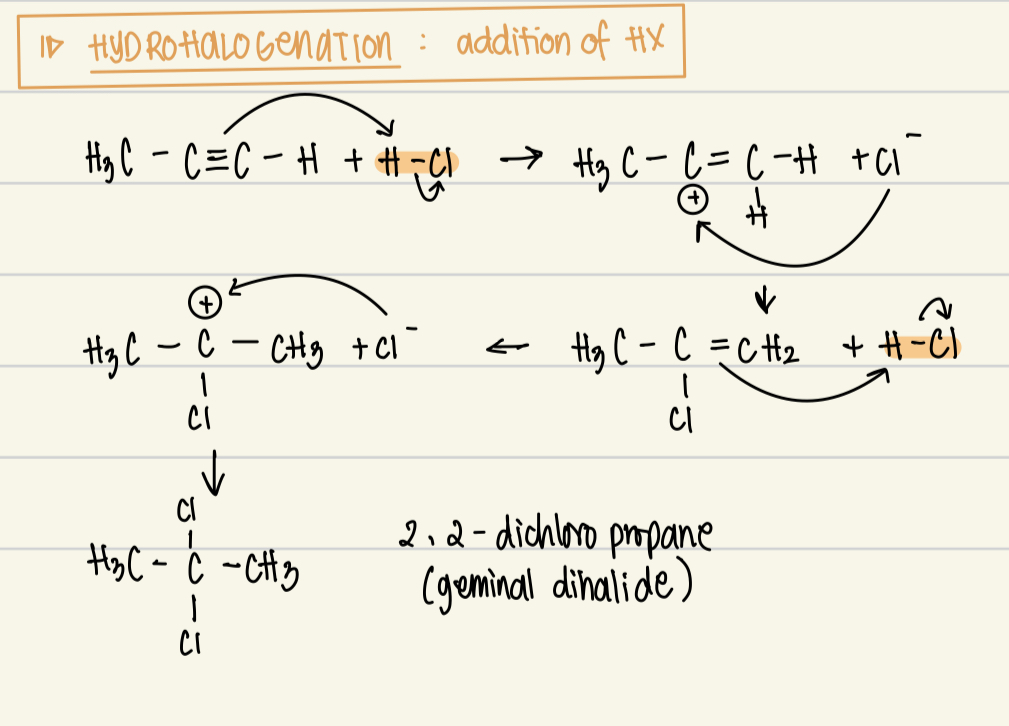
Hydrohalogenation reaction reagent, catalyst, product and mechanism
Reagent: HX
Catalysts: none
Product: geminal dihalide
Mechanism: electrophilic addition
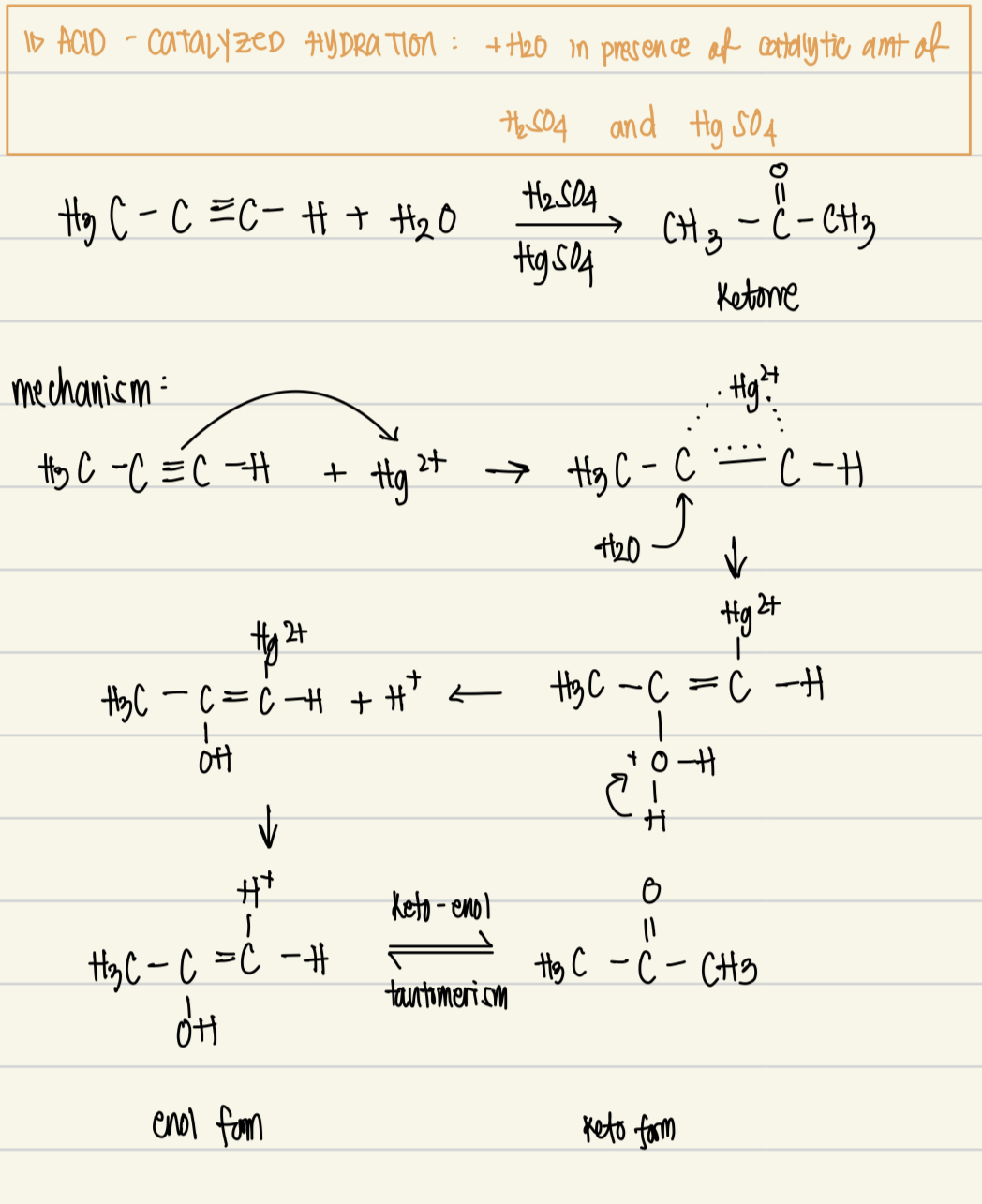
Acid catalyzed hydration reagent, catalysts, product, mechanism
Reagent: H2O
Catalysts: H2SO4 and HgSO4
Product: keto form from enol form (keto-enol tautomerism)
Mechanism: electrophilic addition
Halogenation reagent, catalyst, product, mechanism
Reagent: X2
Catalysts: none
Product: geminal dihalide
Mechanism: electrophilic addition

Hydroboration-oxidation reagent, catalyst, product, reaction
Reagent: (BH3)2
Catalysts: none
Product: aldehyde from enol (keto-enol tautomerism)
Mechanism: electrophilic addition then oxidation
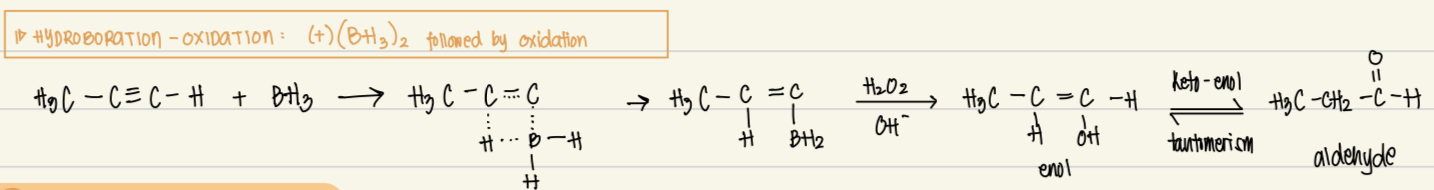
Metal acetylide reaction reagents, catalyst, product, mechanism
Reagent: metal acetylide + RX
Catalysts: none
Product: alkyne-R + metal-X
Mechanism: nucleophilic addition

Catalytic Hydrogenation for Pd/C catalyst
Reagent: 2H2
Catalysts: Pd/C
Product: alkane
Mechanism: reduction of alkynes

Catalytic Hydrogenation for Lindlar Catalyst
Reagent: H2
Catalysts: lindlar caralyst (poisoned Pd or Pd/CaCO3
Product: cis alkene
Mechanism: reduction of alkynes

Catalytic Hydrogenation for NaNH2 or LiNH2 catalyst
Reagent: H2
Catalysts: NaNH2 or LiNH2
Product: trans alkene
Mechanism: reduction of alkynes
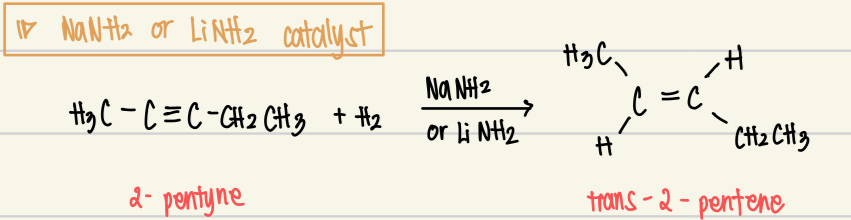
Ozonolysis reagent, catalys, product, mechanism
Reagent: 1. O3
Catalysts: 2. Zn, H3O+
Product: if terminal alkyne, CO2. if internal alkyne, carboxylic acid
Mechanism: oxidation of alkynes