gcse physics topic 6 radioactivity
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
SP6b - what are the three subatomic particles as well as their masses

SP6b - how are the subatomic particles displayed on a periodic table
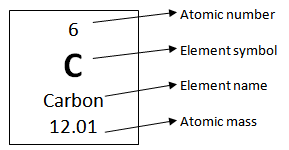
SP6b - what is an isotope
an atom of an element but has a different mass number / number of neutrons
SP6c - how do electrons exist around the nucleus
orbiting in electron shells
SP6c - when electrons absorb energy they can what
jump into higher shells
SP6c - what happens when they lose the energy
they emit visible light
SP6c - what is an ion
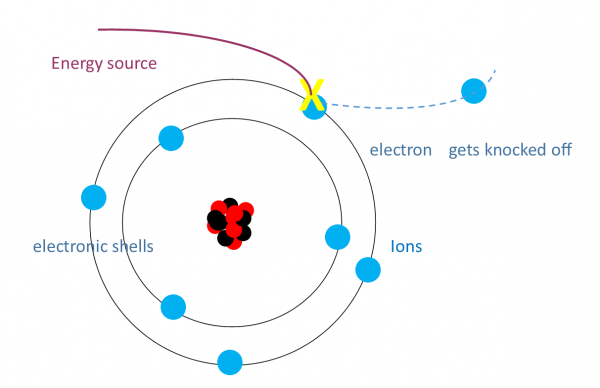
when an electron gets too much energy it can get knocked off which forms a ion with a positive charge (cation)
SP6d - what is background radiation?
natural radioactive substances we are always exposed to
SP6d - what are some examples of background radiation
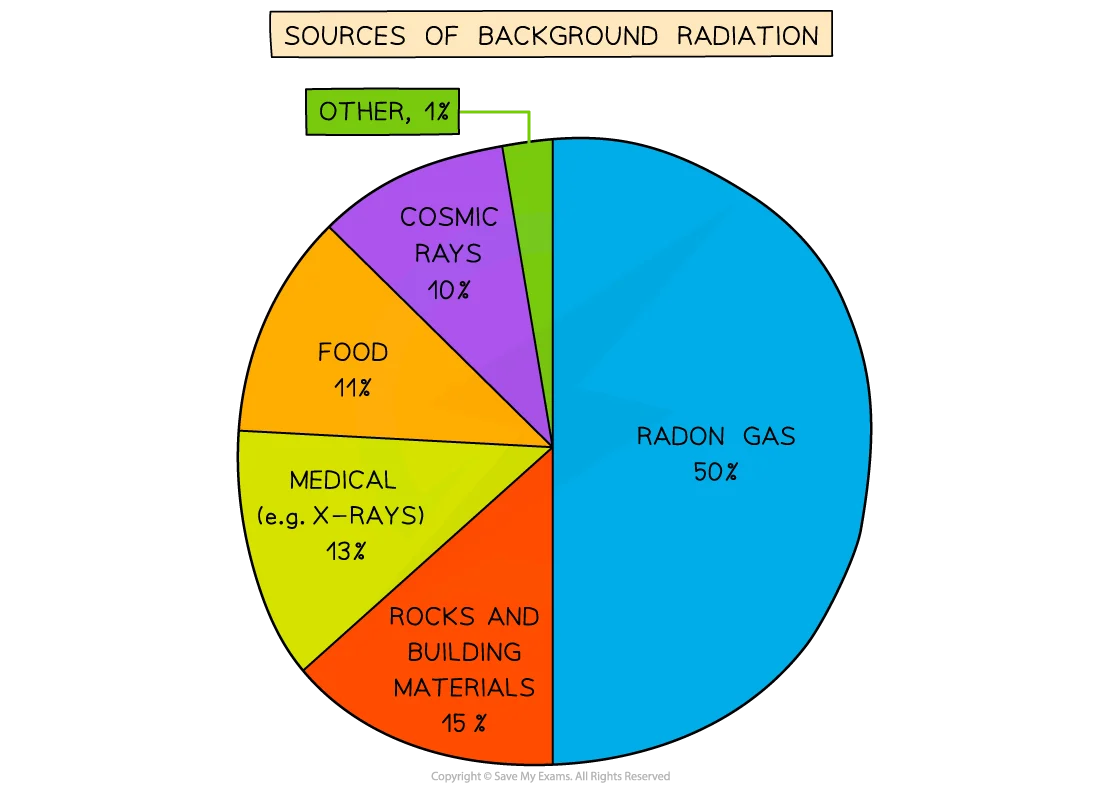
SP6d - how to measure radioactivity
dosimeter badges can be developed to see the dose exposed to
geiger-muller (GM) tube which shows count rate