chem 3.9 - acid-base equilibria
1/48
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
what is an acid?
a proton donor
what is an acid? (lowry-bronsted theory)
a substance that dissolves to give hydrogen ions (H+) when they are dissolved in water
what is a monobasic acid?
an acid that donates one proton (H+) per molecule
HCl(aq) → H+(aq) + Cl-(aq)
what is a dibasic acid?
an acid that can donate two protons per molecule
H2SO4(aq) → H+(aq) + HSO4-(aq)
what is a tribasic acid?
an acid that can donate three protons per molecule
H3PO4(aq) → H+(aq) + H2PO4-(aq)
what is a base?
a proton acceptor
acid-base pair equartion
HA(aq) + H2O ⇌ H3O+(aq) + A-(aq)
HA - acid 1 (conjugate acid of base 1)
H2O - base 2 (conjugate base of acid 2)
H3O+ - acid 2 (conjugate acid of base 2)
A- - base 1 (conjugate base of acid 1)
what does the pH scale measure?
the concentration of H+(aq) ions
how do you calculate pH?
pH = -log10[H+]
How do you calculate [H+]?
[H+] = 10-pH
describe the pH scale?
logarithmic scale
low pH means high [H+], high pH means low [H+]
what does a logarithmic scale for pH mean?
a change of 1 in pH means a change of 10 in the [H+']
what is a strong acid?
an acid that completely dissolves in aqueous solutions to produce H+ ions and equilibria lie completely to the right
HA(aq) ⇌ H+(aq) + A-(aq)
examples of strong acids
HCl
HNO3
H2SO4
what can we assume about [H+] in strong acids?
[H+] is the same as the concentration of the acid
[H+] = [acid]
how do you calculate pH of strong acid?
find [H+] - use concentration of acid
use -log10 to find pH
what is the expression for the equilibrium constant of the self ionisation of water?
not Ka its Kc btw
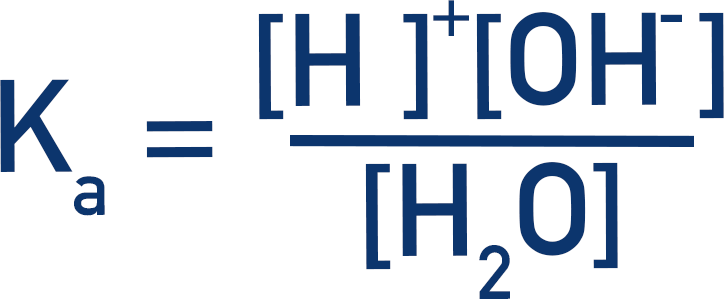
what is Kw?
the ionic product of water
1.01 × 10-14 mol2dm-6 at 25 degrees
what is the expression for Kw?
Kw = [H+][OH-]
can rearrange to find OH- or H+
relationship between [H+] and [OH-] in pure water and neutral solutions?
[H+] = [OH-]
relationship between [H+] and [OH-] in acidic solutions?
[H+] > [OH-]
relationship between [H+] and [OH-] in alkaline solutions?
[H+] < [OH-]
How do you calculate the pH of a strong base?
find H+ using [H+] = Kw/[OH-]
use -log10 to find pH
what are weak acids?
acids that only partially dissociate in aqueous solutions and the equilibria lie to the left
CH3COOH ⇌ CH3COO- + H+
what can we assume about [H+] in weak acids?
[H+] = [A-]
what can we assume about [HA] in weak acids?
[HA] = [acid]
what are the units of Ka?
moldm-3
what does Ka indicate?
the strength of an acid
how can you calculate Ka?
Ka = 10-pKa
how can you calculate pKa?
pKa = -log10Ka
what does a higher Ka mean?
lower pKa
stronger acid
what does a lower Ka mean?
higher pKa
weaker acid
what is the expression for Ka of weak acids?
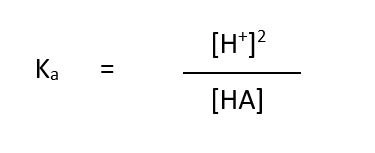
how do you calculate the pH of a weak acid?
use Ka = [H+]2/[acid]
rearrange to find H+ using [H+]2=√Ka x [acid]
use -log10 to find pH
what is a buffer?
a solution that resists change in pH on addition of small amounts of an acid or a base
cannot prevent pH from changing completely but pH changes are minimised
what is a buffer a mixture of?
a weak acid (HA) and its conjugate base (A)
how can you make a buffer? (method 1)
mix together a weak acid and soluble salt of the weak acid
e.g. ethanoic acid and sodium ethanoate
how can you make a buffer? (method 2)
add a strong base to double the quantity of a weak acid
base will neutralise half of the acid -`. forms its conjugate base
conjugate base forms mixture with the other half of the acid which remains in the solution
how does a buffer work if an acid is added?
H+ conc increases
reaction is reversible
so equilibrium moves to left
most of additional H+ is removed
because A- and HA are so big, when equilibrium moves these changes are negligible so H+ remains constant
how does a buffer work if an alkali is added?
H+ conc decreases
reaction is reversible
added OH- ions removes H+ ions
so equilibrium moves to right
because A- and HA are so big, when equilibrium moves these changes are negligible so H+ remains constant
what are some examples where buffer solutions are used?
fermentation using yeast
dyeing cloth
using or storing enzymes of pharmaceuticals
human blood
label the titration curve
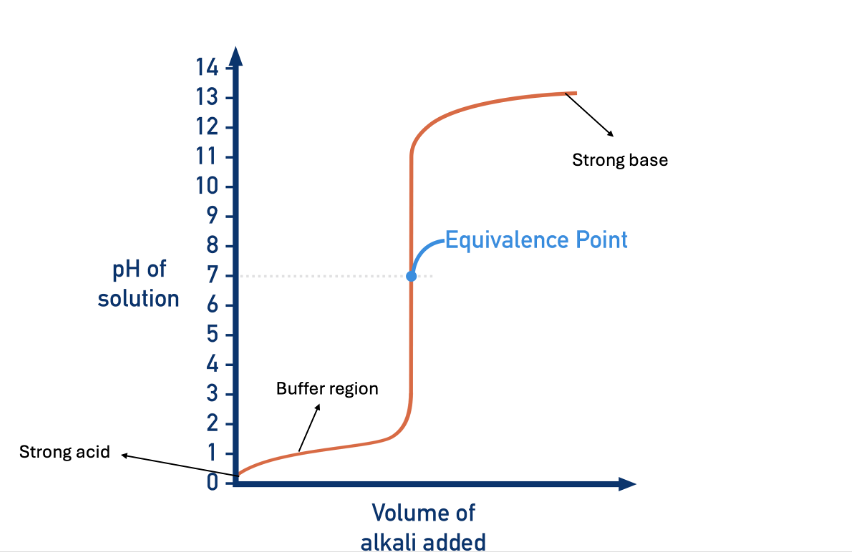
what is the equivalence point of a titration curve?
vertical section
region where an indicator works if the pH range of that indicator falls in the vertical region
how do you calculate the pH of a solution of acid-base reactions?
work out moles of acid
work out moles of base
find difference between moles to get excess mol of acid or base
if H+ (acid) is in excess
conc of H+ = mol/vol
pH = -log[H+]
If OH- (base) is in excess
conc of OH- = mol/vol
H+ = Kw/OH- = 10^-14/OH-
pH = -log[H+]
how do you calculate the pH of a buffer? ( equations)
or
[H+] = Ka x [HA]/[A-]
or
[H+] = Ka x [ACID]/[SALT]
![<p>or </p><p>[H+] = Ka x [HA]/[A-]</p><p>or</p><p>[H+] = Ka x [ACID]/[SALT]</p>](https://assets.knowt.com/user-attachments/111152cb-2ff7-4686-b753-f840517c5c10.png)
how do you step by step calculate pH of a buffer?
calculate mol of acid → use mol = conc x vol
calculate mol of salt → use mol = conc x vol
calculate conc of acid AND salt → mol/vol (use total volume)
sub into [H+] = Ka x [ACID]/[SALT]
pH = -log[H+]
if a salt is formed from a strong base and a strong acid, what will its pH be?
neither of the ions they contain interact with water
neutral salt
pH = 7
e.g. NaCl, KBr
if a salt is formed from a strong base and a weak acid, what will its pH be?
salt will dissociate to produce conjugate base of weak acid
conjugate base will accept proton from surrounding water molecules
there will be excess of OH- in solution
so pH > 7
if a salt is formed from a weak base and a strong acid, what will its pH be?
salt will dissociate to produce conjugate acid of weak base
conjugate acid will donate protons
pH < 7