Alkyl Hallides
1/19
Earn XP
Description and Tags
Chapter 11
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
The larger the halogen, the _____ the carbon-halogen bond, and the _____ the bond length.
The larger the halogen, the weaker the carbon-halogen bond, and the larger the bond length.
Based on electronegativity, we expect the carbon of alkyl halides to be ______ polarized. Making the carbon the __________.
Based on electronegativity, we expect the carbon of alkyl halides to be positively polarized. Making the carbon the electrophile.
the higher the bond strength, ___________, and the more _____ the radical is
the higher the bond strength, the harder it is to rip apart, and the more unstable the radical is
radical stability (most stable to least stable)
3o allylic/benzyllic
2o allylic/benzyllic
1o allylic/benzyllic
3o
2o
1o
acrylic/vinylic
radical substitution requires three steps:
initation
propogation
termination
how to make an alkyl halide
radical halogenation
allylic bromination (use NBS)
monohalogenation
dihologenation
alcohol
oxidation
decreases e- density on carbon by
forming C-O, C-N, C-X
breaking C-H
reduction
increasing e- density on carbon by
forming C-H
breaking C-O, C-N, C-X
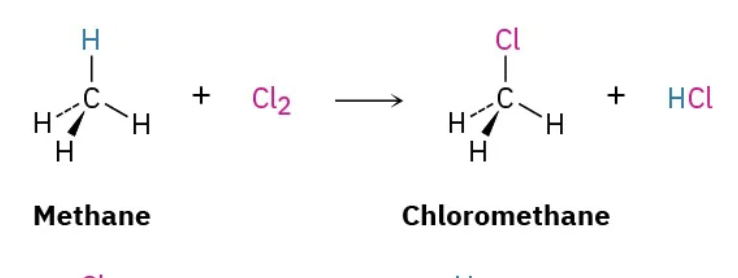
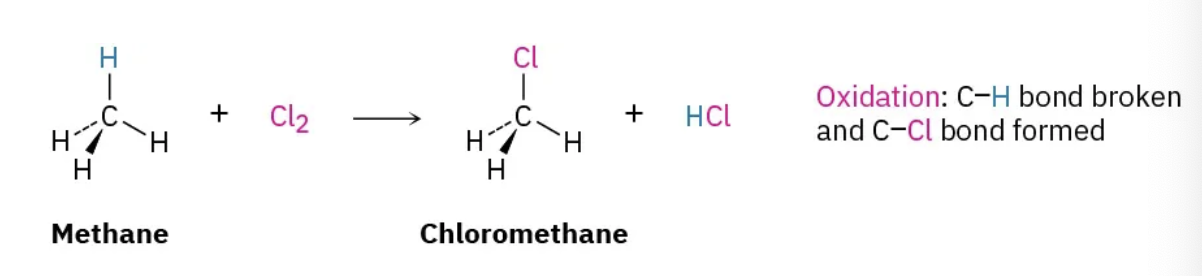
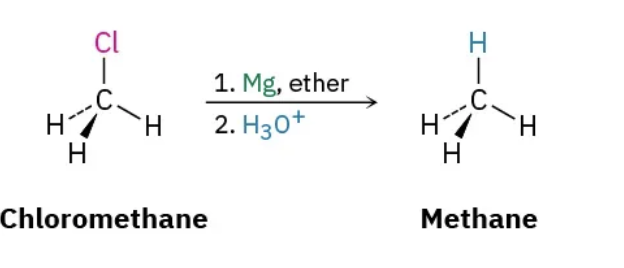
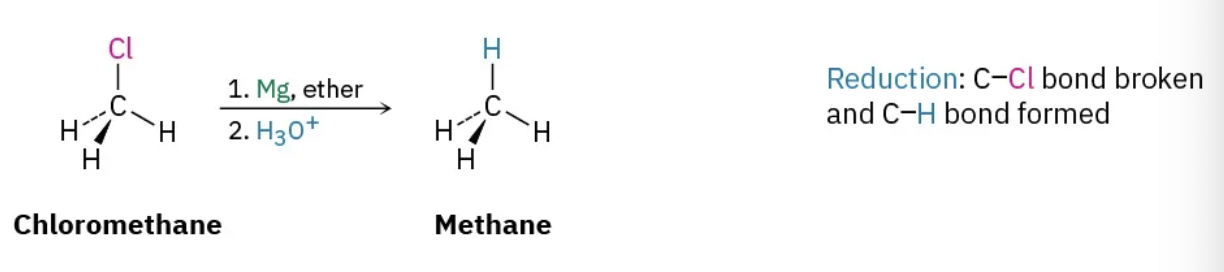
radical halogenation
alkane (no double bonds) + light + Cl2 or Br2 = alkyl Hallie
Cl is not selective
Br is selective
in radical halogenation _____ is not selective, while ____ is selective
in radical halogenation Cl is not selective, while Br is selective
Br is less reactive (less electronegative) than Cl, and as a result only really reacts with the weakest C-H bond (tertairy), resulting in the most stable radical (tertiary radical)
allylic bromation
NBS+ alkene + light = alkyl halide with bromine at the allylic position
alcohol substitution
HCl, HBr, HI (not HF), can rip alcohols off of tertiary carbons
SOCl2 and PBr3 can rip alcohols off of primary and secondary carbons, they also flip the stereocenters
HF can rip alcohols off of primary and secondary carbons, but they are often combined with pyradine for safety reasons
when we start with an alkene we can use
HBr (monohalogenation)
Br2 or Cl2 (dihalogenation)
NBS
formation of Grignard reagents $
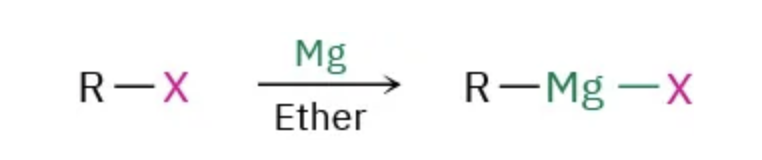
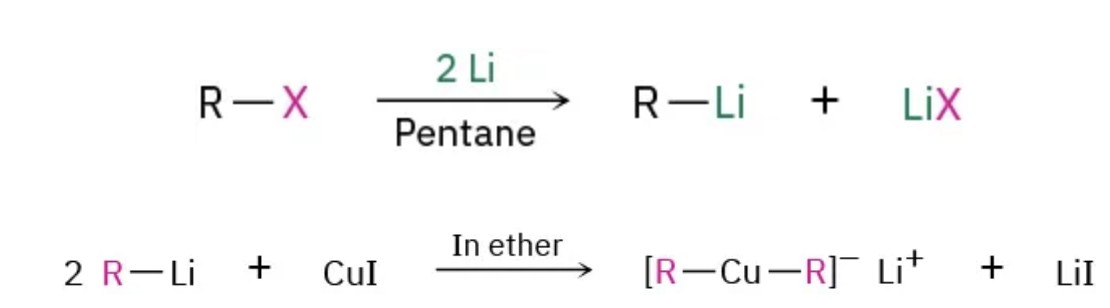
formation of gilman reagents
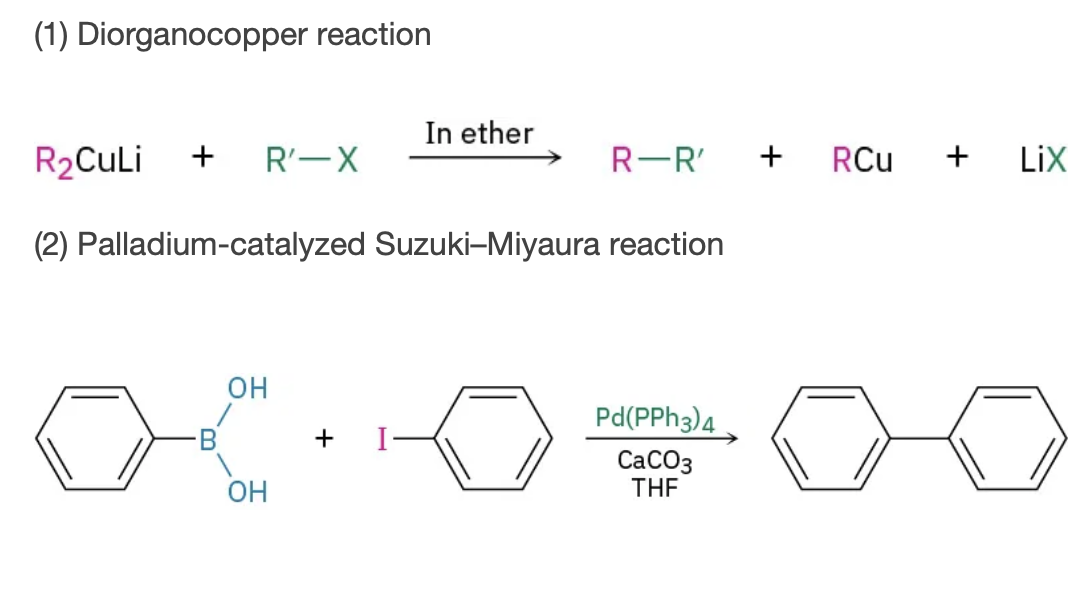
organometalic coupling
we always pair HF with
pyridine
gringard reations occur in what solvents
ether, THF (weakly polor, aprotic solvent)