Enzyme inhibitors
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
What are competitive inhibitors
They have similar shape to substrates and compete against them for the active site, and the one in the greatest concentration has the greater chance of binding to active site. Can still reach V max by adding more substrate.
What are non-competitive inhibitors
They do not have a similar shape to substrate and bind to the allosteric site of enzyme. When they bind this alters the shape of the active site so substrate can no longer bind so active site is no longer complimentary.
Why can V max not be reached with non-competitive inhibitors
Enzymes cannot catalyse reactions properly, total number of functional enzymes reduced so maximal possible rate is lower
What are reversible inhibitors
Inhibitors which have a weak affinity for enzyme so can be easily dislodged
What are irreversible inhibitors
Inhibitors which have stable bonds with enzyme so can’t easily be removed
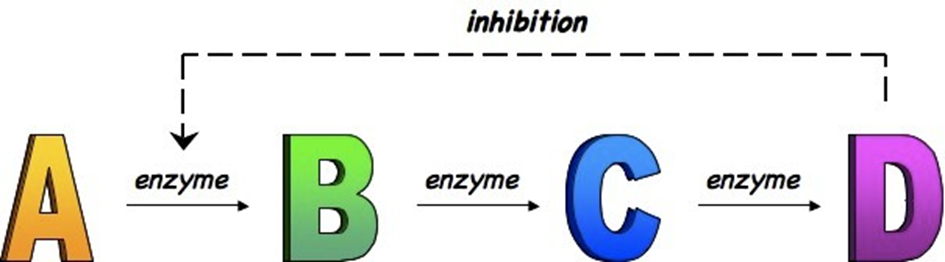
What is end product inhibitlon
When the end product in a metabolic pathway binds to an enzyme at the start of the pathway, the final product as a non-competitive inhibitor to enzymes earlier on chain. This serves as a a negative feedback control to ensure excess product is not made and intermediate metabolic precursors not wasted
Describe how you could investigate the concentration of enzyme inhibitor on the activity of an enzyme
Note: using milk coagulating as an example practical for this
IV: Concentration of enzyme inhibitor e.g. 1%,0.5%,0.25%
By doing a serial dilution by using half enzyme and half dilute water then do same for other conc.
DV: Observe every 30s and record time in seconds when clots
CV: Temperature 37oC using a water bath, volume of inhibitor 1cm3, volume of milk 10cm3, volume of enzyme 1cm3
Apparatus:
Test tubes
Distilled water
Stop clock
Water bath
Apparatus for volume measurements such as syringes 10cm3
Beaker
Repeat for different concentrations of inhibitors three times and calculate the mean
Why is it important correct concentration of enzyme is used in an reaction
If too much inhibitor is used then it could affect the quality of the final product, and if there is not enough inhibitor used the reaction could continue and excess product