science CAT test physical and chemical change
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
What are compounds?
Pure substances containing more than one different atom bonded together.
What is a physical change?
When a substance looks different but its particles have not changed
What is a chemical change?
When a new substance has been formed that has different particles then the original substance
What is evidence that a chemical change has taken place?
a gas is produced
there is a permanent change in colour
light or heat is produced
they are irreversible
a solid is formed from 2 solutions
What is some evidence a physical change has taken place?
changes to the objects shape
is reversable
phase changes like melting or freezing
What are some examples of a chemical change?
burning wood
rusting iron
baking
food going off
What are some examples of a physical change?
melting ice
boiling water
chopping wood
breaking glass
What is an endothermic reaction
Reactions that absorb energy energy from its surroundings causing them to become colder.
What is an example of an endothermic reaction?
instant cold packs
melting ice
baking a cake
What is an exothermic reaction?
Reactions that release energy/ heat into its surroundings.
What is an example of an exothermic reaction?
lighting a match
explosions
magnesium reacting with oxygen
What is an element?
Substances with atoms of the same element bonded together.
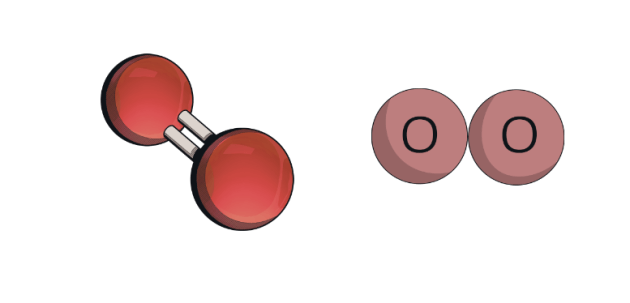
Is this an element or compound?
element
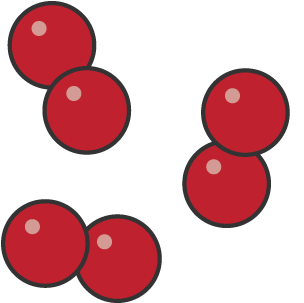
Is this an element or compund?
element
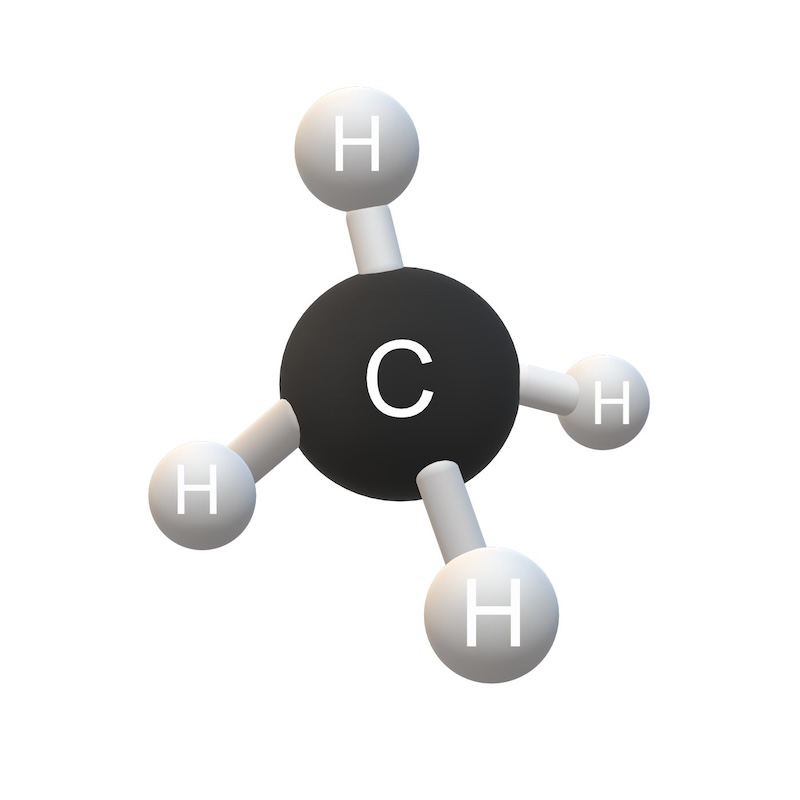
Is this an element or compund?
compound
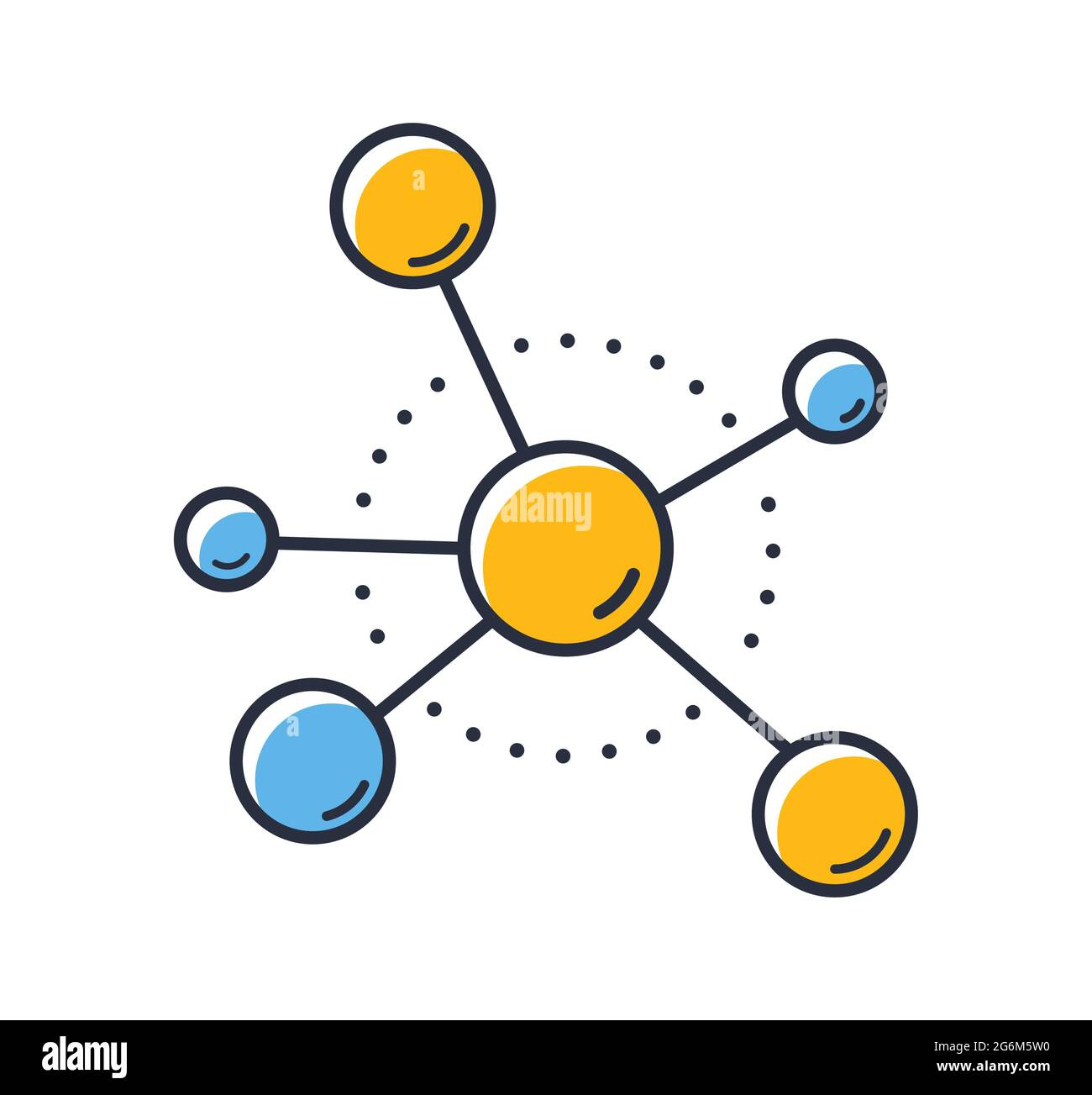
Is this an element or compound?
compound
What is a chemical formula?
Something that scientists use to show types and numbers of atoms that make up a compound.
What are the different types of compounds?
Ionic
Covalent
What is an ionic compound?
Something that has a metal bonded to a different non metal
Give an example of an Ionic compounds
Carbon oxide
Sodium chloride
What in a covalent compound?
Something that has a non-metal bonded to a different non-metal
Give an example of a covalent compound
water
carbon dioxide
What are the rules for naming chemical compounds?
The ending of the final element is changed to -ide
What are the rules for naming ionic compounds?
write the name of the metal
write the name of the non-metal second and change ending to -ide
How do you name covalent compounds?
Write elements in the order they appear
A prefix goes in front of each element
If there is one atom of the first element no prefix is needed
The ending of the last element is changed to -ide
What is the prefix for one?
mono
What is the prefix for two?
di
What is the prefix for three?
tri
What is the prefix for four?
tetra
What are chemical changes also called?
Chemical reactions
What are products?
The new substance formed
What are reactants?
The original substance
Where do the reactants go in a word equation?
First
Where do products go in word equations?
second
How do you seperate the reactant and product in a word equation?
an arrow (→)
What is a synthesis reaction?
When two reactants combine to form one product
What is a decomposition reaction?
When one reactant breaks down into two products
What is a displacement reaction?
When one element in substituted for another
How do you represent Endo and Exo reactions in word equations?
By adding the word ‘energy’ to either the reactants or products
How do you know what side the energy should go on in a word equation?
If energy is absorbed → reactants side
If energy is released → products side