Enzyme
1/5
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
how do enzymes work
made of protein mainly
act as biological catalysts which hold active sites which is a region where the substrate/reactant binds to for the process of enzyme catalysis
included fit model
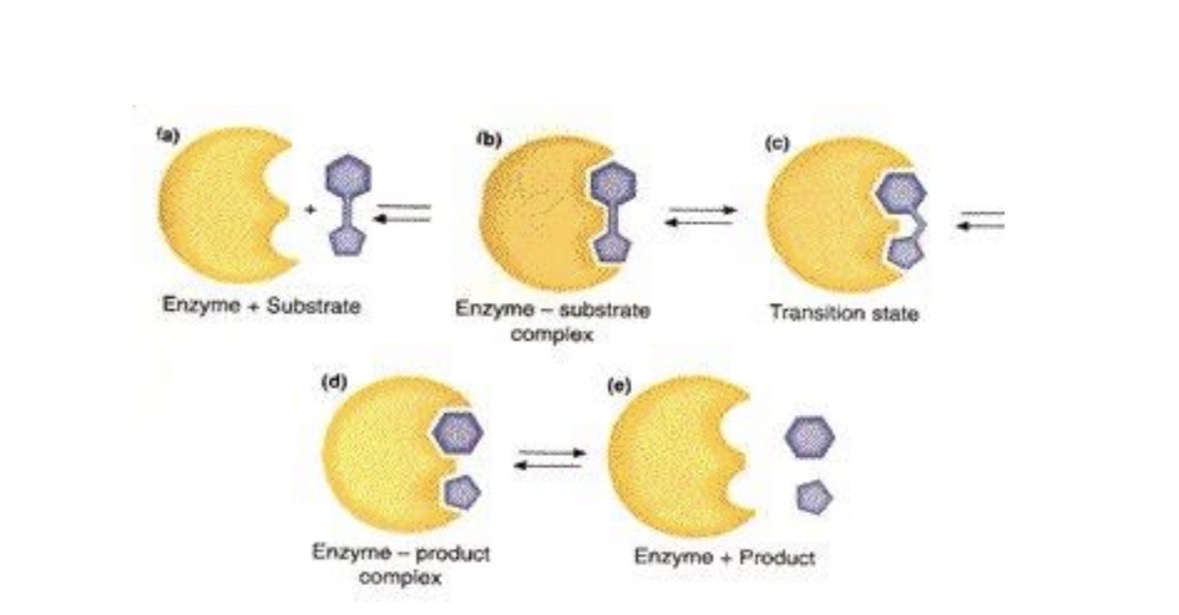
the substrate will attach to enzyme by H-bonding then break off into 2 monosaccharides
what is inhibition
its the control of an enzyme based on a molecule called an inhibitor
The types of inhibitor
Competitive Inhibition: The inhibitor looks like the substrate and competes for the active site, blocking the reaction.
Non-Competitive Inhibition: The inhibitor binds to a different spot on the enzyme and changes its shape so the substrate can’t bind.
Allosteric Regulation: A molecule binds to a different spot on the enzyme and can either turn it on (activator) or off (inhibitor) by changing its shape.
Feedback Inhibition: The final product of a reaction stops an earlier step to prevent too much product from being made.
Factors affecting
Temperature:
Increasing temperature generally increases enzyme activity because particles move faster
and collide more often.
If the temperature gets too high, the enzyme can change shape (denature) and stop
working.
If it’s too cold, reactions slow down.
- Enzyme concentration:
More enzyme = faster reaction, as long as there is enough substrate to work on.
If the substrate runs out, the rate levels off even if enzyme amount keeps increasing.
- Substrate concentration:
More substrate = faster reaction until all enzyme active sites are “busy.”
Once enzymes are saturated, the rate plateaus and won’t go faster.
- pH:
Each enzyme has an optimal pH (best pH for its shape/function).
Too high or too low pH can denature the enzyme or change its active site, slowing or
stopping the reaction.
Extra
The enzyme has an active site that is specific to a particular substrate.
The substrate will be attracted to the active site by h-bonds, and electrostatic attractions. The substrate binds into the active site to form the enzyme substrate complex.
Any time something attaches to the enzyme it causes a change in the enzyme's 2°, 3°, 4° structures by altering the H-bonds and electrostatic attractions between amino acids within the protein. This change in shape by the enzyme causes the reaction to occur.
Once the reaction occurs the products are released and the enzyme returns to normal ready to start all over again.