Thermochemistry Review
Calorimetry
Measuring the amount of heat released or absorbed during a chemical reaction.
q = m ⋅ c ⋅ ΔT
- q = heat (J)
- m = mass of water (g)
- c = specific heat capacity (J/(g ⋅ °C))
- ΔT = change in temperature (°C; Final - Initial)
Specific Heat Capacity
The amount of energy required to raise the temperature of 1kg or 1g of the substance by 1°C.
The specific heat capacity of water is 4.186 J/(g ⋅ °C)
Heat flow (Enthalpy)
Increases the thermal energy of one body and decreases the thermal energy of the other.
Enthalpy (ΔH) is the amount of heat transferred during a reaction.
Both open and closed systems have heat transfer.
ΔH = q / moles
- ΔH = enthalpy change (J / mol)
- q = heat energy (J)
- moles = number of moles reacted (mol)
Standard conditions
Thermochemical standard state conditions
- T = 298.15 K or 25°C
- P = 1.00 atm
- 1M concentration in an aq solution
Endothermic & Exothermic
| Endothermic | Exothermic |
|---|---|
| Absorbs heat / energy. Feels cold. +ΔH. ↓ΔT. Hp > Hr.S → L → G | Releases heat / energy. Feels hot. -ΔH. ↑ΔT. Hp < Hr.G → L → S |
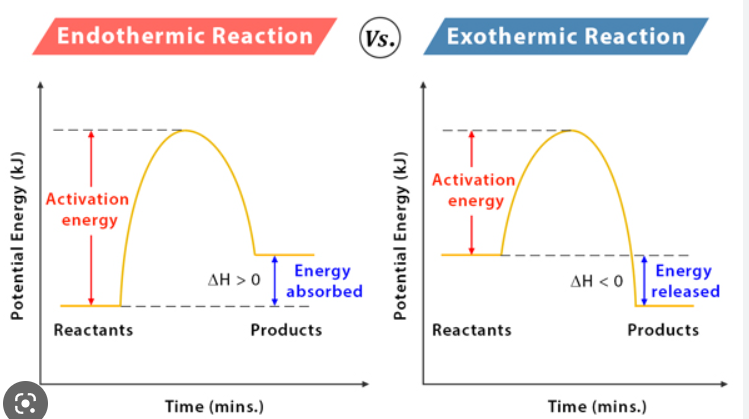
All Ways To Calculate Enthalpy Change
- Graphs
- The difference between the starting and ending energy. Found at the beginning and end of a graph.
- Activation energy (Ea) is the difference between the highest point of the graph and the starting energy.
- Hess’s law
- Give multiple reactions and their corresponding enthalpy you must get certain molecules to cancel out by either multiplying the whole equation or flipping them.
1. Flipping = flip sign of ΔH. 2. Multiplying = multiply sign of ΔH.
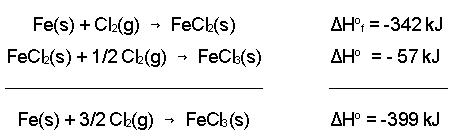
- ΔH0 = ΣHf0p - ΣHf0R = q / moles
For all of the following use:
ΔH0 = ΣHf0p - ΣHf0R
- ΔH0 = standard enthalpy change (kJ/mol^-1)
- ΣHf0p = sum of standard heat of formation for the products
- ΣHf0R = sum of standard heat of formation for the reactants
- You will be given Hf values for each of the reactants and products.
- Heat of formation
- From its elemental form.
- 2C(s) + 3/2H2(g) + 1/2Cl2(g) → C2H3Cl(g)
- Heat of combustion
- CxHy + O2 → H2O + CO2
- Heat of fusion / vaporization
- Solid to a liquid to a gas
- H2O(s) → H2O(l)
1. ΔH = 1.43 Kcal (1 Kcal = 4.184 kJ)
Maxwell Boltzmann
Describes the distribution of speeds among the particles in a sample of gas at a given temperature.
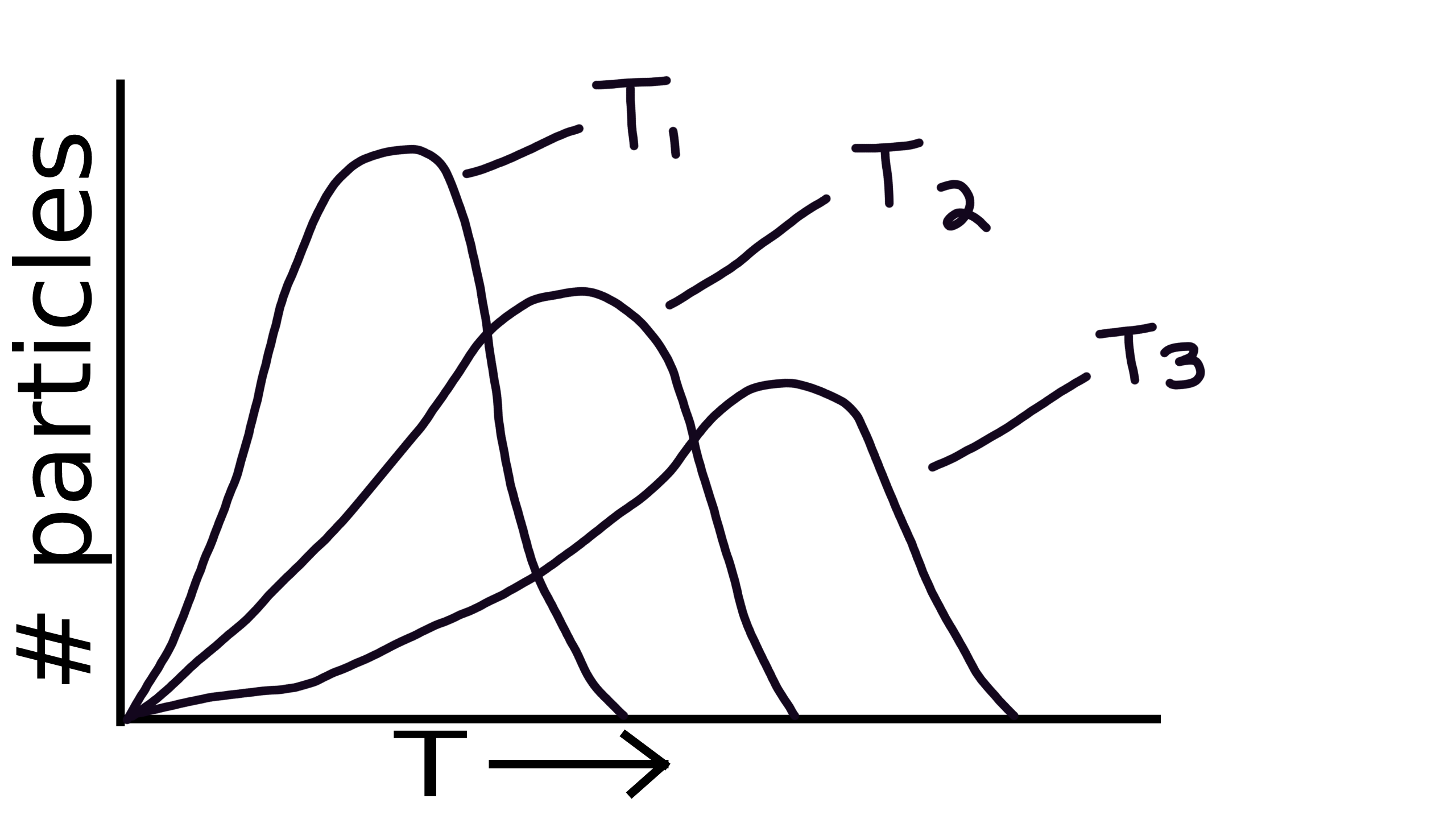
T1 < T2 < T3
Higher temperature means lower peak and moved farther right.
Low temperature will have higher peak and moved father left.