module 4 - core organic chemistry
1/116
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
117 Terms
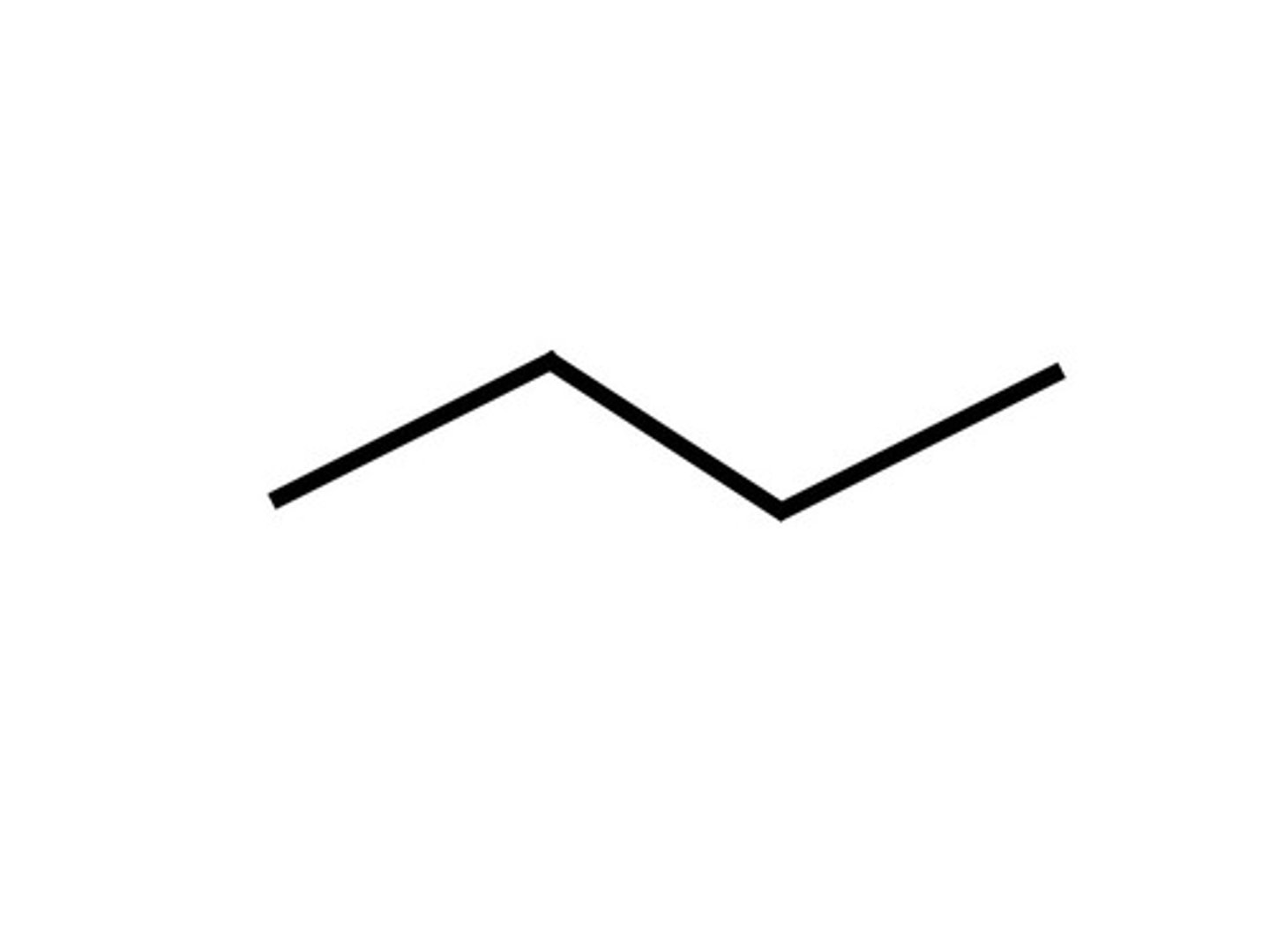
What is the skeletal formula?
shows the simplified organic formula, shown by removing hydrogen atoms from alkyl chains leaving just a carbon skeleton and associated functional groups
What is an alderhyde and how would you name one?
Alderhyde = compound with a carbonyl C=O group at the end of a chain. Written as -CHO not -COH as that's an alcohol
Suffix is -al e.g. ethanal
What are carboxylic acids and how would you name one?
Carboxylic acids = contain a -COOH group at the end of a carbon chain only but could be on the end of a side chain as well as the main chain.
Suffix is -oic acid
What is the difference between sigma and pi bonds?
Sigma bonds = single covalent bonds
Pi bonds = double covalent bonds
Sigma bonds are stronger than pi bonds so pi bonds are more reactive but overall they're non-polar bonds so they aren't very reactive.
What is a mechanism?
Mechanisms show how the molecules interact and how the bonding electrons are reorganised during a reaction.
What is a homologous series?
A series of organic compounds with the same functional group but with each successive member differing by CH2
What is a molecular formula?
shows the number and type of atoms of each element present in a molecule - doesn't provide any structural info

What is the structural formula?
a formula that shows the arrangement of atoms in the molecule of a compound - smallest amount of detail but shows which groups are bonded together
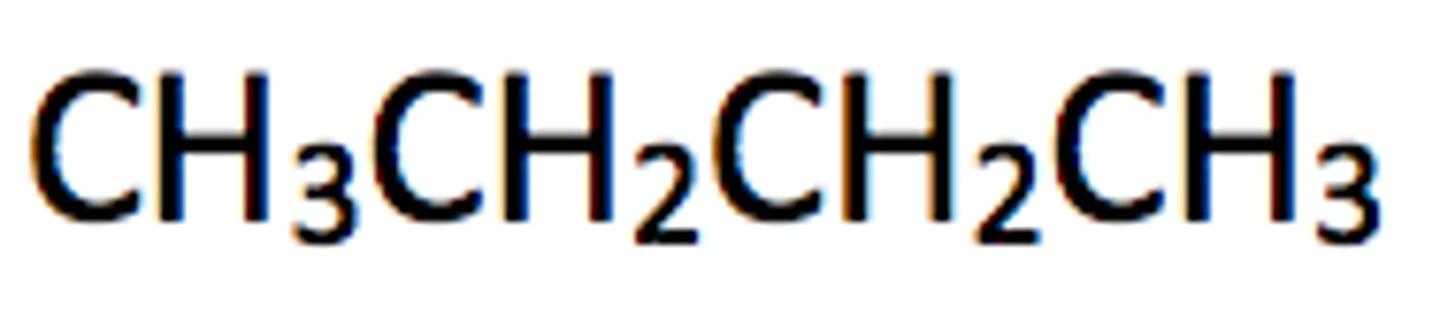
What are the rules for naming alkenes?
Suffix of -ene.
Position of the double bond must be stated for alkenes with 4 or more carbons e.g. pent-2-ene
What are the rules for naming alcohols?
Either suffix -ol or prefix hydroxy
However the prefix is only used when there's another suffix functional group.
Position of the hyroxy group must be stated with alcohols with 3 or more carbons in their chain e.g. Butan-1-ol
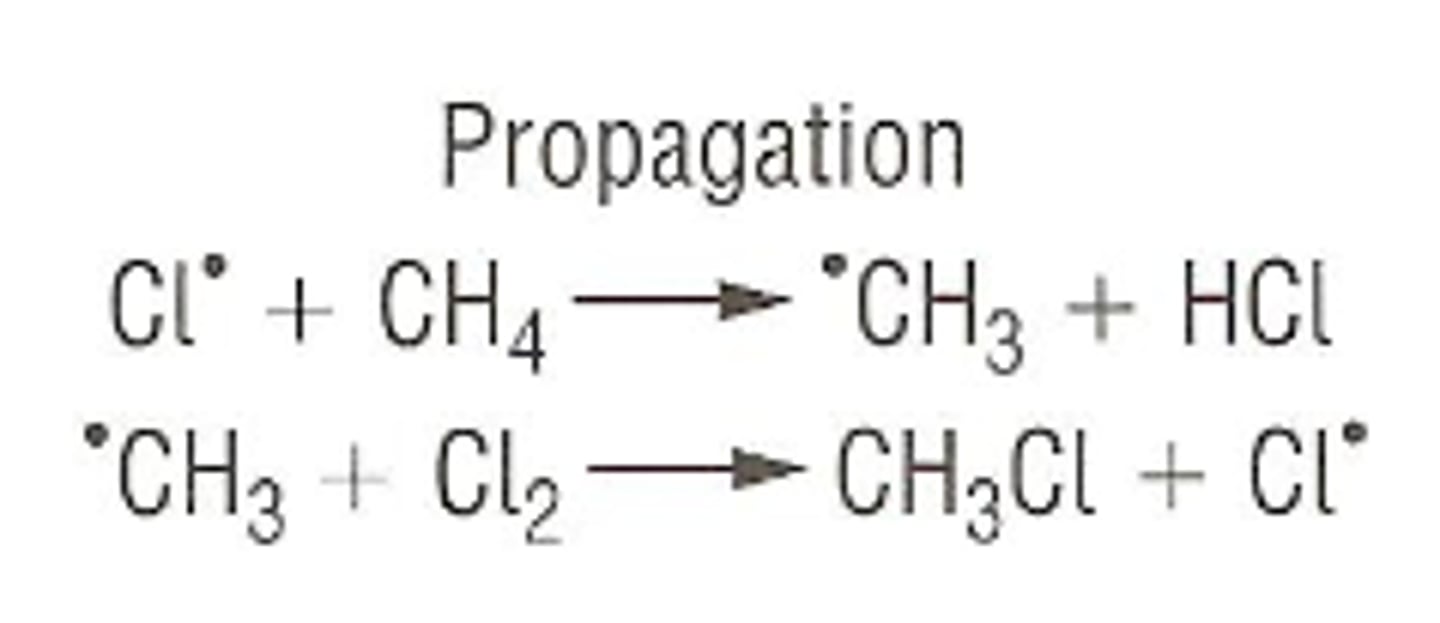
What is free radical substitution?
A 3 step reaction mechanism when a halogen free radical replaces a hydrogen atom - shows the movement of electrons in a reaction between an alkane and halogen.
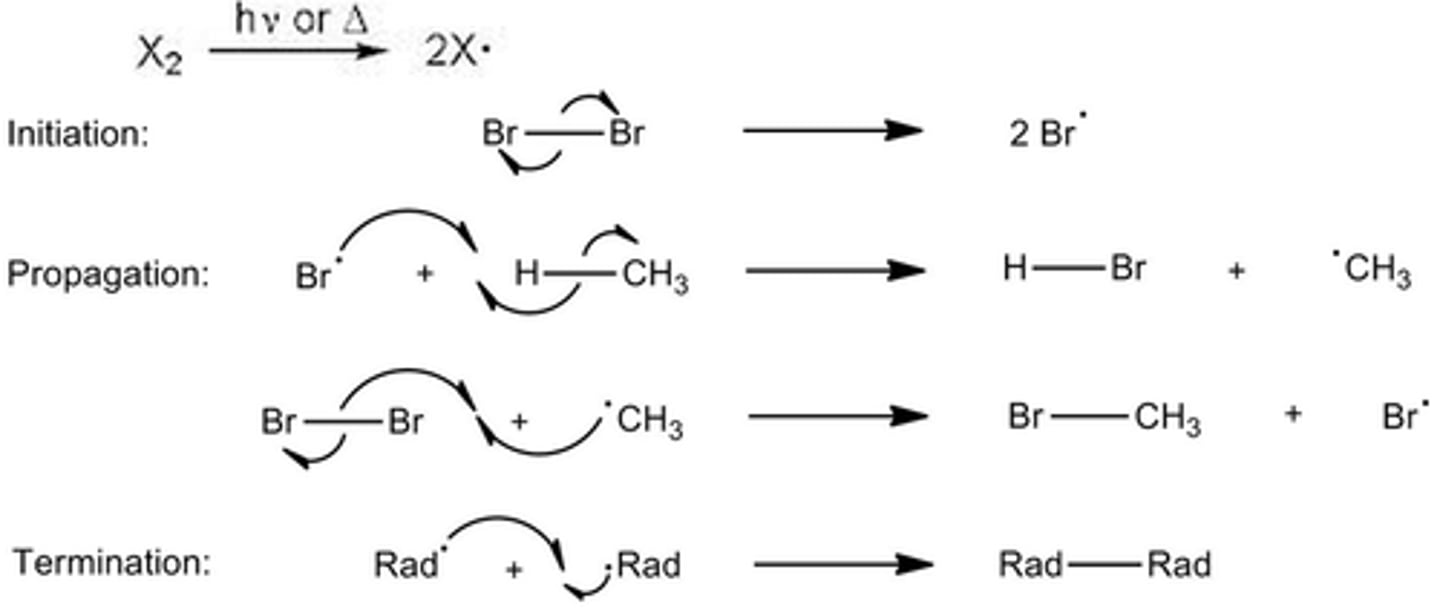
What are the different stages in free radical substitution?
Initiation, Propagation, Termination

What happens in propagation?
Free radicals react with other molecules forming another radical.
What is an isomer?
compounds with the same molecular formula but different structural formulas
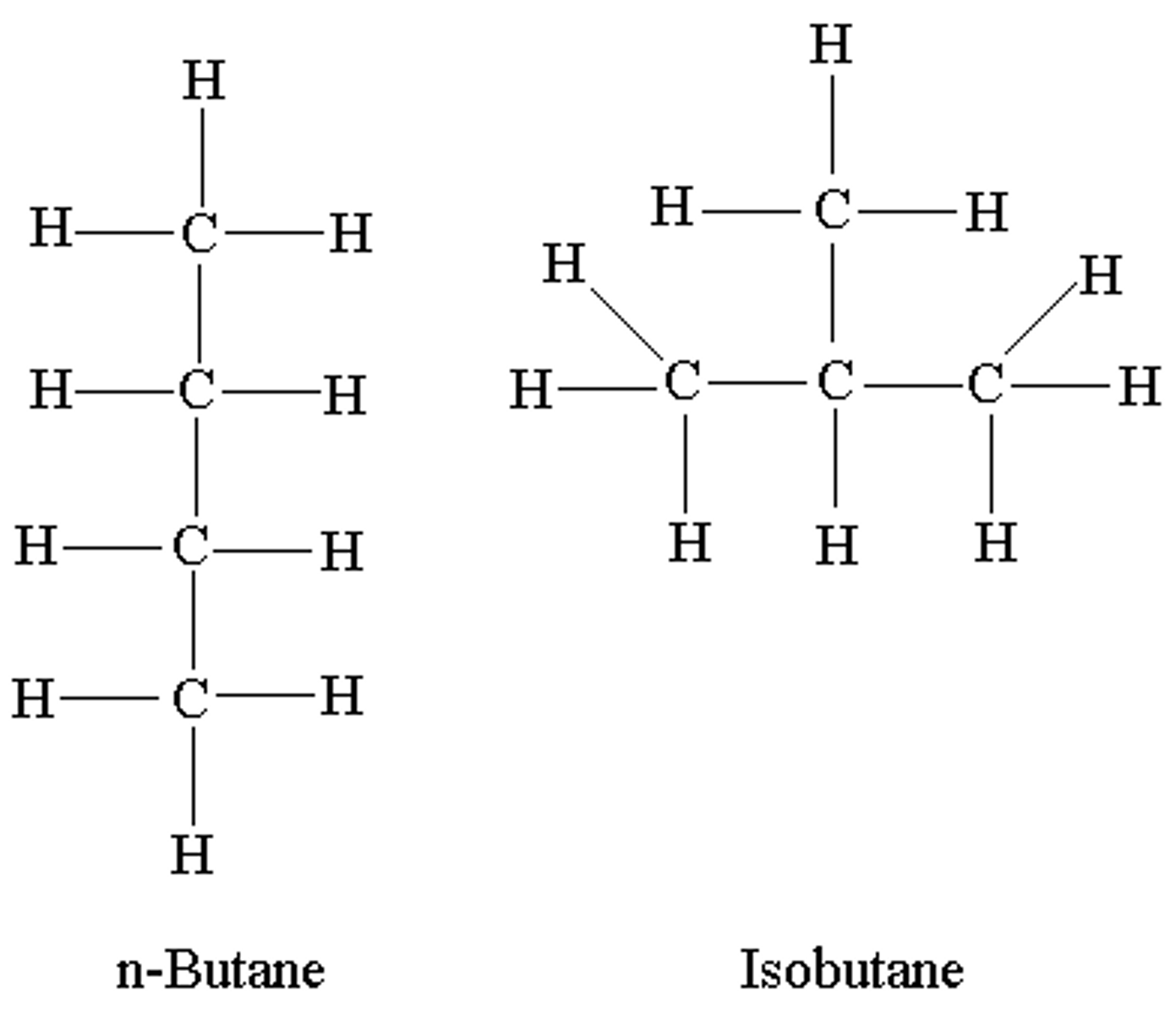
What is structural isomerism?
Compounds that have the same molecular formula but different structural formula
What are the different ways you can make haloalkanes?
- Alkane ----> Haloalkane through free radical substitution
- Alcohol ----> Haloalkane through nucleophillic substitution where the OH group swaps with the halogen
- Alkene ---> Haloalkane through electrophillic substitution where a HX ( X = halogen) breaks/replaces the double bond
What is organic chemistry?
the study of compounds that contain carbon
What is a hydrocarbon?
a compound made of hydrogen and carbon atoms only - alkanes & alkenes
What is the difference between saturated & unsaturated?
Saturated hydrocarbons/organic molecules have single carbon bonds only.
Unsaturated hydrocarbons/organic molecules have at least one carbon-carbon multiple bond (double or triple)
What are the alkanes?
The simplest homologous series - saturated hydrocarbons
What is a functional group?
An atom or group of atoms responsible for the characteristic reactions of that compound.
What are examples of a functional group?
Oxygen, nitrogen & halogen atoms.
What is the general formula?
The simplest algebraic formula for any member of a homologous series e.g. for alkanes Cn H 2n+2
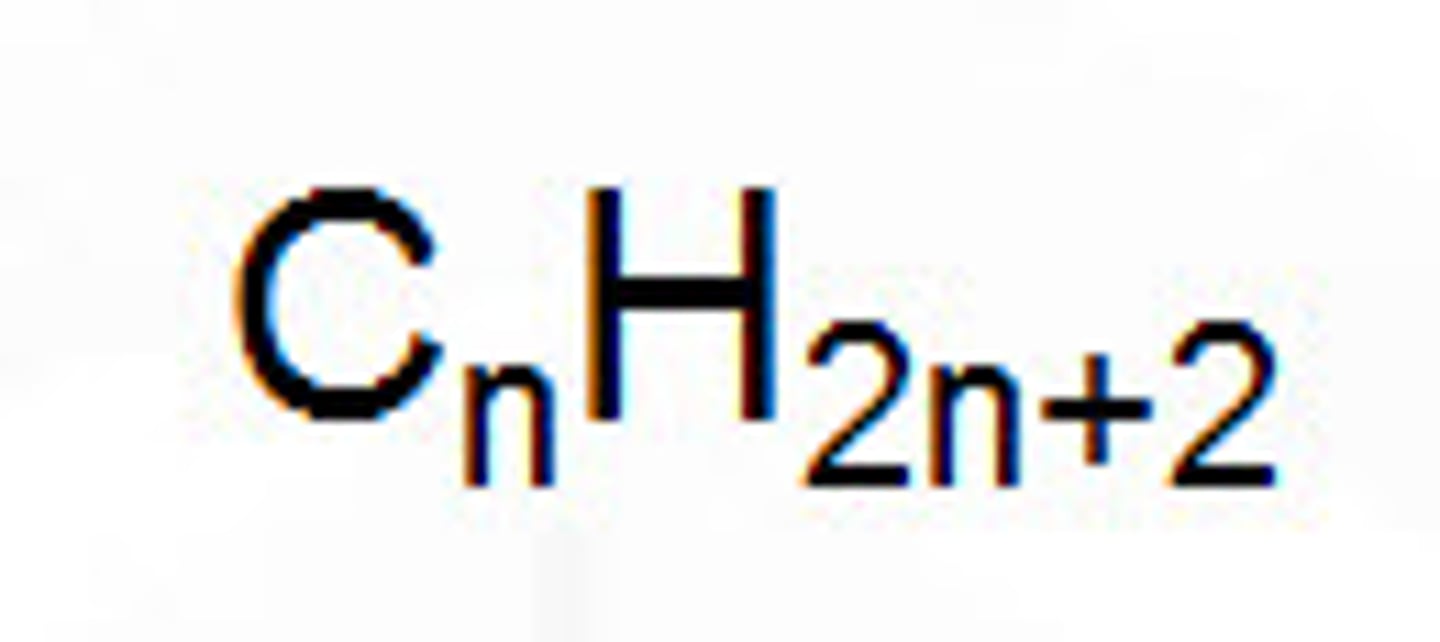
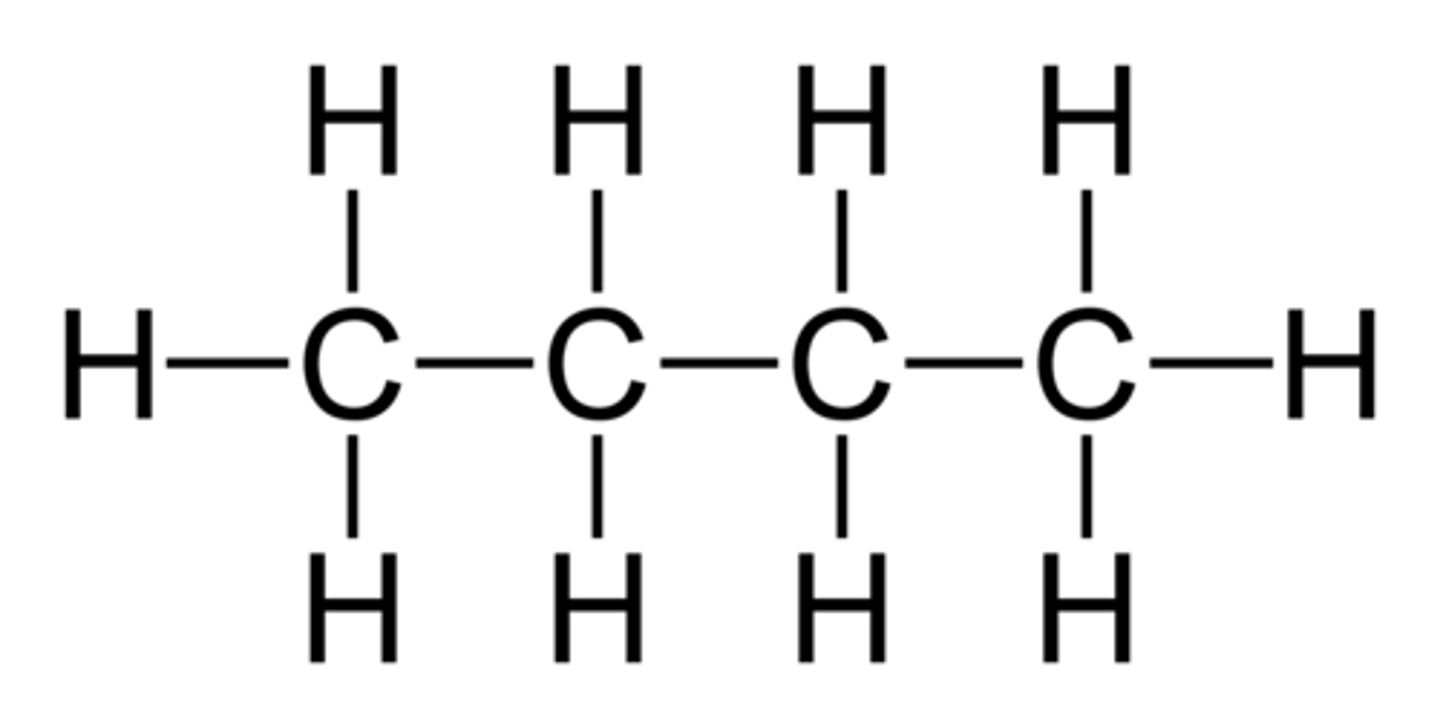
What is the displayed formula?
The relative positioning of atoms and the bonds between them
What are the different "stems" for naming organic molecules?
1 carbon - meth
2 - eth
3 - prop
4 - but
5 - pent
6 - hex
7 - hept
What is the 'stem' for naming organic side chains?
1 carbon - methyl
2 - ethyl
3 - propyl
4 - butyl
5 - pentyl
6 - hexyl
7 - heptyl
What is nomenclature?
Naming system for organic compounds
How can you classify hydrocarbons?
- Aliphatic - a hydrocarbon in the form of only straight chains, branched changed or non-aromatic rings
- Alicyclic - an aliphatic compound arranged in non-aromatic rings with or without side chains
- Aromatic - a hydrocarbon containing a benzene ring
How do you represent multiple side chains in a hydrocarbons name?
With prefix di- or tri- before the side chain e.g Dimethyl
Numbers of which carbon the side chains are bonded to before the prefix e.g. 2,3-dimethyl
If there are 2 different types of side chains it must be written in the mane alphabetically e.g. 3-ethyl-2-methyl...
What are the rules for naming hydrocarbons with halogen functional groups?
Prefixes:
- Flourine - fluoro-
- Chlorine - chloro-
- Bromine - bromo-
- Iodine - iodo-
If there are multiple halogens in one order them alphabetically e.g. 2-Bromo-2-Chlorobutane
What is a ketone and how would you name one?
Ketones = compound with a carbonyl group but it's located on a carbon in the middle/not on the end.
Suffix -one
The simplest ketone is propone as ketones require a middle group.
What is an alkane?
A hydrocarbon with only sigma covalent bond - single covalent bonds - which make alkanes quite inert.
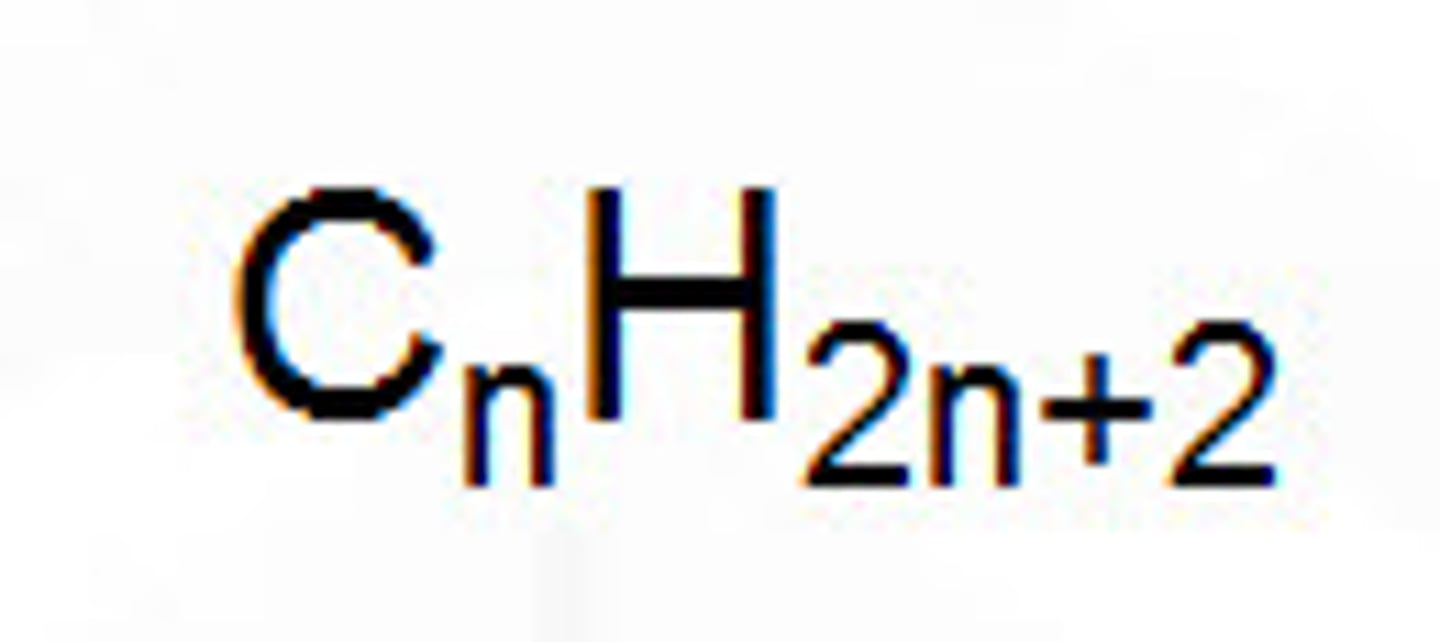
What is the relationship between carbon chain length and boiling point for alkanes?
The longer the carbon chain the higher the boiling point due to the increase in London/induced dipole-dipole forces between the molecules.
--> this is because a larger compound = more electrons in total
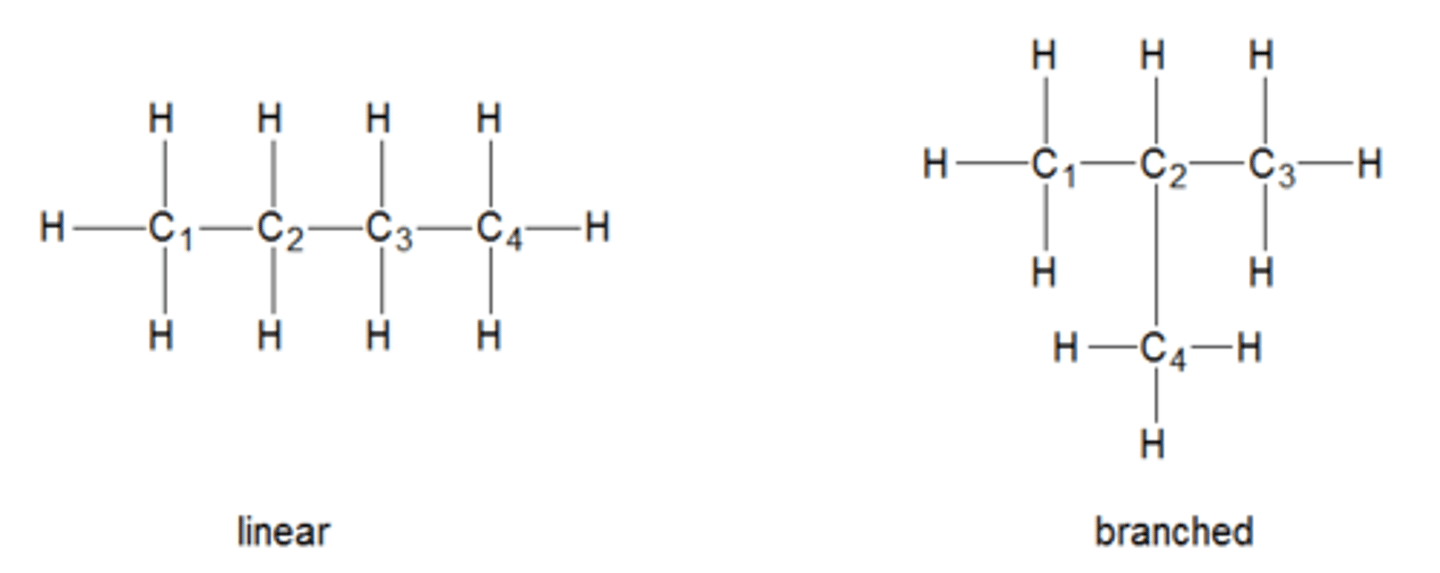
What is the relationship between alkanes being branched and boiling point?
A branched alkane isomer will have a lower boiling point than a linear alkane isomer because the London/induced dipole-dipole forces weaken.
---> this is due to the molecules not being able to have as many points of contact/ not being able to pack as close together
What is a free radical?
A molecule or atom with an unpaired electron - free radicals are very reactive because of this.
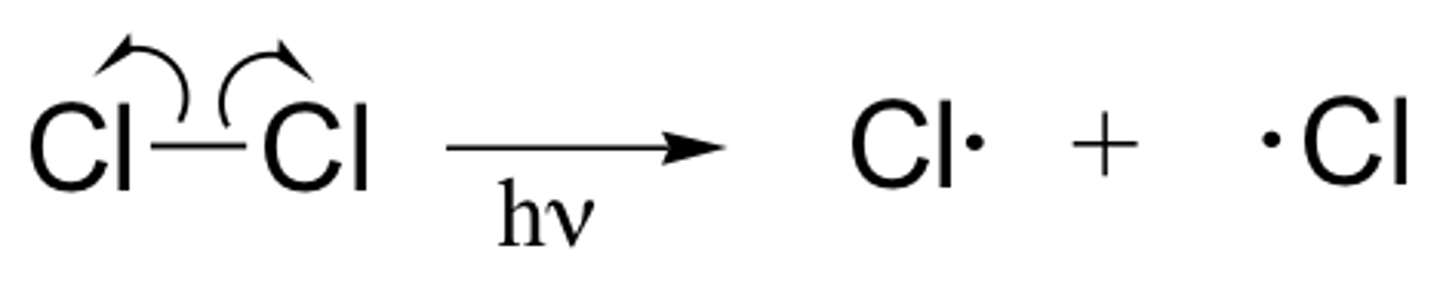
How are halogen free radicals formed?
Free radicals are formed through homolytic fission where the energy required for the electrons to be shared equally is provided from UV light.
What happens in initiation?
The chlorine molecule is split into 2 chlorine free radicals by UV light.
What happens in termination?
Any two radicals react together to form a normal molecule.
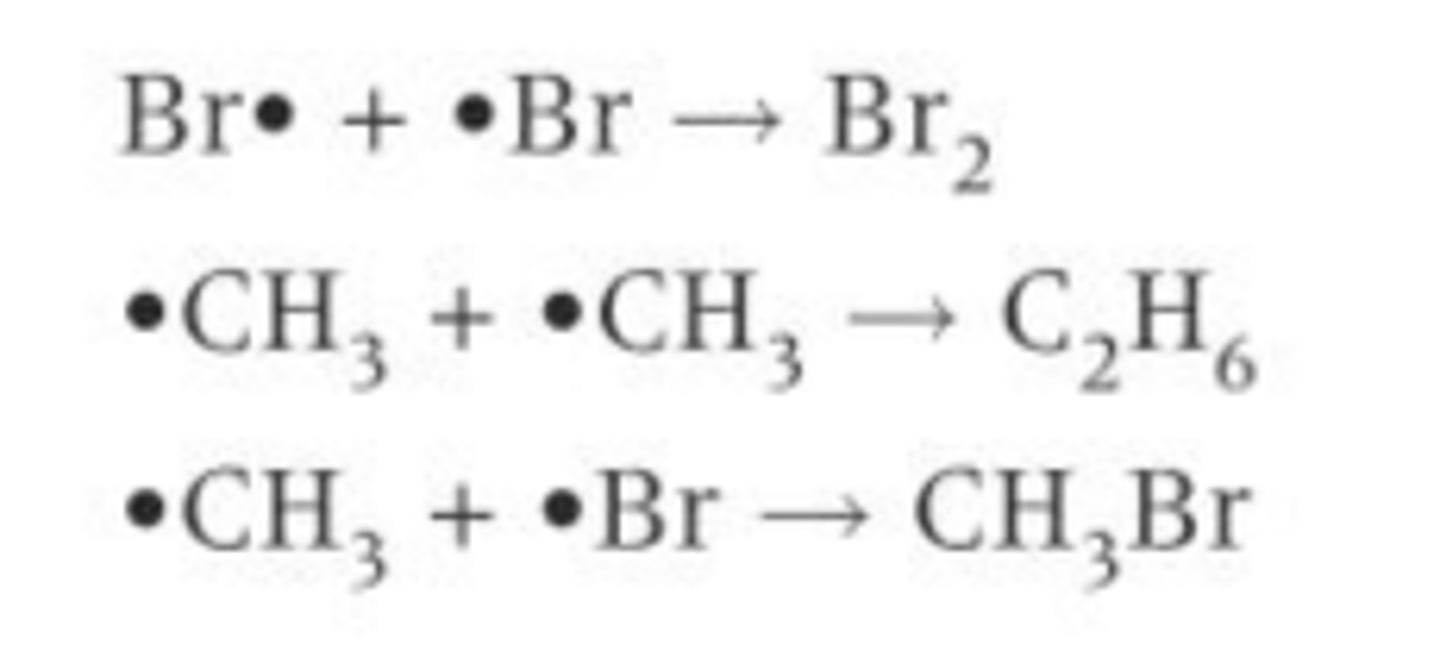
What can happen when there's an excess of a halogen in a free radical substitution reaction?
Excess halogen gives multiple substitutions to form e.g. CH2Cl2 or CHCl3 or CCl4 as the original e.g. CH3Cl can bump and react with the halogen e.g. chlorine again and replace another hydrogen with a halogen e.g. chlorine
What are the different types of structural isomerism?
Chain isomerism - isomers with different chain lengths
Positional isomerism - changing the position of the functional group
Functional group isomerism - changing the position of the atoms changes what functional group there is e.g. alderhydes vs ketones
What are the different advantages and disadvantages of the methods of halogen synthesis haloalkanes?
- Alkane ---> Haloalkane = very poor yield and no control over the number of halogens on the haloalkane or their position
- Alcohol ---> Haloalkane = slow reaction but can control position etc
- Alkene ---> Haloalkane = fast reaction and can control position of halogen so is the best method
What are the different types of reactions?
Addition - 2 reactants join together to form one product e.g. polymerisation
Substitution - an atom or group of atoms is replaced by a different atom or group of atoms e.g. free radical substitution
Elimination - a small molecule is removed from a larger molecule so one reactant forms 2 products
Why is the carbon-halogen bond in a haloalkane polar?
There is a large difference in electronegativity between carbon and any halogen with the halogen being electronegative.
This means the carbon is delta positive & the halogen is delta negative.
Since the carbon is delta positive it attracts species containing a lone pair which it can donate.
What is a nulceophile?
A nucleophile is an atom or group of atoms that is attracted an electron deficient carbon atom where it donates a pair of electrons to form a new covalent bond.
= an electron pair donor which has a lone pair of electrons
e.g. hydroxide ions, water molecules & ammonia molecules
What happens when a haloalkane goes through nucleophillic substitution?
A haloalkane reacts with a nucleophile and replaces the halogen forming a new compound with a new fuctional group.
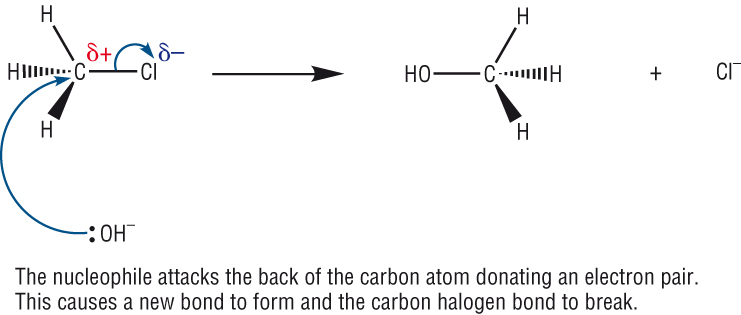
What happens in hydrolysis of haloalkanes?
Hydrolysis is a nucleophillic substitution reaction where the halogen atom is replaced by a hydroxide group :
the nucleophile OH- approaches the delta positive carbon on the other side of its bond with a halogen
this direction of attach minimises repulsion between the delta negative halogen & hydroxide anion
a lone pair of electrons on OH- is attracted and donated to the delta positive carbon
a new bond is therefore formed between the oxygen on the hydroxide and the carbon atom
the halogen-carbon bond is broken by heterolytic fission
an alcohol is made as the product as well as a halide ion
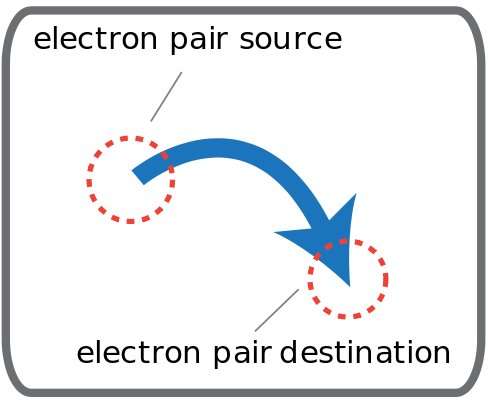
What do curly arrows show in a mechanism?
A curly arrow show the movement of electron pairs and must start from either a lone pair or bonding pair of electrons and point to the atom where the electron pair will move to.
What are the uses of different haloalkanes?
Polymerisation of tetrafluoroethene produces PTFE or Teflon which is used in non-stick pans, plumbing & GORETEX
Polymerisation of chloroethene makes PVC which is used in vinyl records & to cover copper cable as it has a high melting point and low electrical conductivity.
CHClF2 is the main HCFC used today as a refridgerant - it’s inert and volatile gas but very persistent so will destroy the ozone layer for a long time
What is ozone?
Ozone is an allotrope of oxygen - O3 instead of O2
It is naturally found in the stratosphere in the Earth’s atmosphere and there it acts as a barrier to harmful UV rays.
However when it’s found in the troposphere this can be dangerous as ozone can form photochemical smog & carcinogenic compounds.
How is ozone formed in the stratosphere?
Ozone is formed when O2 reacts with UV solar radiation with a wavelength larger than 240nm/UVc to form 2 Oxygen free radicals - homolytic bond fission.
A free radical formed can then bond to an O2 molecule forming a O3 molecule.
It can then be split into O2 + O by reacting with/absorbing UV radiation with a wavelength greater than 320 or UVb & UVa.
The O2 molecule can then be photolysed to form Oxygen radicals etc.
Why is ozone so important for the atmosphere?
The ozone in the stratosphere:
completely absorbs UVc which is the most harmful type of UV blocking it from reaching Earth as UVc is needed to form Oxygen free radicals
the interconversion process from ozone to oxygen molecules to ozone etc converts UV radiation in thermal energy as the formation of ozone is exothermic and heats the atmosphere
What happens in the natural decomposition of ozone?
O3 ——→ O2 + O radical :
It is a slow reaction that abosrbs UVc & UVb blocking this radiation from Earth.
Overall the rate of decomposition & formation is the same so the conc of ozone is constant and UVc & UVb are converted into heat.
What happens in the unnatural removal of ozone and why is this bad?
O3 can be removed by Cl* and NO* and acts as catalysts to the natural decomposition of ozone.
NO* + O3 → NO2* + O2
NO2 + O* → NO* + O2
OVERALL REACTION - O3 + O —> 2O2
This reaction is therefore much quicker and if Cl* - released by CFCs- or NO* - released by lightening & aircraft engines - are present in the stratosphere this means too much ozone is being removed and not enough is being reformed.
What is a sigma/σ bond?
A sigma bond is a type of covalent bond that is the result of 2 overlapping orbitals each containing one electron.
A σ-bond is positioned on a line directly between the bonding atoms.
In terms of organic chemistry they are single carbon bonds.
What is an alkene?
An unsaturated hydrocarbon.
Aliphatic alkenes that contain at least one carbon double bond.
Homologous series with the general formula CnH2n - only for alkenes that aren’t cyclic & contain only 1 double bond
What is a pi/π bond?
Carbon has 4 outer shell electrons which can be used to form bonds - in an alkane all 4 electrons are in sigma bonds.
In an alkene only 3 are leaving one electron in a p orbital uninvolved in any sigma bonds.
This electron is able to form a pi bond by overlapping its orbital sideways with another electron in a p orbital. The π bond’s electron density is concentrated either below or above the straight sigma bond.
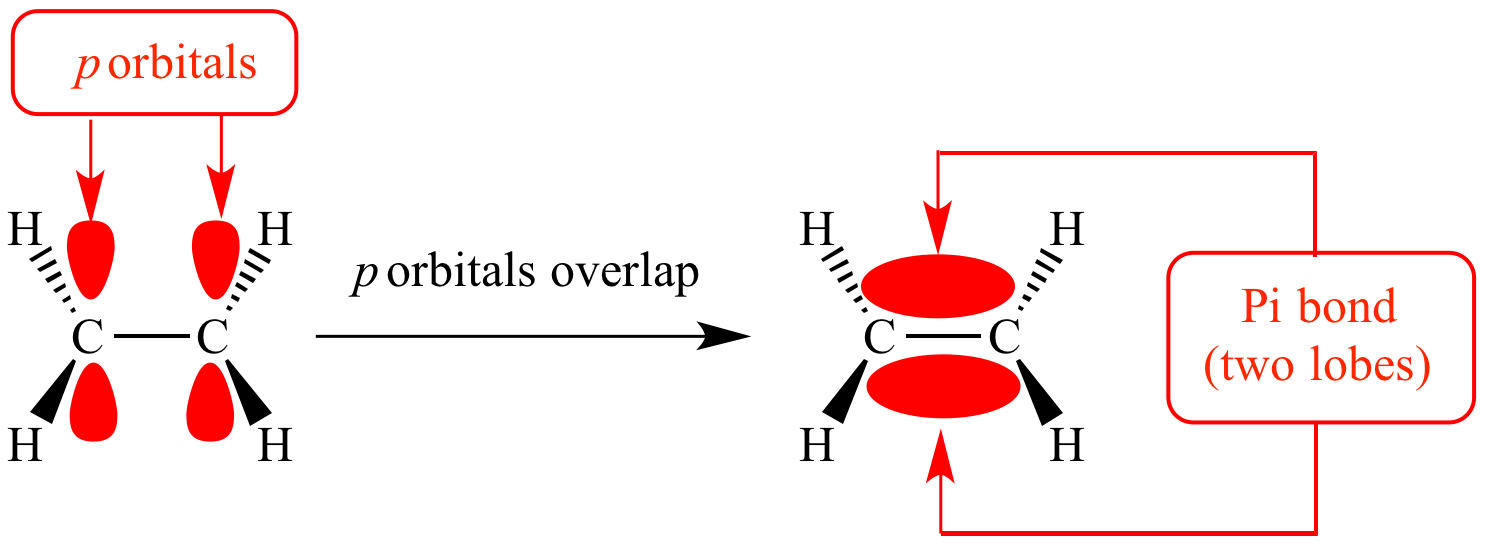
How does having a pi bond effect an alkene?
The pi bond limits the geometry of an alkene as the carbon atoms involved in the double/pi bond are unable to freely rotate around each other- the pi bond “locks” the 2 carbons in position.
The shape around each carbon involved in the double bond is trigonal planar.
Alkenes are more reactive compared to alkanes due to the double bond.
What happens when a double carbon bond breaks?
The pi bond breaks but the sigma bond doesn’t break.
What are the properties of alkenes?
Insoluble in water - alkenes have no polar bonding so are non-polar molecules therefore they will only dissolve in non-polar solvents
Alkene boiling point increases with chain length - longer chains have more points of contact so more London forces etc
Lower boiling points than their equivalent alkane - alkenes are less able to pack closely together due to lack of free rotation around double bond so fewer London forces & effect of less total electrons
What are stereoisomers?
Compounds with the same structural formula but a different arrangement of atoms in space.
This means the groups bonded to each carbon in a double bond are fixed in position relative to each other.
Why does stereoisomerism occur?
The restricted rotation around a double bond due to the pi bond.
What is E-Z isomerism?
A type of stereoisomerism that occurs when :
there is a a carbon double bond in a compound
there are different groups attached to each carbon atom in the double bond - the carbons can have the same different groups
What is the basic difference between an E stereoisomer compared to a Z stereoisomer?
E - the different groups are on opposite sides of the sigma bond/ one below & one above
Z - zame zide - the different groups are on the same side of the sigma bond
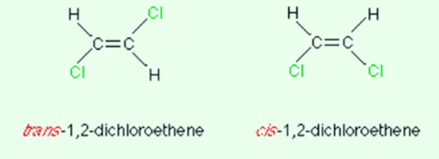
What is the proper definition of E isomers vs Z isomers in terms of priority groups?
According to Cahn-Ingold-Prelog rules all the atoms attached to each carbon in the C=C bond are given a priority based on atomic number :
E.g. Carbon > Hydrogen in terms of priority because 12>1
E isomer - the groups of higher priority are on the opposite side of the molecule
Z isomer - the groups of higher priority are on the same side of the molecule
What is cis-trans isomerism and how is it different to normal E/Z isomerism?
Cis-trans isomerism is a special type of E/Z isomerism so all cis-trans isomers are E/Z isomers but not all E/Z isomers are cis-trans isomers as well.
All the criteria for a cis-trans isomer is the same for an E/Z isomer but one group on each carbon atom must be the same - or else it isn’t a cis-trans isomer.
Why are alkenes more reactive than alkanes?
The presence of an electron dense area - the pi bond - which have pi electrons that are more exposed allows for alkenes to undergo reactions more easily.
What reactions do alkene mainly undergo?
Addition reactions.
What are all the different types of addition reactions that alkenes undergo & the conditions needed?
Hydrogenation = alkene + hydrogen —> alkane
423K & a nickel catalyst
Halogenation = alkene + halogen —> dihaloalkane
room temperature
Reaction with an hydrogen halide = alkene + hydrogen halide —> haloalkane
Hydration = alkene + steam —> alcohol
must be steam not water, in the presence of a phosphoric acid catalyst & at 300 degrees celsius & 60atm
Why are different conditions needed for a hydration reaction depending on how many double bonds there are?
Since hydrogen is added across a double bond the more double bonds there are the more hydrogen is needed for all of the bonds to be broken.
Aka. for a diene to be completely converted to an alkane an excess of hydrogen is required.
What is the mechanism that allows for addition reactions to happen with alkenes?
Electrophillic addition.
What is an electrophile?
An electron pair acceptor/ an atom or groups of atoms that is attracted to an electron dense area e.g. a Carbon double bond.
they are usually positive ions or a molecule containing a delta positive sign
How can electrophiles be produced?
Heterolytic bond fission forms cations & so produces the elcetrophiles needed in electrophilic addition.
In which addition reactions is electrophilic addition used by alkenes?
Addition reaction with halogens to form a dihaloalkane.
Addition reaction with hydrogen halides to form a (mono)haloalkane.
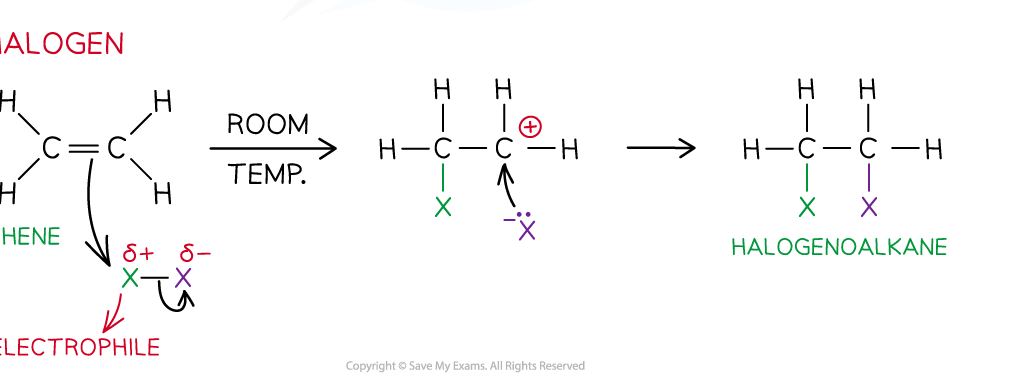
What is the process of electrophilic addition in the halogenation of an alkene?
Bromine molecule isn’t already an electrophile but once it approaches the C=C double bond it gets polarised - so there is a delta positive Bromine
Electron pair in pi bond is attracted to delta positive Bromine & the electrons are transferred
This causes the C=C bond to break & the Br — Br bond to break as well via heterolytic fission
A carbocation intermediate is formed which is very reactive
Negative Bromine formed via heterolytic fission bonds to positive carbon on the carbocation
A dihaloalkane is formed
How is the process of electrophilic addition with a hydrogen halide different to an electrophilic addition with a halogen?
The process is exactly the same but H — Br is already polar with a delta positive H & delta negative Br so the H will always be the one to bond first.
With the halogenation either Bromine could become the delta positive one.
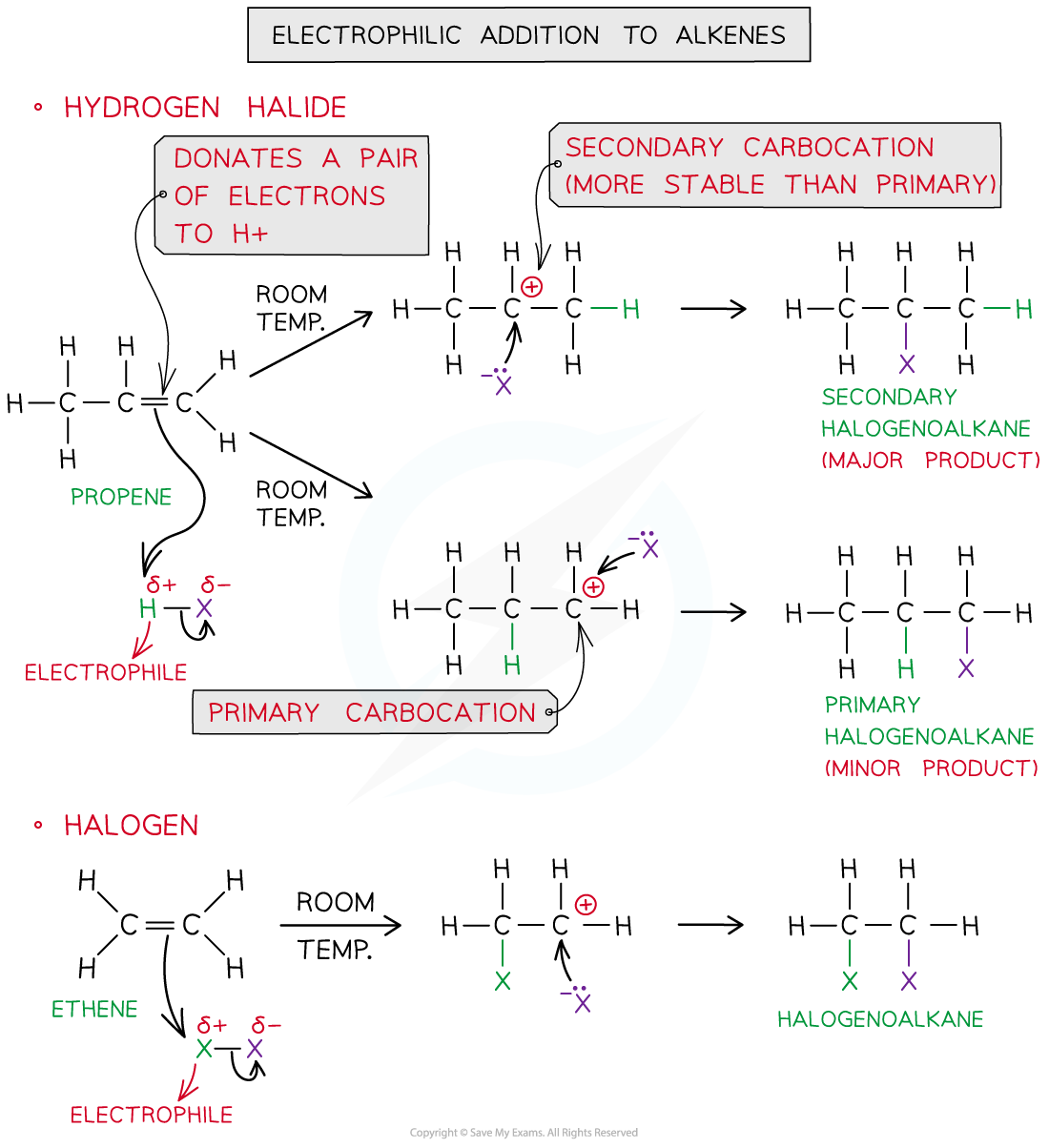
Why can there be two different products formed from one electrophilic addition reaction with a hydrogen halide + alkene?
If the alkene is unsymmetrical reacting with a hydrogen halide can form 2 different products.
An unsymmetrical alkene doesn’t have the same groups bonded to each carbon in the double bond e.g. propene vs ethene.
What are the reaction conditions required to required to convert an alkene to an alkane?
Alkene + Hydrogen —> Alkane
Requires 423K & a nickel catalyst
What is Markownikoff’s Rule?
When a hydrogen halide reacts with an unsymmetrical alkene, hydrogen will attach to the carbon with a greater number of hydrogens
This leads to the formation of a major & minor product with the yield of the major product being higher
What effects the formation of a major or minor product in electrophilic addition of alkenes?
It depends on how stable the carbocation intermediate is - the major product will form via the most stable carbocation.
A mixture will still always be formed
In what categories are carbocations classified as?
Primary - positive carbon is bonded to 1 other carbon/alkyl group
Secondary - positive carbon is bonded to 2 other carbon/alkyl groups
Tertiary - positive carbon is bonded to 3 other carbon/alkyl groups
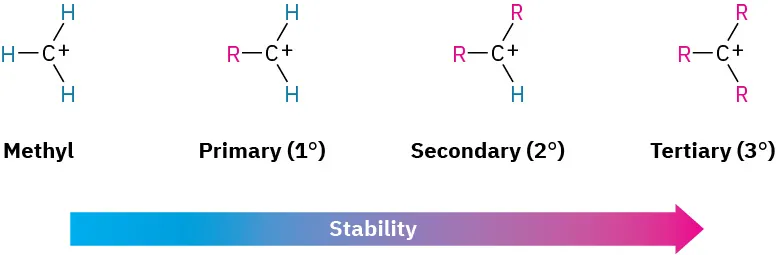
Why are tertiary carbocations more stable than primary carbocations?
The stability is linked to the electron-donating ability of the alkyl groups.
Carbon is unstable when positive but the alkyl groups push electrons towards it so it’s less positive making the molecule overall more positive
This means the greater the number of alkyl groups the more stable the carbocation is.
What is addition polymerisation?
A reaction between unsaturated organic products to form saturated polymer chains.
What conditions does addition polymerisation require?
High temperature
High pressure
A catalyst
This is in general but conditions for addition polymerisation change depending on the alkene used and the polymer you want produced as multiple polymers can be formed from the same alkene depending on the conditions.
How do you draw out addition polymerisation?
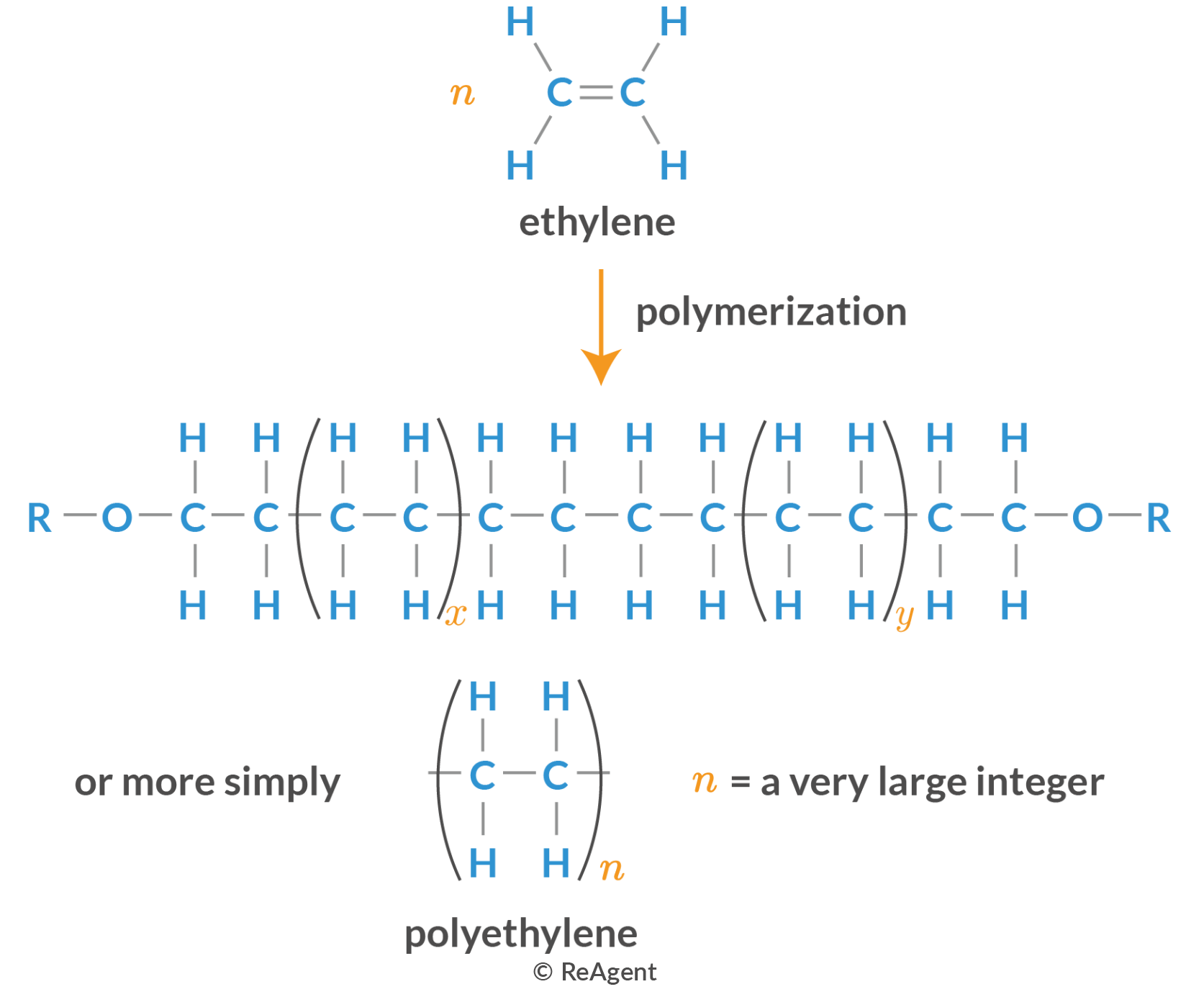
What are some additions polymers and their uses?
Polyethene - straws
Polypropene - toys & guttering
Polystyrene - packing material
PTFE - non-stick coatings
Why are polymers hard to dispose of?
Lack of reactivity makes them suitable for safe food & chemical storage but it means they can’t be biodegraded & instead have to be burnt or recycled.
These come with their own problems as incineration can release toxic gases & recycling requires sorting of plastics.
How does recycling polymers reduce their environmental impact?
Finite fossil fuels are conserved
Amount going to landfill is reduced - reduced methane released
What are the (general) steps for recycling a polymer?
Plastics must be sorted by type
Once sorted they are chopped into flakes
Washed & dried
Melted
Chopped into pellets & then used to make new products
How is recycling PVC different to recycling plastics in general?
Recycling PVC is hazardous due to the high Chlorine content & additives present.
PVC can be burnt but the HCl gas & other pollutants produced have to be removed by passing the gas through an alkali solution.
PVC can also now be recycled through using solvents to dissolve the polymer so high grade PVC can be recovered through precipitation from the solvent + the solvent can be reused.
Why are some polymers incinerated instead of recycled?
They are too hard to be recycled so are instead incinerated to produce heat generating electricity.
What is feedstock recycling?
Processes that reclaim monomers, gases or oil from waste polymers
Unlike in recycling polymers can be unsorted
The products resemble the original ones from crude oil so close the loop
What are bioplastics?
Plastics produced from plant starch, cellulose, plant oils & proteins
Offer a renewable & sustainable alternative
Protects planet from non-biodegradeable polymers & conserves finite fossil fuels
Broken down by microorganisms to make water etc
Many bioplastics contain additives that alter the polymer structure to make this easier
What are photodegradeable polymers?
Polymers that have bonds that are weakened by absorbing light to begin degredation
Can be used when plant-based polymers aren’t suitable
What are alcohols?
Alcohols are a homologous series that contain a hydroxyl group -OH.
They are often used as chemical feedstock & solvents.
How are alcohols classified?
They are classified by the number of alkyl groups bonded to the carbon which is bonded to the hydroxyl group and so can be:
Primary (1°) - 1 alkyl group
Secondary (2°) - 2 alkyl groups
Tertiary ( 3°) - 3 alkyl groups
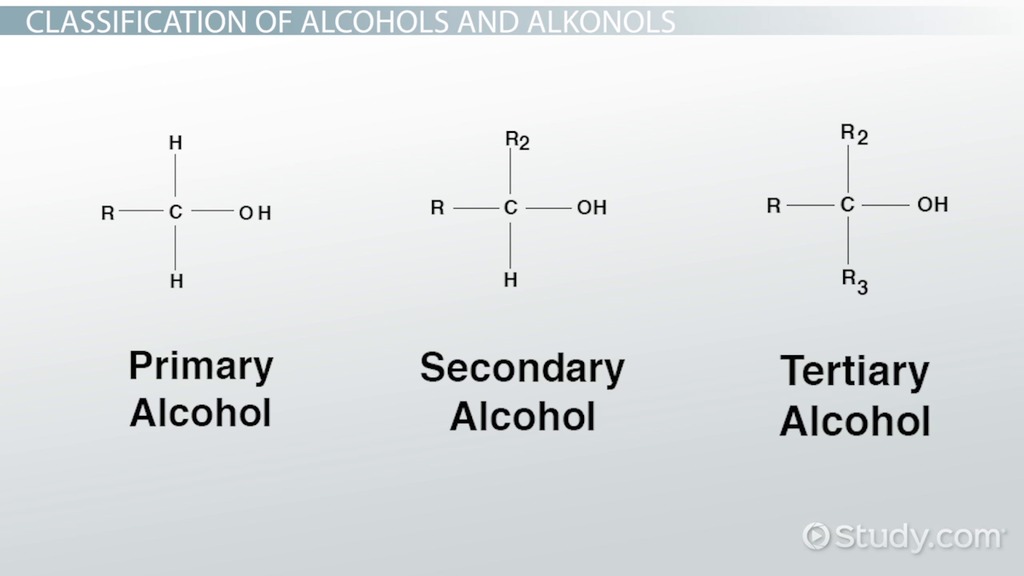
What are the differences between alcohols and their corresponding alkane e.g. ethanol & ethane?
Alcohols are less volatile
They have higher melting points
They have greater water solubilty
However, these differences become less significant with the longer chain alcohols & corresponding alkenes.
Why are there differences between alcohols & their corresponding alkanes?
Alkanes are non-polar as they only have weaker non-polar bonds - London forces
Alcohols are polar because of the difference in electronegativity between O & H and C & O which creates a polar bond - stronger permanent induced dipole-dipole interactions & hydrogen bonding
This means more energy is required to overcome the hydrogen bonding than the London forces - higher boiling points & lower volatility
Water is polar and like dissolves in like as water forms hydrogen bonds with the -OH on the alcohols - greater water solubility
Why are alcohols more reactive than their alkane counterparts?
The presence of polar O-H and C-O bonds.
What reactions can alcohols undergo?
Combustion
Oxidation
Elimination/dehydration
Subsitution
What happens in a combustion reaction with alcohols?
All alcohols can burn completely in excess O2 to produce CO2 & H2O (duh)
As the length of the carbon chain increases the quantity of heat & moles produced increases