Crystal Structures and Defects
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
22 Terms
Crystalline (smal scale)
atoms/molecules are arranged in a regular array that repeats (fixed bond lengths and angles) - in the solid state, metals and many ceramics have crystalline structures under normal conditions
Amorphous (small scale)
atoms'/molecules have an irregular arrangement to each other (bond lengths and angles may be distorted) and there is no long range order
What are some large scale SOLID material structures?
single crystal, amorphous, semi-crystalline, polycrystalline
single crystal
whole material has one arrangement of atoms e.g silicon
amorphous (large scale)
whole material is amorphous (atoms/molecules have an irregular arrangment)
semi-crystalline
material has crystalline regions and amorphous regions
polycrystalline
material is composed of many small single crystal regions (grains) that are oriented at random to each other
what is a unit cell?
the smallest segment that can be repeated in three dimensions
What can polymorphic materials do?
Adopt more than one crystal structure
What can allotropic elements do?
adopt different crystal structures at different temperatures and pressures
What are some examples of metallic crystal structures?
face centred cubic, body centred cubic and hexagonal close packed
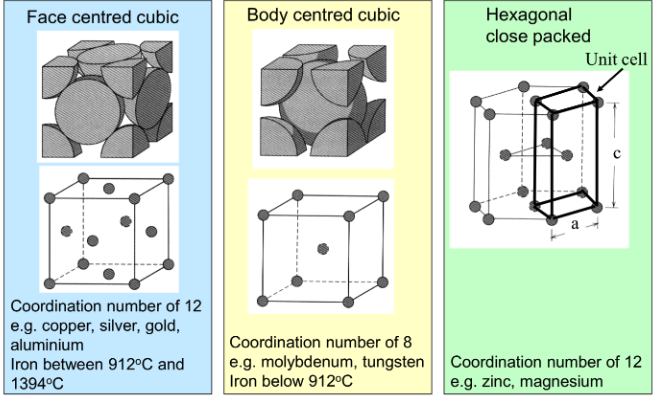
What factors affect material strength?
bonding, grain boundaries and defects
What are grain boundaries?
regions of crystal planes that don’t match (transition is a few atomic layers wide), these have higher energy than the bulk of the material due to the irregular bond lengths and coordination numbers of the atoms
What are vacancies?
hole formed at a specific location in the crystal structure due to the absence of an atom - also known as a point defect
how do vacancies arise?
as a result of solidification or deformation, higher temperature = more vacancies,
what are dislocations?
defects that involve planes of atoms, two types are screw and edge or a combination
how are edge dislocations formed?
an extra, incomplete plane of atoms in the crystal structure
what is an edge dislocation slip?
dislocation moves across atom rows due to shear stress, atoms break bonds and form new ones (this can continue until the dislocation moves to the crystal edge)
How are dislocation slips relevant in metals?
the metallic bond enables dislocations to slip relatively easily past each toher hence metals are ductile
How are dislocation slips relevant in ionic compounds?
for slips to occur, ions with like charge must be forced past each other hence ionic compounds are brittle
How are dislocation slips relevant in covalent compounds?
the bonds are strong and very directional, it’s difficult for atom planes to slide past each other hence covalent compounds are brittle
When are alloys weakest?
When the different elements making them up are similar size