Natures chemistry flashcards
1/171
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
172 Terms
How is 'an alcohol whose -OH group is attached to a carbon attached to no other hydrogens' described?
tertiary
Fehlings solution changes colour from blue to which colour?
brick red
What are tertiary alcohols oxidised to?
They cannot be oxidised
What type of compound can be used to prevent non-polar and polar substances from separating into layers?
emulsifier
Which group is circled in the diagram?
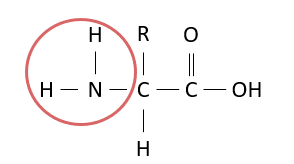
amino
How is 'an alcohol whose -OH group is attached to a carbon attached to 2 other hydrogens' described?
primary
Essential amino acids are amino acids which must be acquired from the ____.
Identify the missing term.
diet
What are major structural materials of animal tissue?
protein
What are molecules that prevent unwanted oxidation?
antioxidants - free radical scavengers
Oils have a lower melting point because they are more __________. Identify the missing term.
unsaturated
What can be defined as 'compounds with the same molecular formula, but different structural formulae'?
isomers
Which oxidising agent changes colour from blue to brick red?
fehlings solution
What can be described as 'molecules that react with free radicals to form stable molecules' ?
free radical scavenger
Many flavour and aroma molecules are __________.
Identify the missing term.
aldehydes
Name the salt that is produced when potassium hydroxide reacts with butanoic acid.
potassium butanoate
Name the following carboxylic acid.
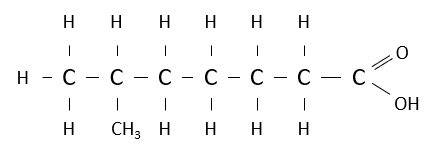
6-methylheptatanoic acid
Metal carbonate + acid --> salt + water + ______________. Identify the missing term.
carbon dioxide
Name the following carboxylic acid.
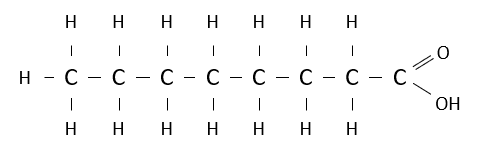
octanoic acid
Hot copper (II) oxide changes from black to which colour?
brown
The volatility of a compound is affected by the ____________ forces present. Identify the missing term.
intermolecular
Which type of addition reaction is shown below?
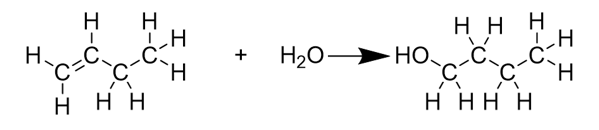
hydration (adding water)
What type of compounds will have no reaction with bromine water?
saturated (alkane/single bond)
What type of compounds will quickly decolourise bromine water?
unsaturated (alkenes/double bond)
A hydration reaction involves the addition of what to an alkene?
water
As alkanes increase in molecular size, their melting point also increases. This is due to the presence of which intermolecular force?
London dispersion forces
What happens to the melting point as the size of a compound increases?
it increases
A polar compound will be soluble in __________ compounds. Identify the missing term.
polar
Which term can be used to describe the following compounds?
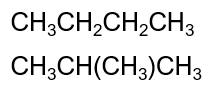
isomers
A hydrogenation reaction involves the addition of what to an alkene?
hydrogen
How can alkanes be described in terms of saturation?
saturated
How can the molecule in the diagram be described in terms of saturation?
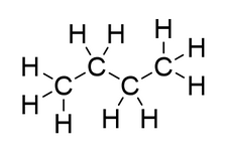
saturated
What will happen to bromine water if an alkene is added to it?
quickly decolourises
How can alkenes be described in terms of saturation?
unsaturated
What will happen to bromine water if an alkane is added to it?
nothing
What type of alcohol is shown: primary, secondary or tertiary?
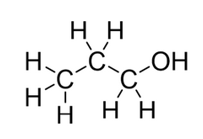
primary
Name the following alcohol.
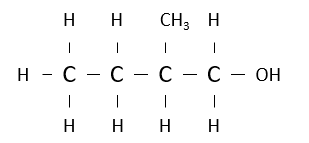
2-methylbutan-1-ol
The number of hydroxyl groups on an alcohol affects its melting point. This is due to a greater degree of ___________ _________. Identify the missing term.
hydrogen bonding
What is the name of the functional group found in an alcohol?
hydroxyl
What type of alcohol is shown: primary, secondary or tertiary?
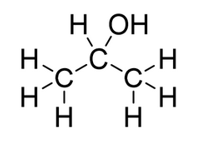
secondary
Name the following alcohol.
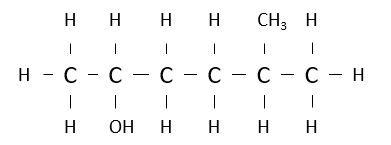
5-methylhexan-2-ol
Name the following alcohol.
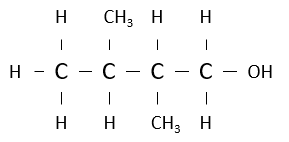
2,3-dimethylbutan-1-ol
Name the following alcohol.
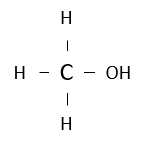
methanol
Alcohols with more hydroxyl groups will have a ______boiling point. Identify the missing term.
higher
How would this alcohol be described?
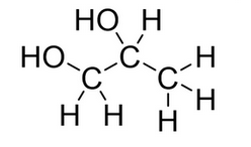
diol
Name the following alcohol.
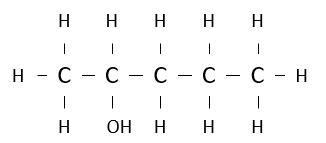
pentan-2-ol
Name the following alcohol.
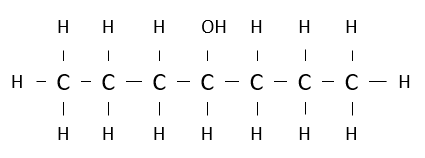
heptan-4-ol
What type of alcohol is shown: primary, secondary or tertiary?
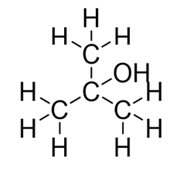
tertiary
How would this alcohol be described?
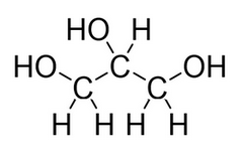
triol
How is 'an alcohol whose -OH group is attached to a carbon attached to 1 other hydrogen' described?
secondary
Name the following alcohol.
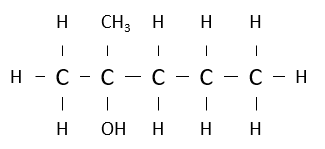
2-methylpentan-2-ol
Name the following carboxylic acid.
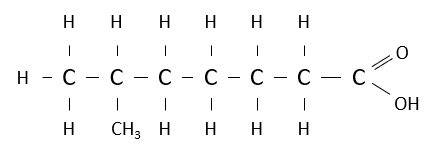
6-methylheptanoic acid
Metal carbonate + acid --> salt + water + ______________. Identify the missing term.
carbon dioxide
What is the name of the functional group found in a carboxylic acid?
carboxyl
Name the following carboxylic acid.
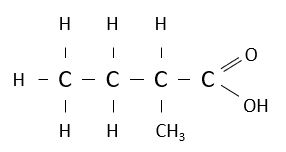
2-methylbutanoic acid
Name the salt that is produced when lithium carbonate reacts with ethanoic acid.
lithium ethanoate
Name the following carboxylic acid.
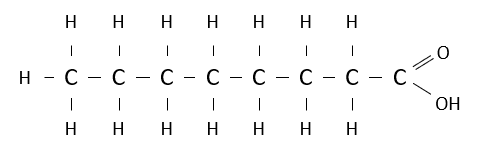
octanoic acid
Name the salt that is produced when potassium hydroxide reacts with butanoic acid.
potassium butanoate
Name the following carboxylic acid.
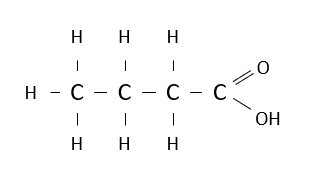
butanoic acid
metal oxide + acid --> water + ____. Identify the missing term.
salt
Name the salt that is produced when sodium oxide reacts with propanoic acid.
sodium propanoate
Name the following carboxylic acid.
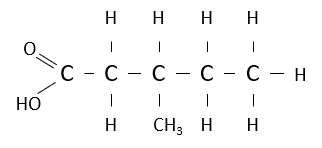
3-methylpentanoic acid
Name the following carboxylic acid.
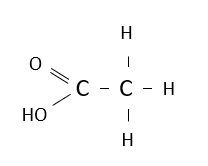
ethanoic acid
Name the following ester.
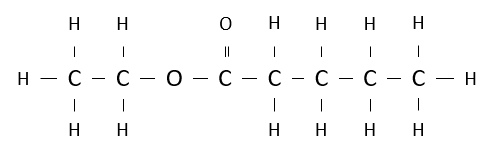
ethyl pentanoate
What is the name of the reaction that forms esters from an alcohol and a carboxylic acid?
condensation
Name the reaction in the diagram.
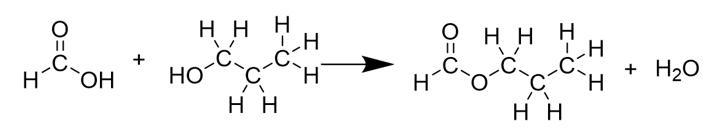
condensation
What is the carboxylic acid that is produced during hydrolysis of ethyl hexanoate?
hexanoic acid
Name the following ester.
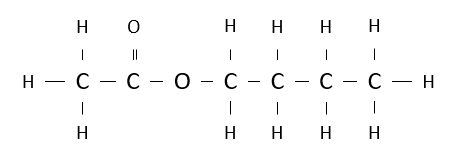
butyl ethanoate
The higher saturation of fats means they are more tightly packed together. This causes stronger_____________ _________. Identify the missing term.
intermolecular forces
Which has the higher melting point? Oils or Fats
Fats
Which chemical is used to test for saturation?
bromine water
Fats have a higher melting point because they are more __________. Identify the missing term.
saturated
Esters are used as solvents, fragrances and ________. Identify the missing term.
flavourings
What type of compound will quickly decolourise bromine water?
unsaturated
Name the carboxylic acid that is used in the production of butyl pentanoate.
pentanoic acid
During a condensation reaction an ester is formed, and what else?
water
Name the alcohol that is used in the production of methyl ethanoate.
methanol
How many carboxylic acids are used to produce edible fats and oils?
3
Name the alcohol which is used to make edible fats and oils.
glycerol / propane-1,2,3-triol
Which reaction makes edible fats and oils from glycerol and three carboxylic acid molecules?
condensation
What is the alcohol that is produced during hydrolysis of pentyl butanoate?
pentanol
Name the functional group.
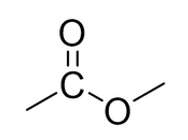
ester link
Which process is defined as ' the breaking apart of a large molecule into to smaller molecules by the adding water' ?
hydrolysis
Name the reaction in the diagram.
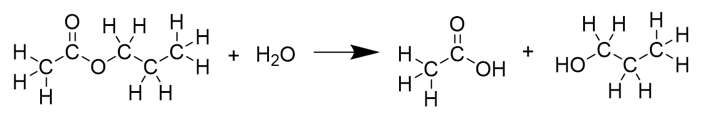
hydrolysis
Oils have a lower melting point because they are more __________. Identify the missing term.
unsaturated
Fats and oils are needed because they are a concentrated source of ______ and essential for the transport and storage of fat-soluble vitamins.
Identify the missing term.
energy
Name the following ester.
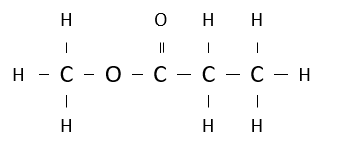
methyl propanoate
Name the following ester.
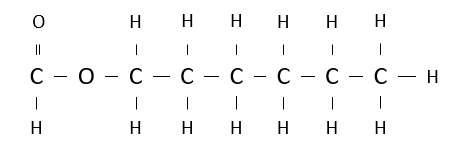
hexyl methanoate
What is the name of the reaction that breaks esters into alcohols and carboxylic acids?
hydrolysis
What term is given to substances with non-polar hydrophobic tails and ionic hydrophilic heads which act in the same way as soaps but don't produce scum in hard water?
detergents
Soaps are made from the alkaline hydrolysis of edible ____ and oils.
Identify the missing term.
fats
How is the tail of a soap described?
hydrophobic / non-polar
Soaps are made from the alkaline hydrolysis of edible fats and ____.
Identify the missing term.
oils
An ________ contains small droplets of one liquid dispersed in another liquid.
Identify the missing term.
emulsifier
Emulsifiers for use in food can be made by reacting edible oils with ___________.
Identify the missing term.
glycerol
Soaps are used to remove __________ substances such as grease and oil.
Identify the missing term.
non-polar
How is the head of a soap described?
hydrophilic / polar
Hard water is water with high levels of ______ ions.
metal
What type of compound can be used to prevent non-polar and polar substances from separating into layers?
emulsifier
What is formed when soap is used in areas of hard water?
scum / insoluble precipitate
Name the reaction that produces soaps from edible fats and oils.
alkaline hydrolysis