AP Biology Review
1/559
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
560 Terms
Elements
Elements are substances that cannot be broken down into simpler substances by chemical means.
Oxygen
Oxygen is an element with symbol O and atomic number 8. It is a highly reactive nonmetal and oxidizing agent. In living organisms, oxygen is used in respiration and in a number of organic molecules.

Carbon
Carbon is an non mental element with symbol O and atomic number 6. It is the second most abundant element in living organisms and is present in all organic compounds.

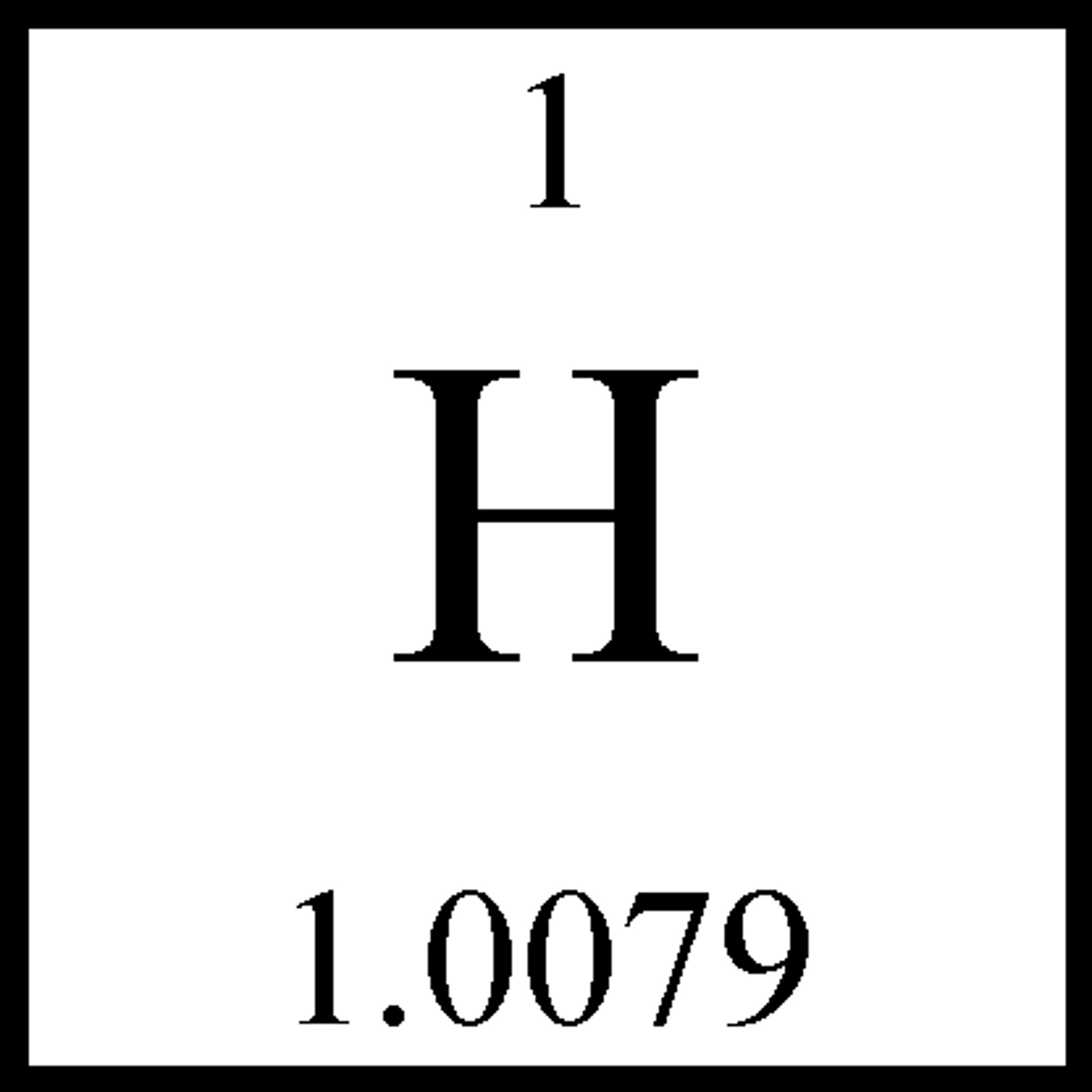
Hydrogen
Hydrogen is an element with symbol H and atomic number 1. Hydrogen combines with non-metallic elements to form water and other organic compounds.
Nitrogen
Nitrogen is an element with the symbol N and atomic number 7. In biology, nitrogen is important as it is found in a number of organic compounds and is used in fertilizers and antibiotics.

Trace elements
Trace elements are elements required by an organism in very small quantities. Trace elements include iron, iodine, and copper.
Atom
An atom is the smallest unit of an element that retains its characteristic properties; they are the building blocks of the physical world.
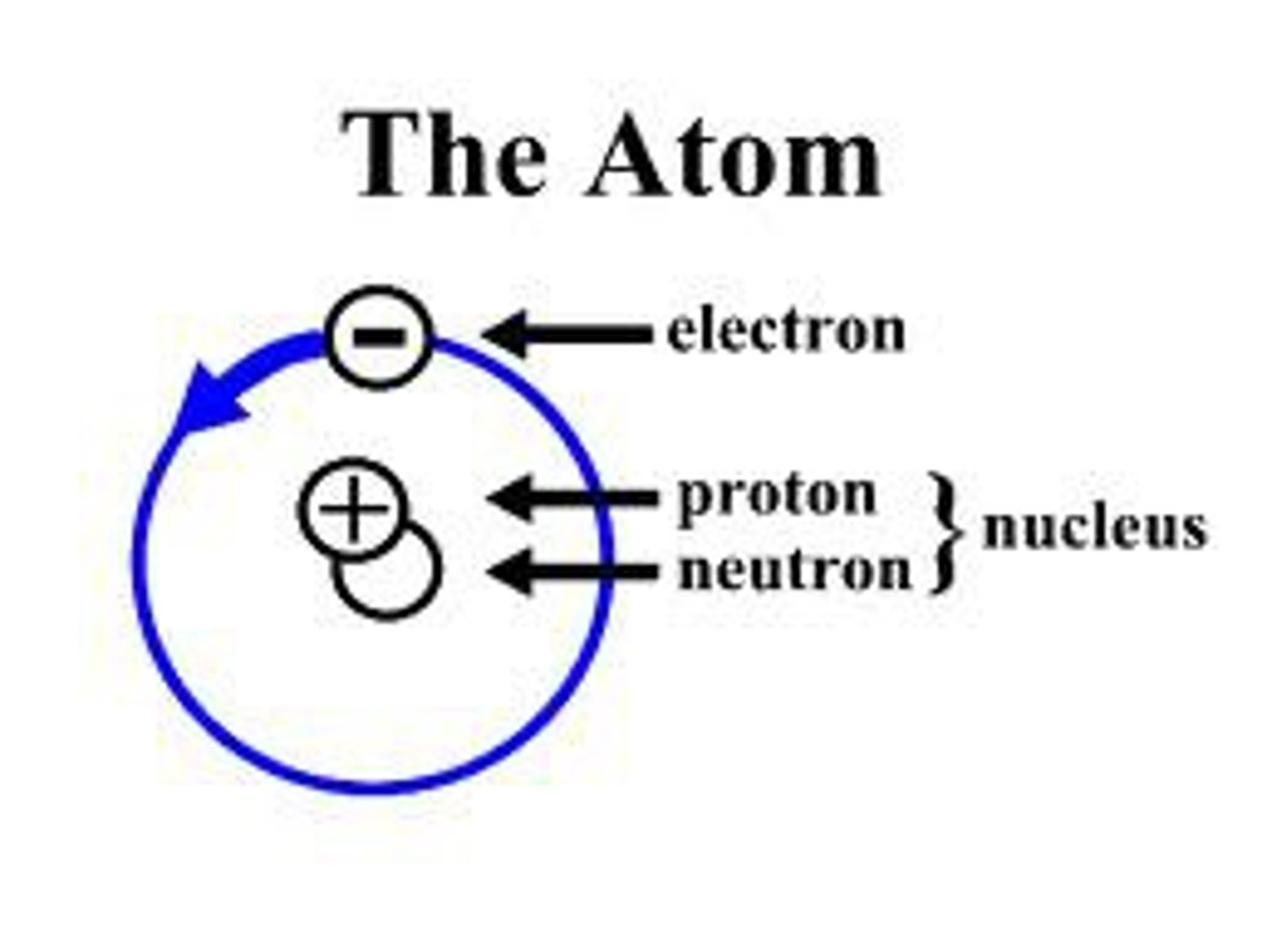
Protons
Protons are positively charged subatomic particles that are found in an atom's nucleus.

Neutrons
Neutrons are uncharged subatomic particles found in an atom's nucleus.

Electrons
Electrons are negatively charged subatomic particles that spin around the nucleus.
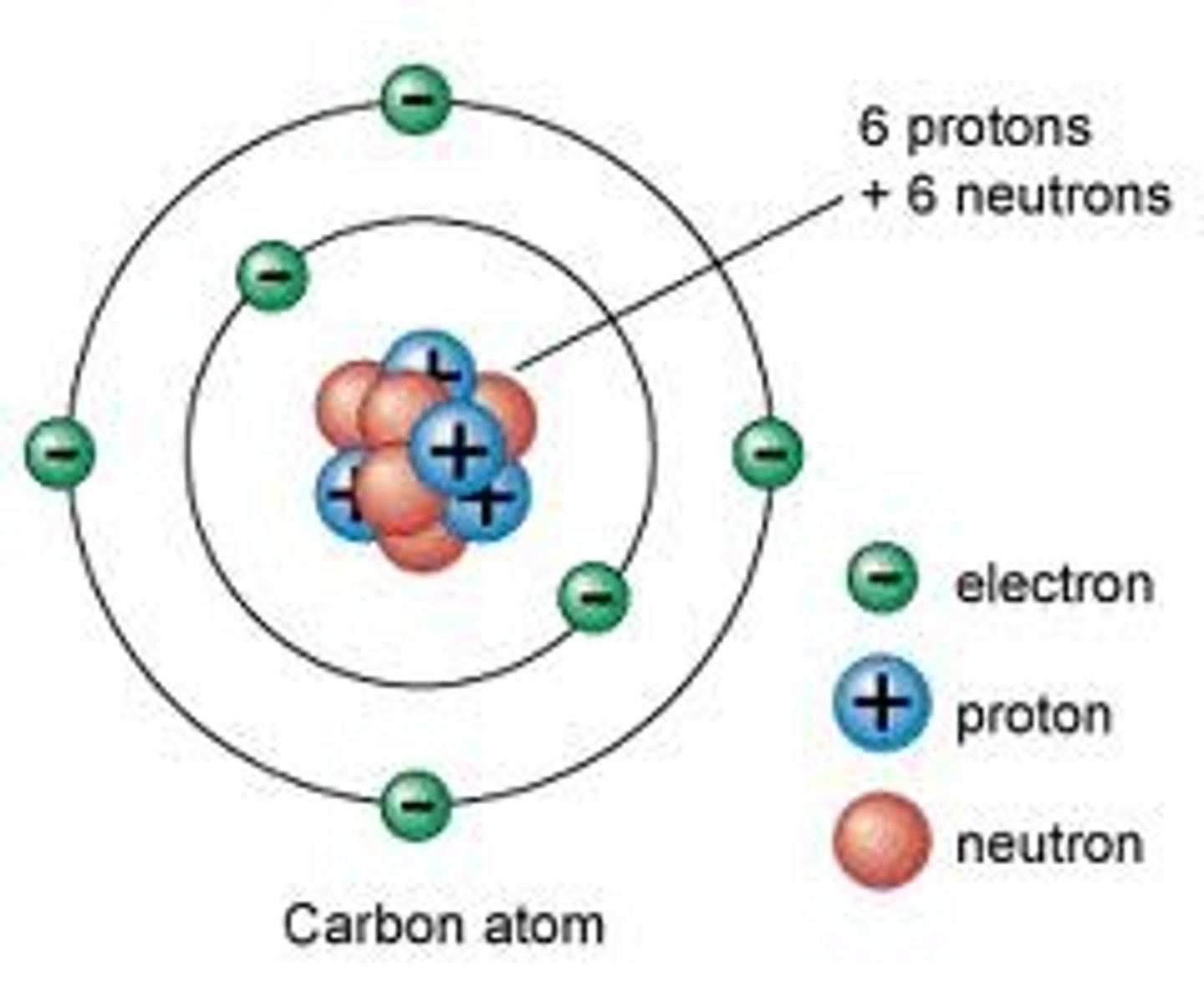
Nucleus
The nucleus is the positively charged core in an atom made up of neutrons and protons.
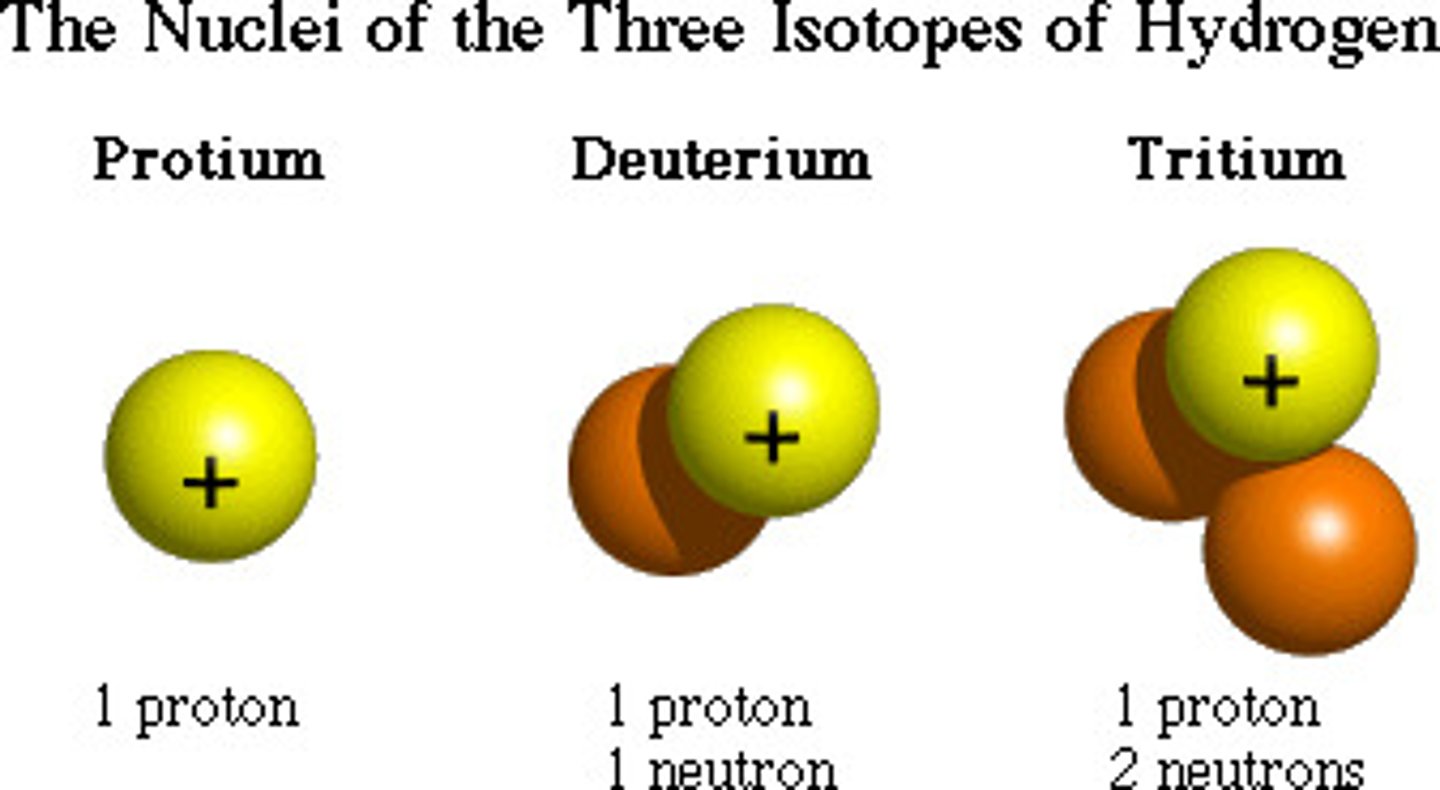
Isotopes
Atoms that have the same number of protons but differ in the number of neutrons in the nucleus are called isotopes.
Compound
A chemical compound is formed when two or more different types of atoms are combined in a fixed ration.
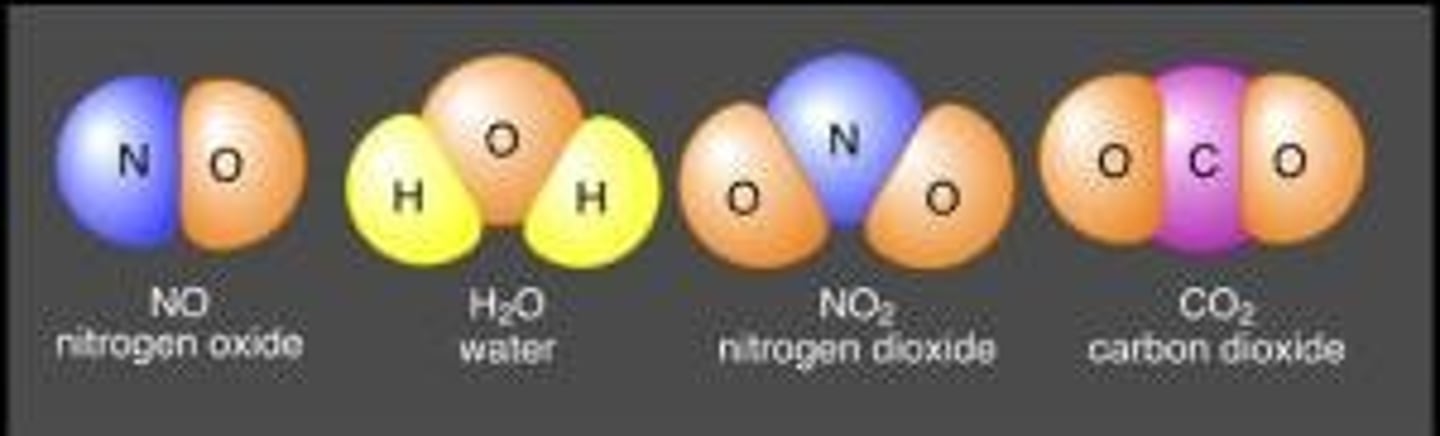
Chemical reaction
A chemical reaction describes a chemical change in which reactants react to form products chemically different from the reactants.
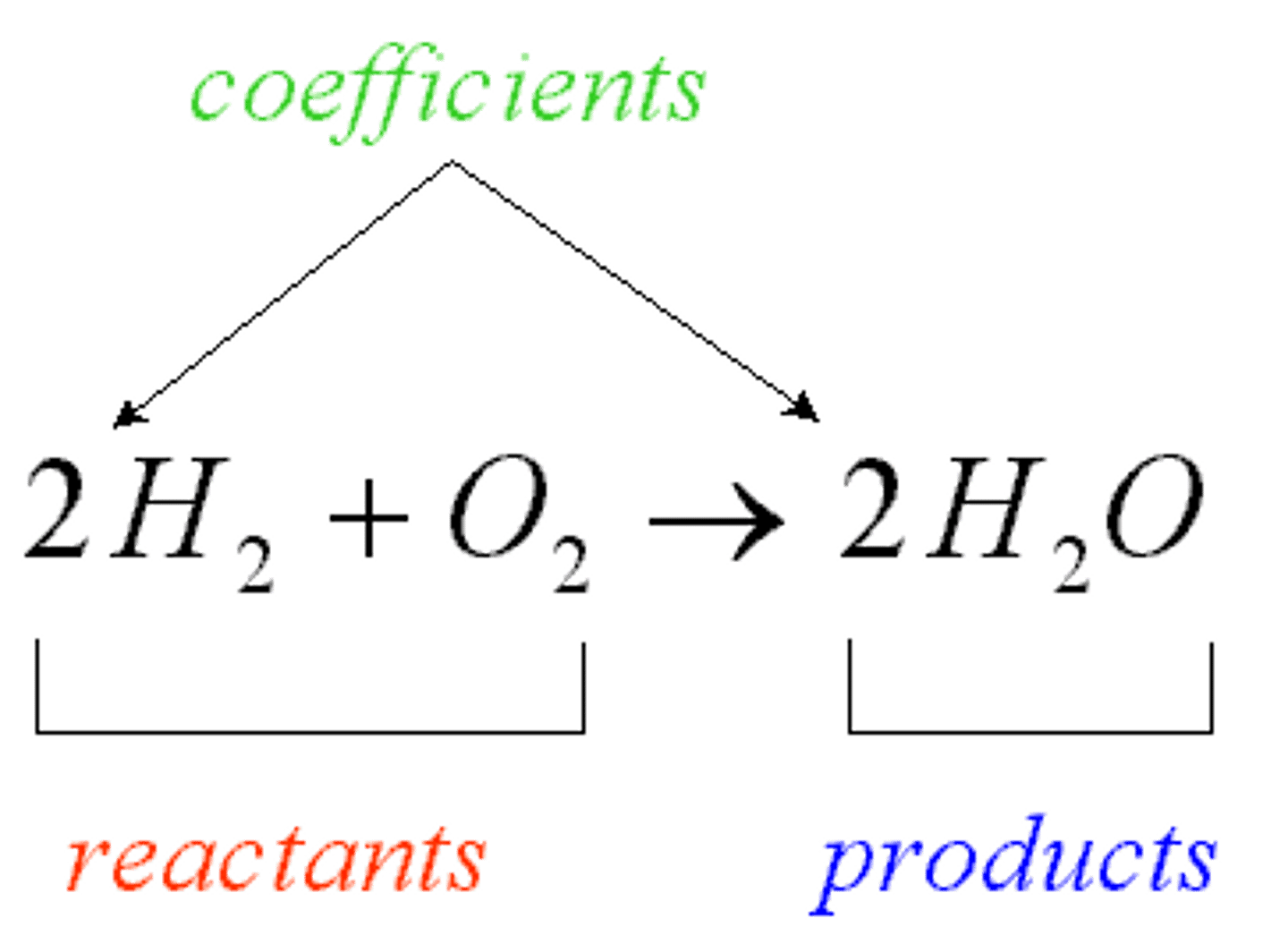
Chemical bond
The atoms of a compound are held together by chemical bonds, which may be ionic bonds, covalent bonds, or hydrogen bonds.

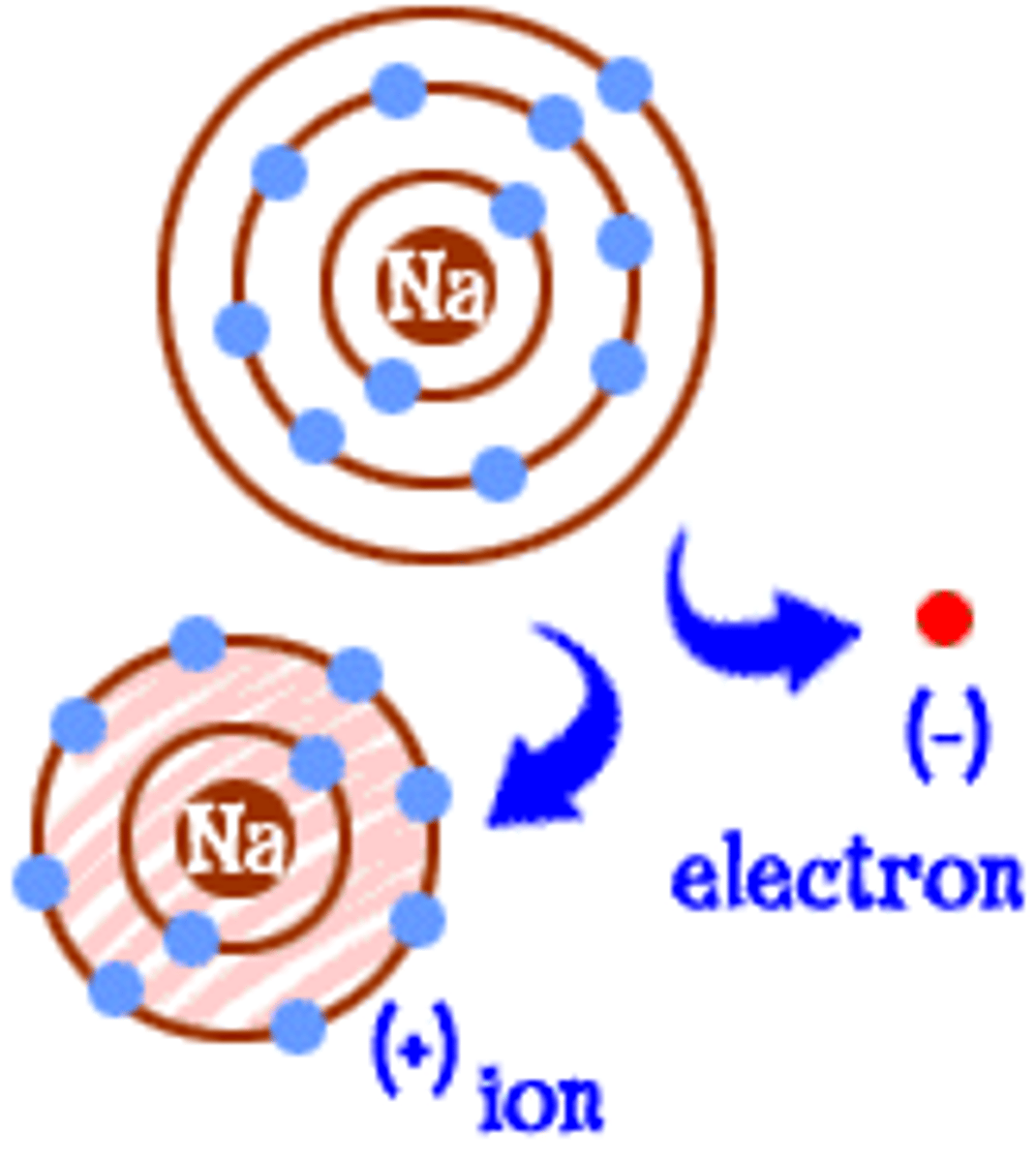
Ionic bond
An ionic bond is formed between two atoms when one or more electrons are transferred from one atom to the other. In this case, one atom becomes negatively charged and one atom becomes positively charged.
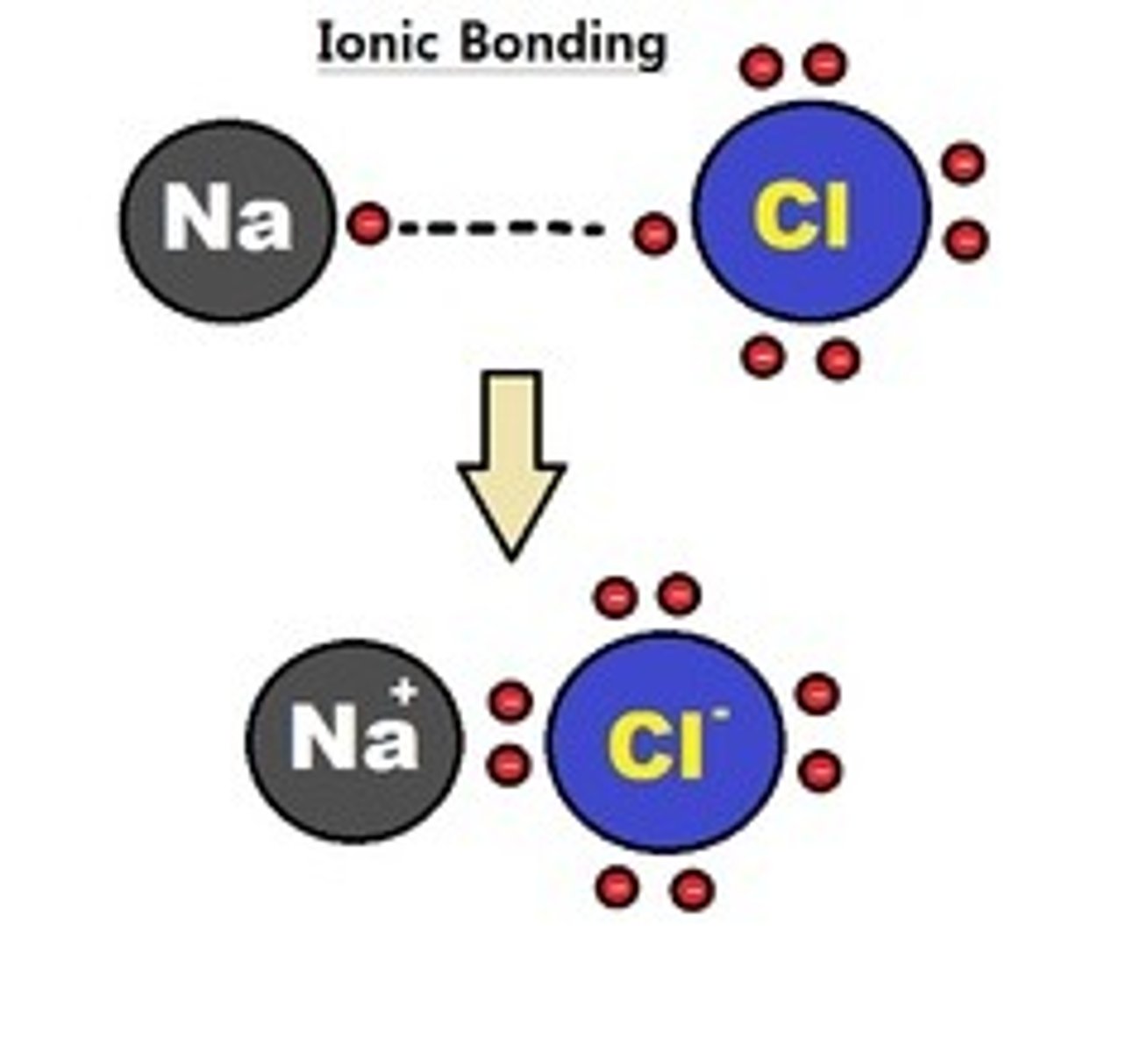
Ions
An ion is a charged form of an atom.
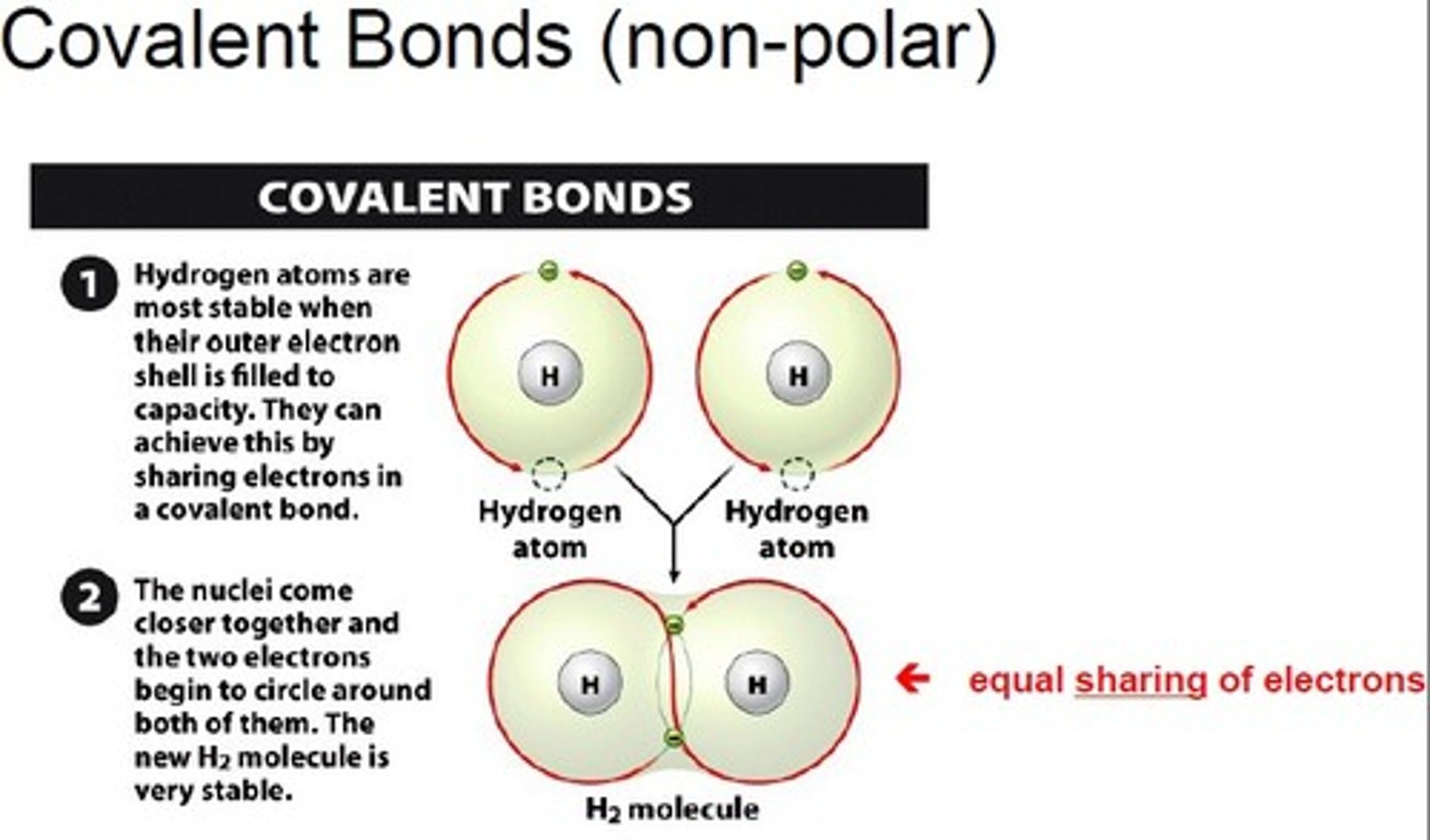
Covalent bond
A covalent bond is formed when electrons are shared between atoms and can be polar or non-polar.
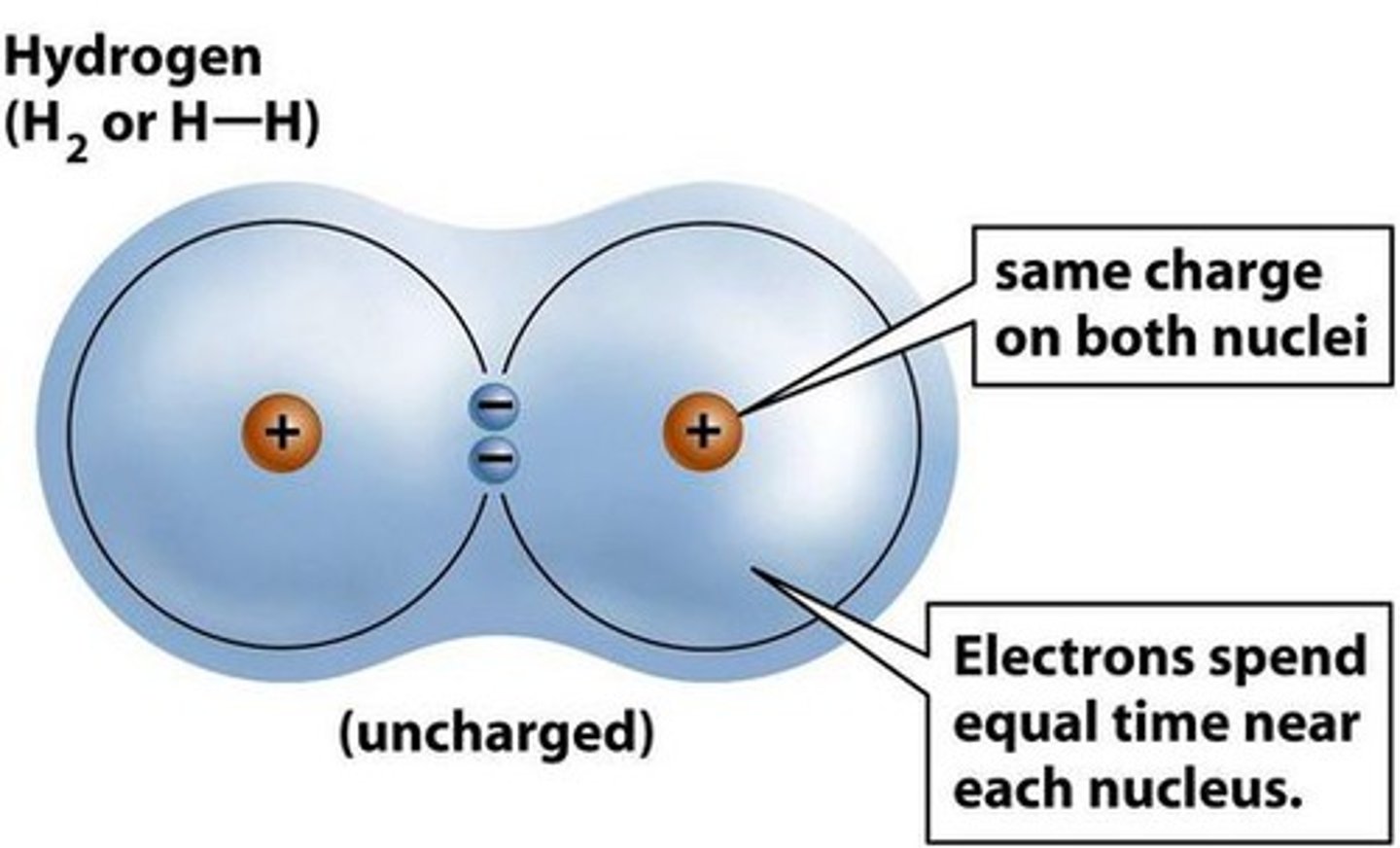
Non-polar covalent
A non-polar covalent bond is a covalent bond in which the electrons are shared equally between the atoms.
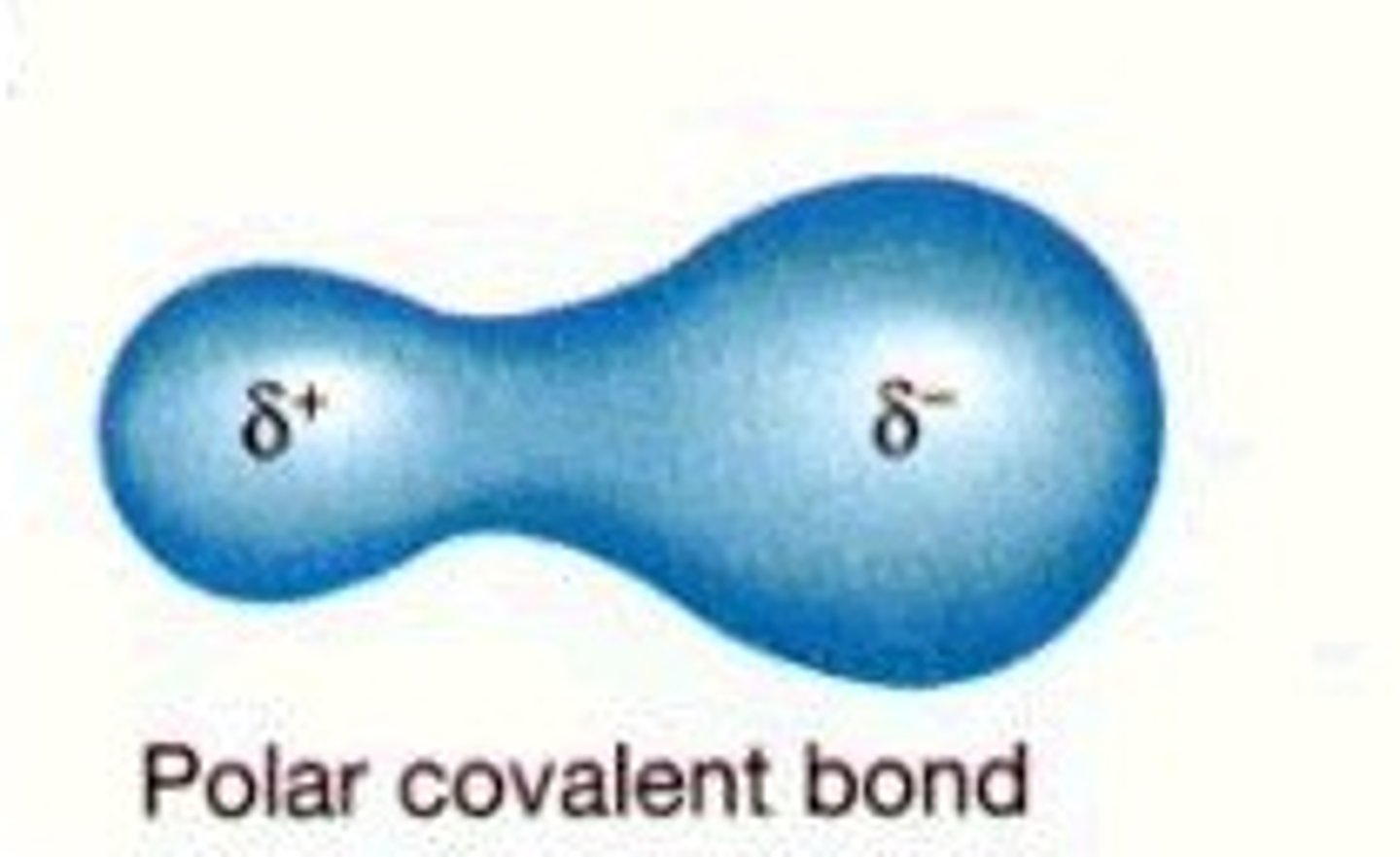
Polar covalent
A polar covalent bond is a covalent bond in which the electrons are shared unequally - one atom attracts electrons more than the other.
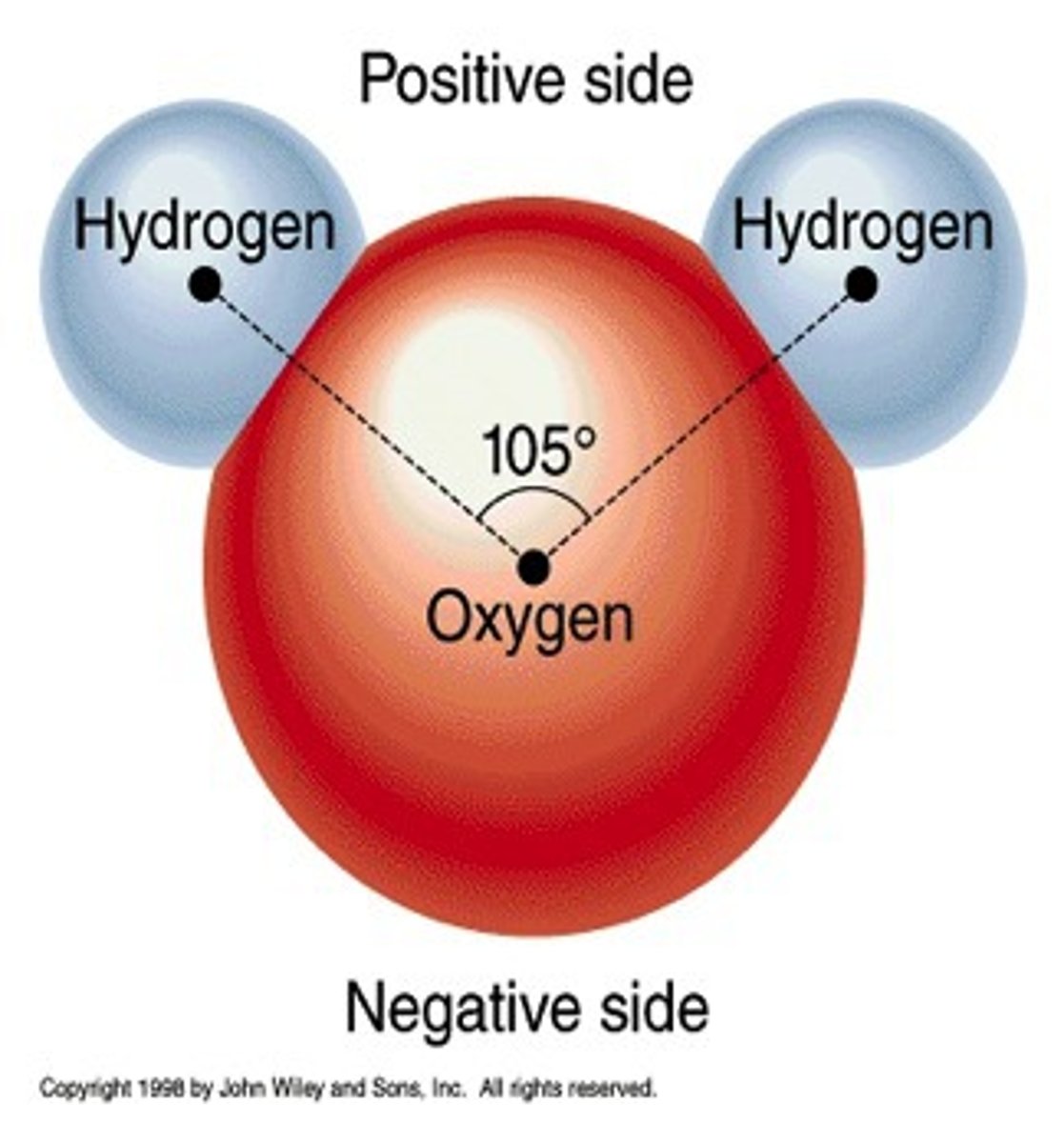
Polar
A molecule is polar if it has partially positive and partially negative charged ends, such as water.
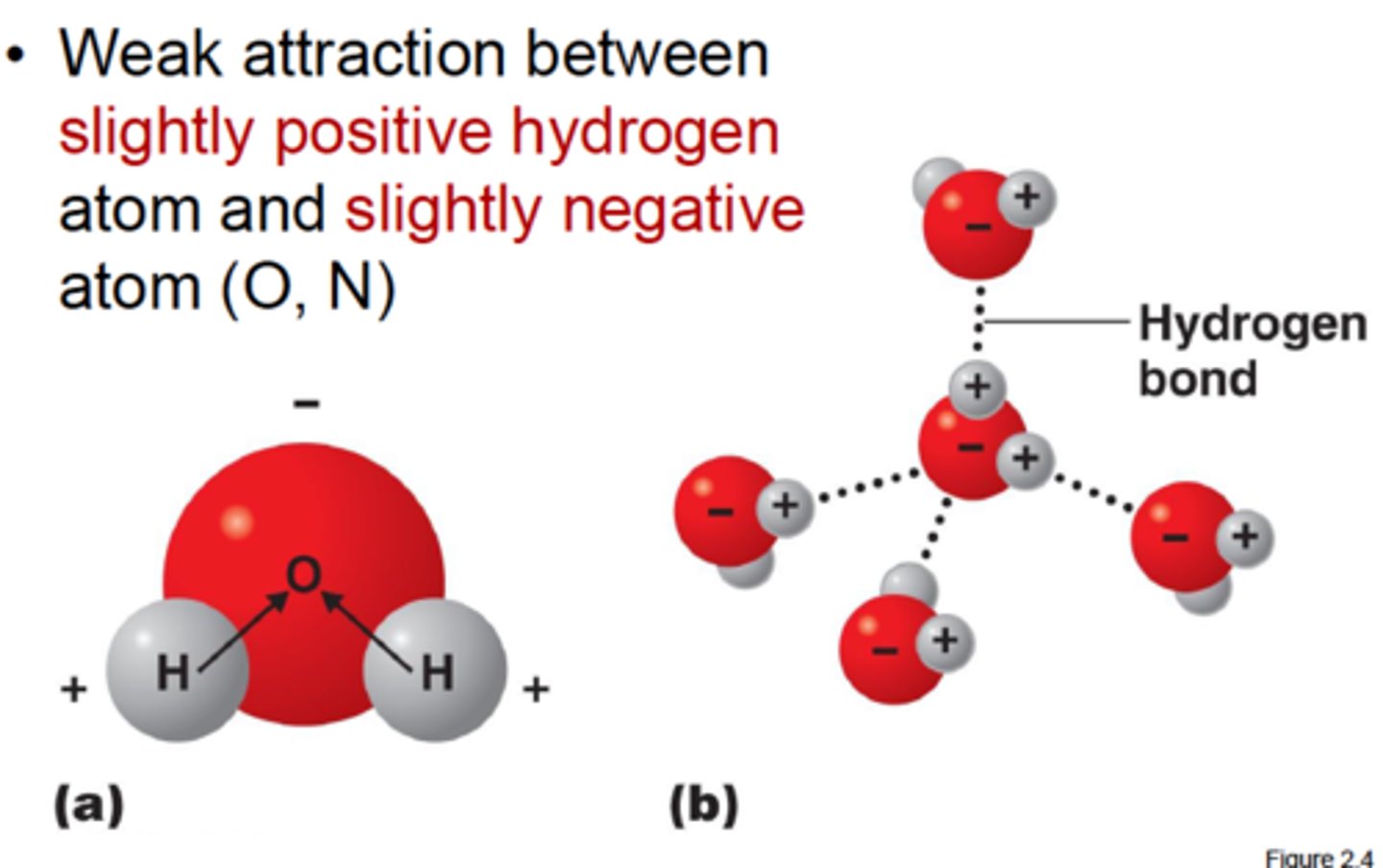
Hydrogen bonds
Hydrogen bonds are intermolecular attractions that form when a hydrogen bond that is covalently bonded to one electronegative atom that it also attracted to another electronegative atom. Hydrogen bonds are individually weak but are strong when present in large numbers.
Cohesion
Cohesion refers to the tendency of water molecules to stick together and allows water to have a high surface tension.
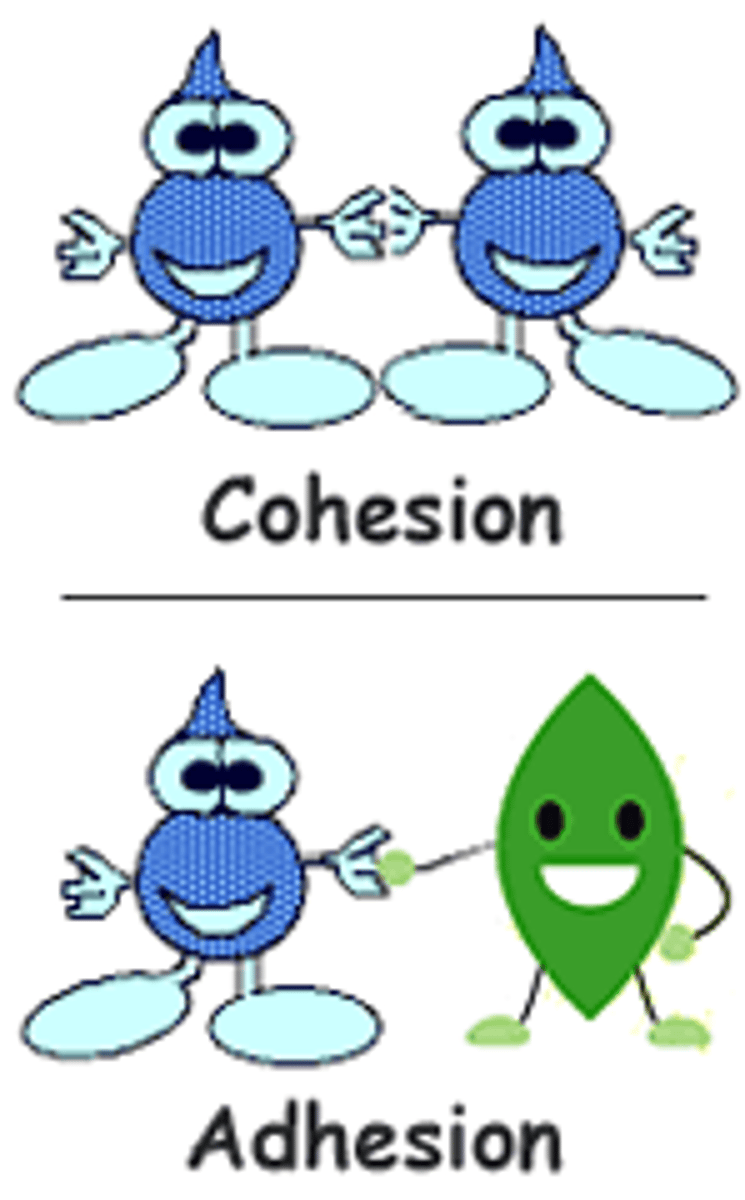
Adhesion
Adhesion refers to the tendency of water molecules to stick to other substances, which accounts for the phenomenon of capillary action.
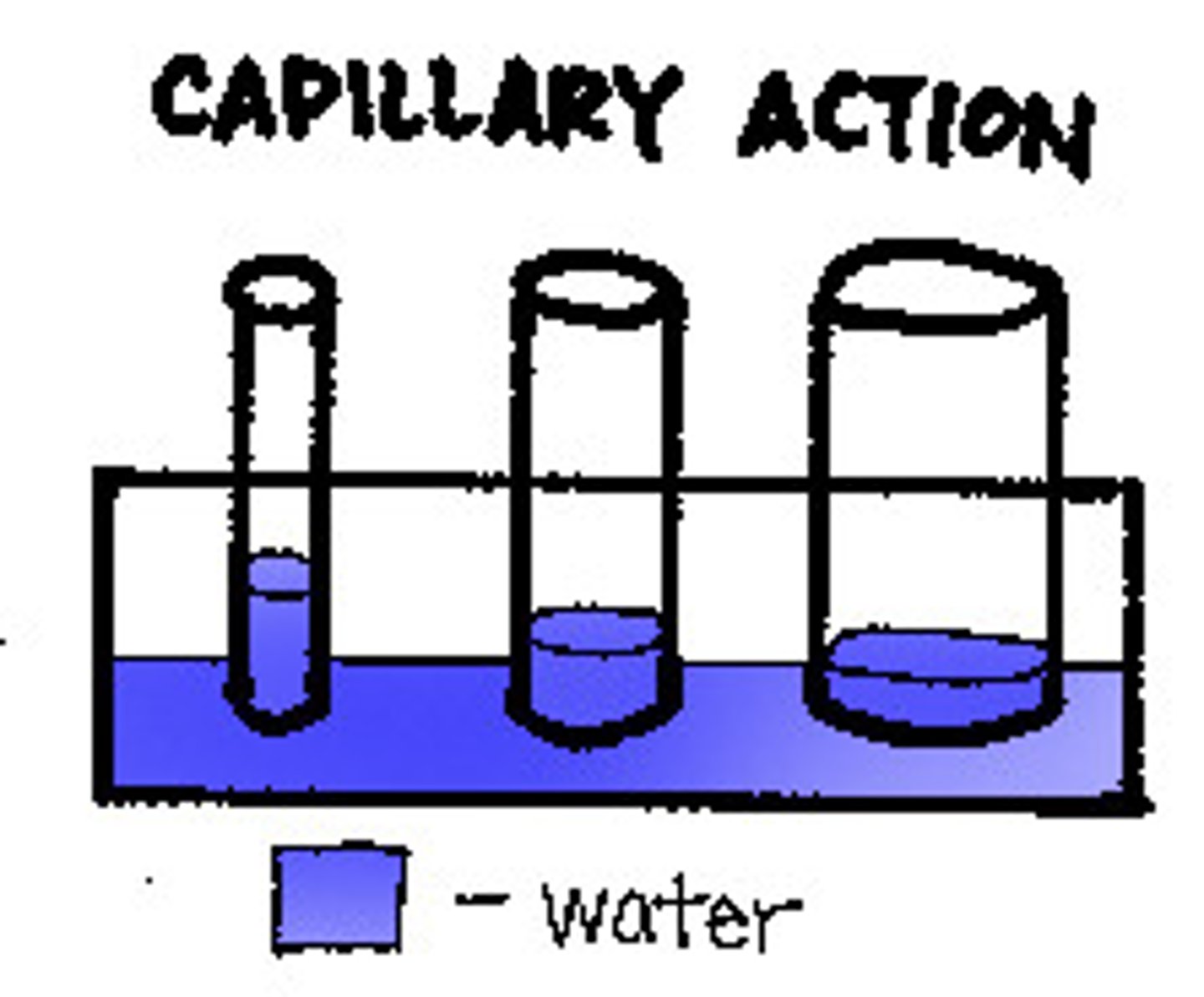
Capillary action
Capillary action is the ability of water, or other liquids, to travel against gravity in a thin vessel and accounts for the ability of water to rise up the roots, trunks, and branches of trees.
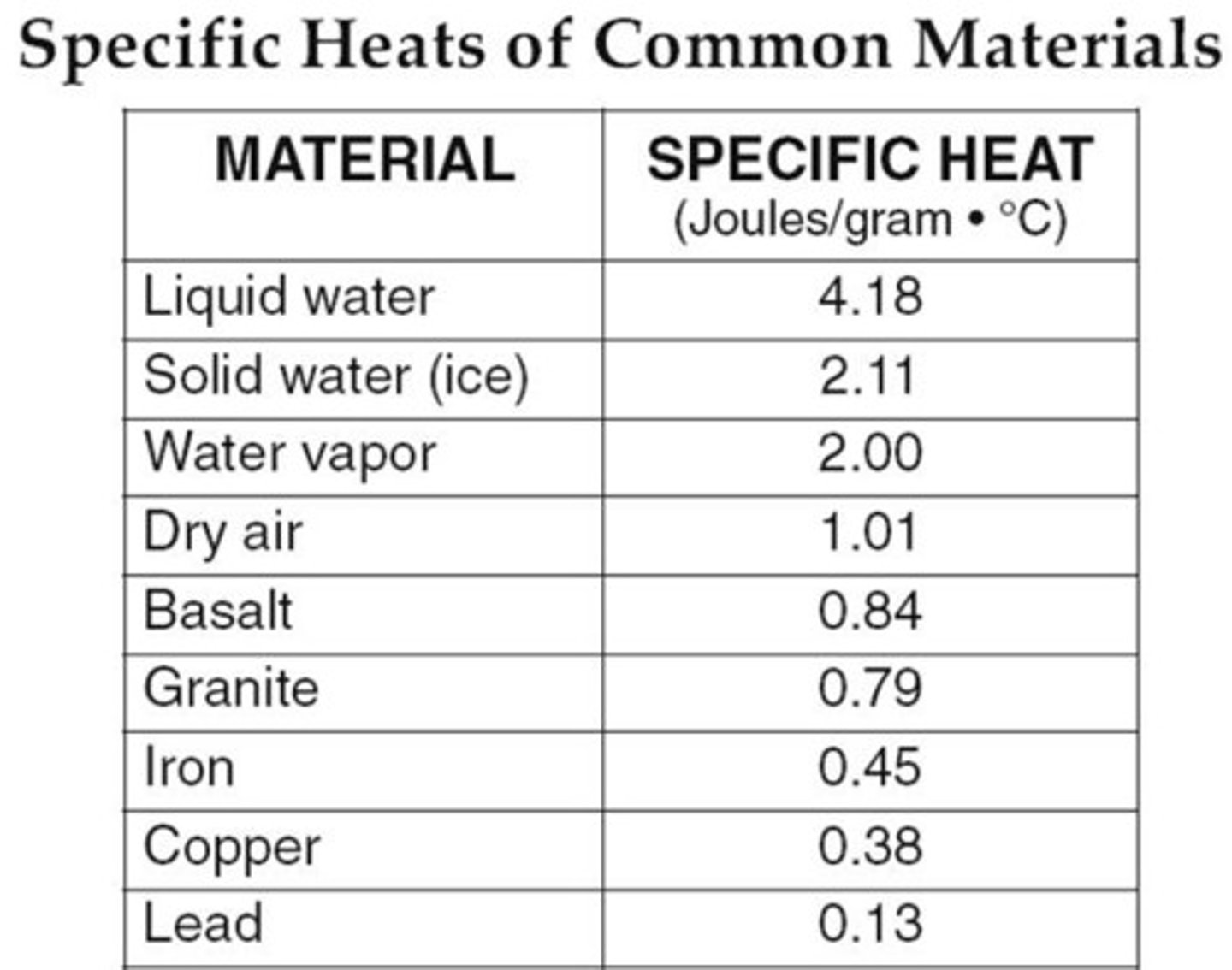
Heat capacity
Heat capacity refers to the ability of a substance to store heat and is the quantity of heat required to change the temperature of a substance by 1 degree. Water has a high heat capacity, allowing it to keep a fairly stable temperature in our bodies and in the environment.
Surface tension
Surface tension is a property of water, due to the cohesiveness of its molecules, that allows things (sometimes organisms) to float and stride on its surface without sinking.

Acidic
A solution is acidic if it contains excess hydrogen ions. It will have a pH less than 7.

Basic
A solution is basic if it releases hydroxide ions when added to water. These solutions are said to be alkaline and are usually slippery.
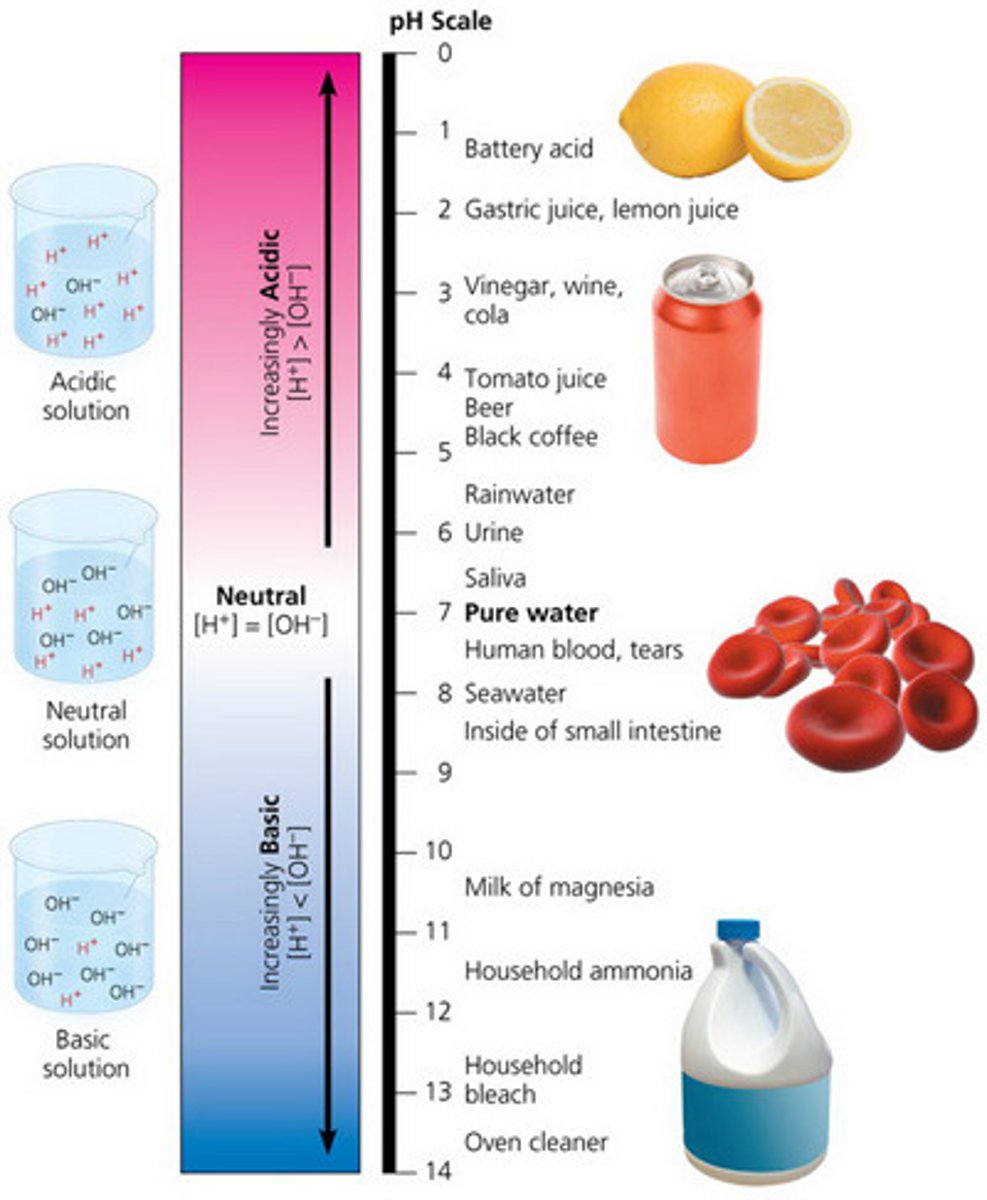
Neutral
A solution is neutral, neither basic nor acidic, if it has a pH of 7.
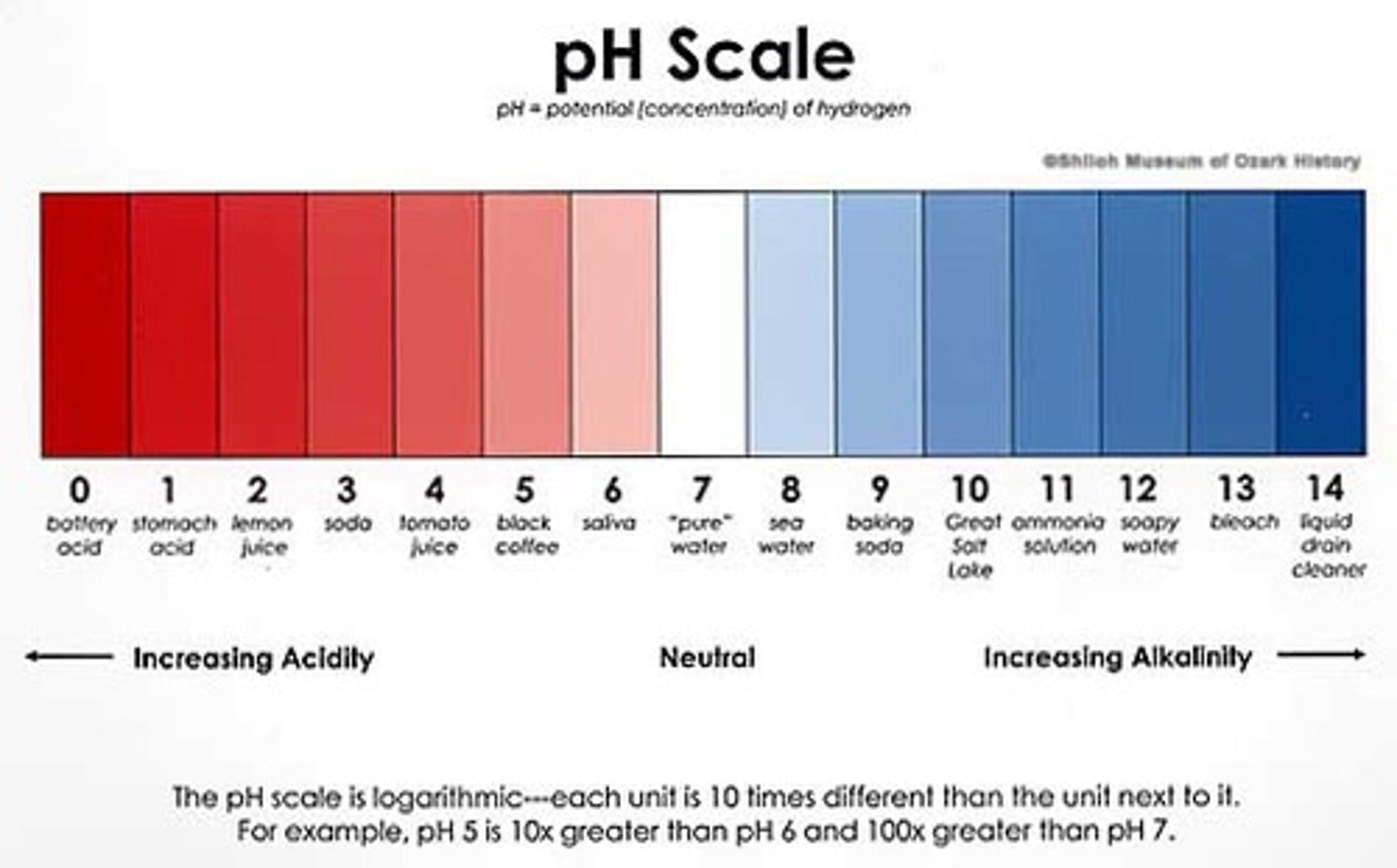
pH scale
The pH scale is a logarithmic scale to measure acidity, with 1 being most acidic, 14 being most basic, and 7 being neutral.

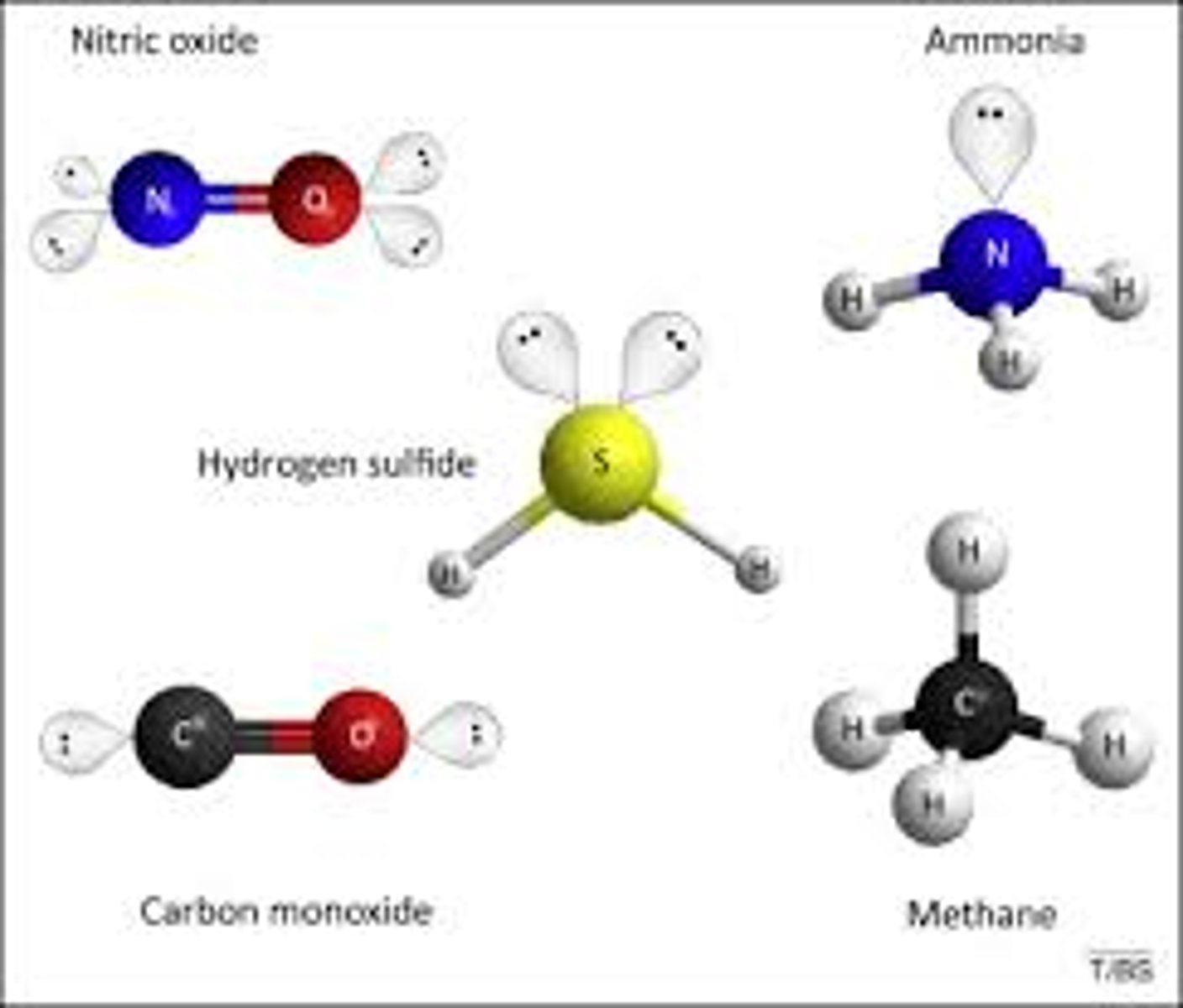
Organic compounds
Organic compounds are compounds that contain carbon and include carbohydrates, lipids, proteins, and nucleic acids.
Inorganic compounds
Inorganic compounds are compounds that do not contain carbon atoms (except for some simple carbon compounds such as carbon oxides)
Carbohydrates
Carbohydrates are organic compounds that contain carbon, hydrogen, and oxygen, usually in a ratio of 1:2:1.
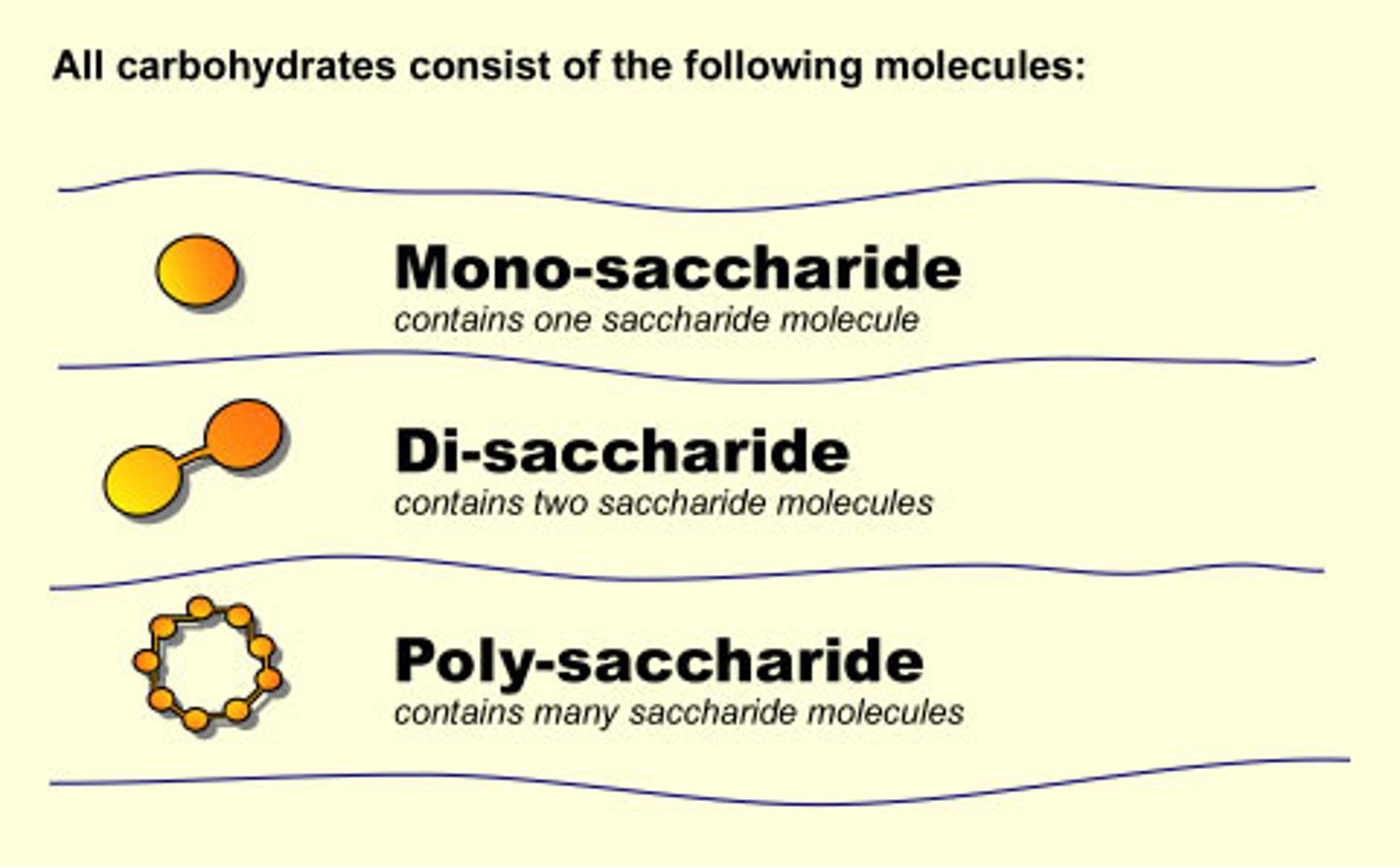
Monosaccharides
Monosaccharides are the simplest sugars which serve as an energy source for cells. The two most common monosaccharides are glucose and fructose.

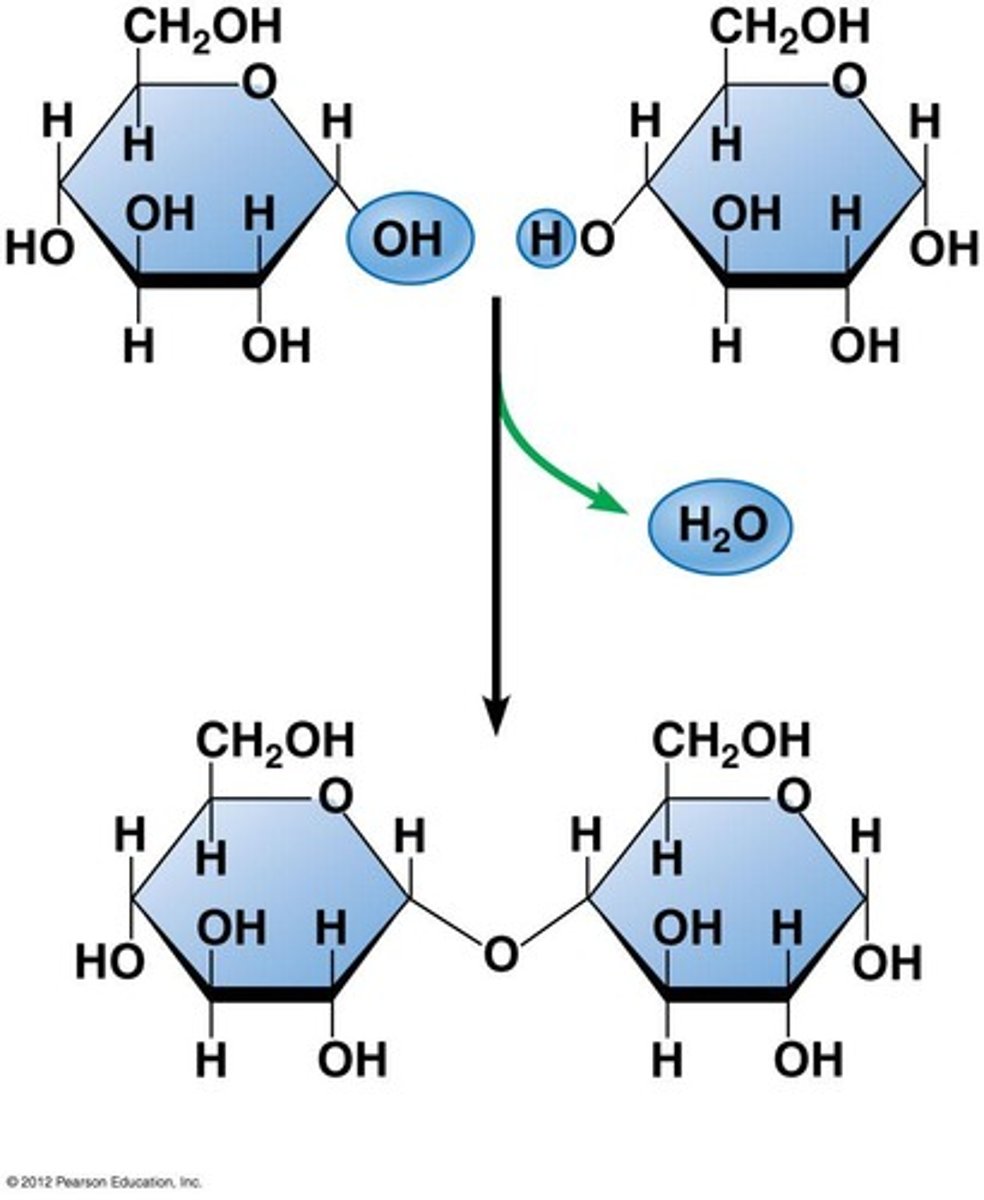
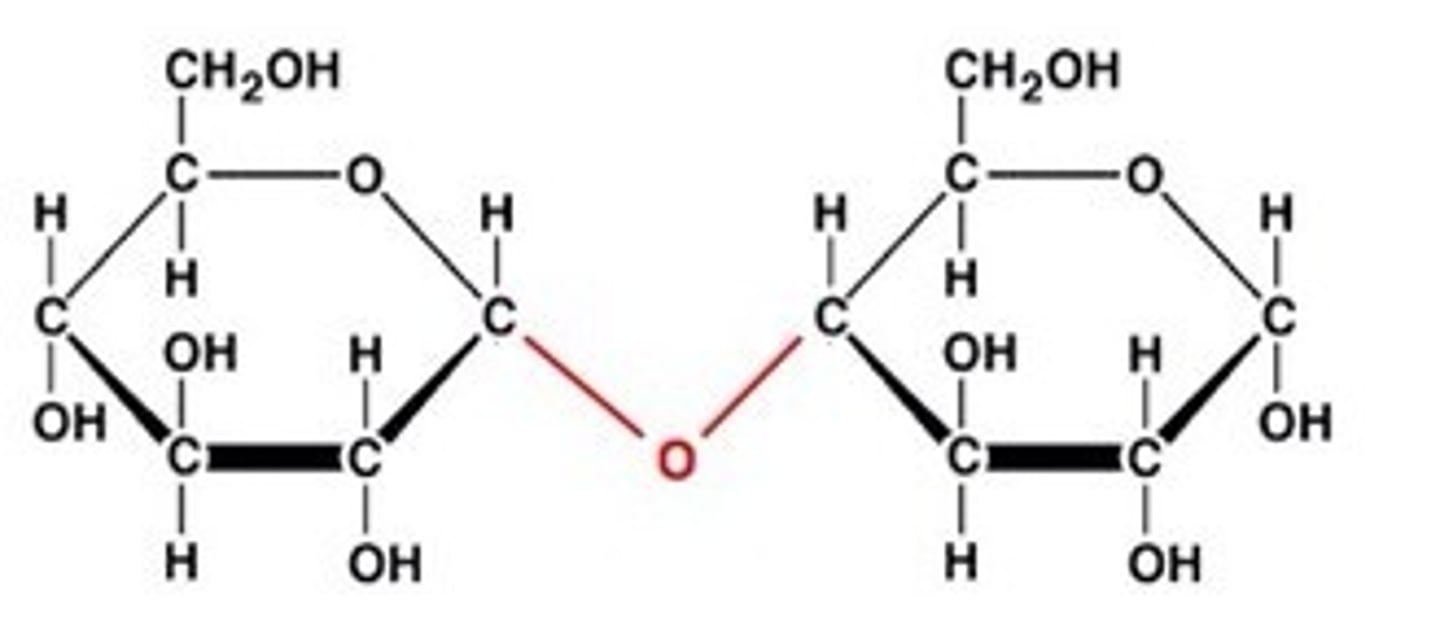
Disaccharides
Disaccharides are formed by two sugar molecules combining together through dehydration synthesis. An example of a disaccharide is maltose, make by linking two glucose molecules.
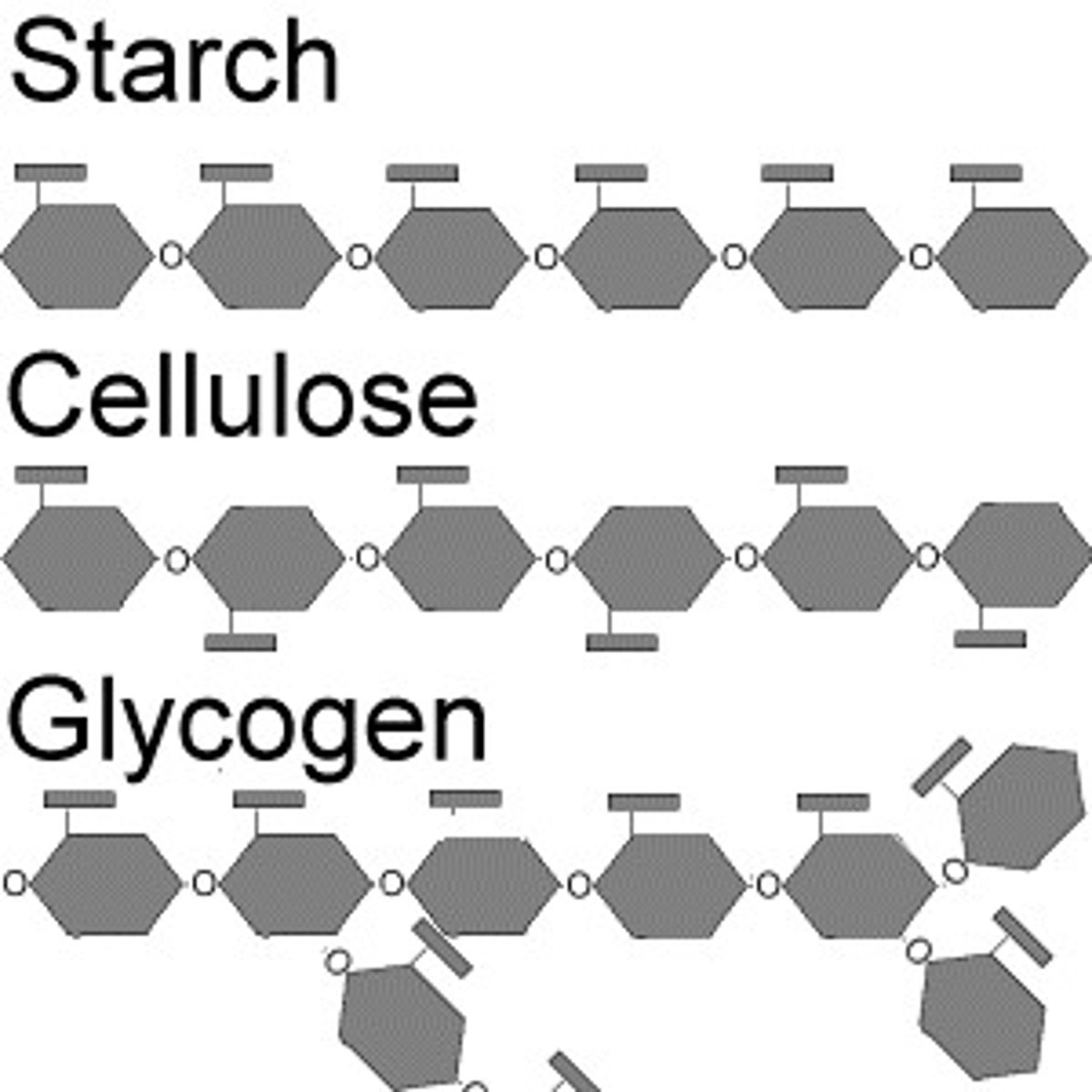
Polysaccharides
Polysaccharides are made up of many repeated unites of monosaccharides, and is therefore a type of polymer. The most common polysaccharides are starch, cellulose, and glycogen. Polysaccharides are often storage forms of sugar.
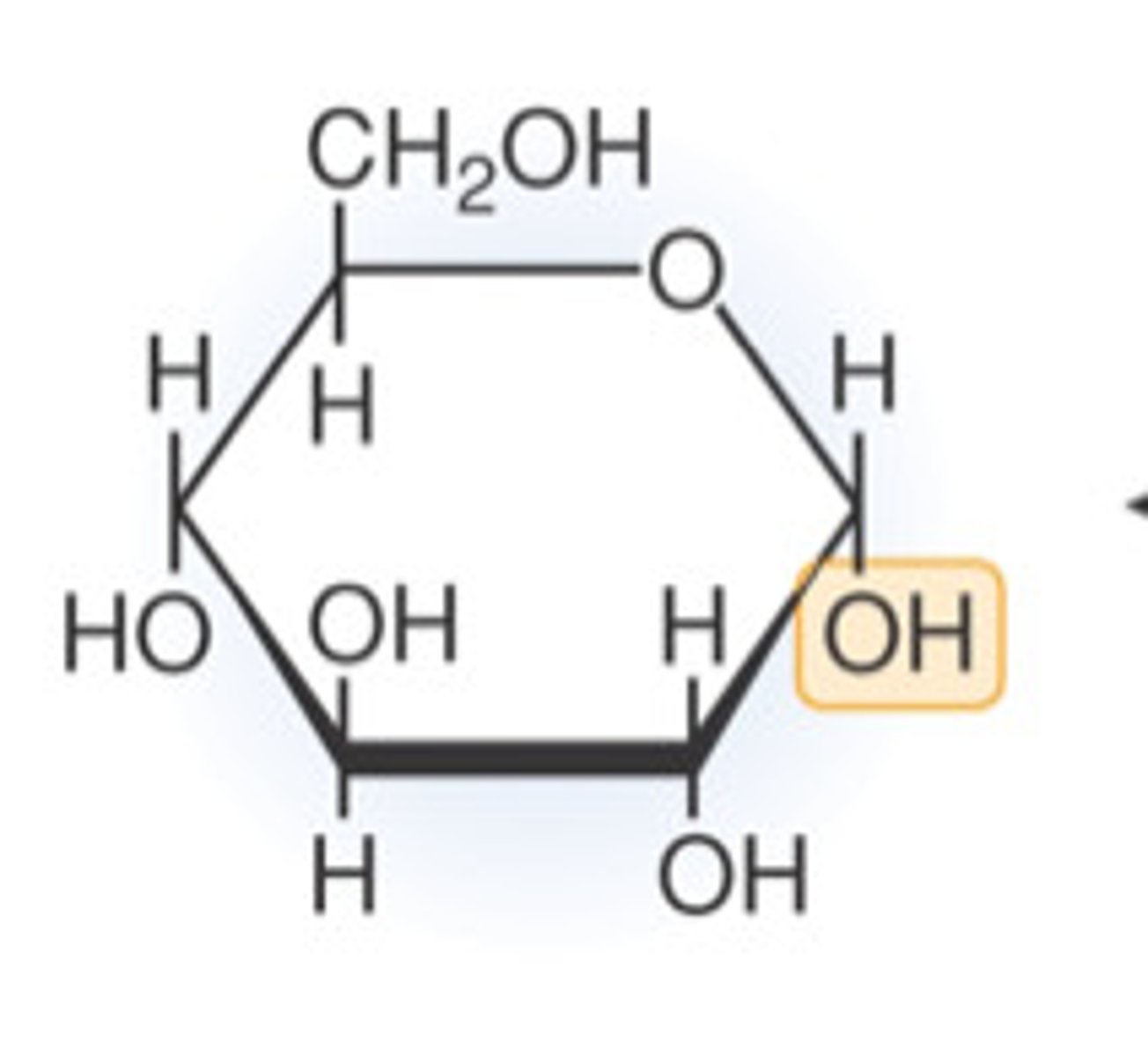
Glucose
Glucose is the most abundant monosaccharide and is a six-carbon sugar with the chemical formula C-6, H-12, O-6. Glucose comes in two forms: alpha glucose and beta glucose, which differ simply by a reversal of the H and OH of the first carbon.
Glycosidic bond
A glycosidic bond is a covalent bond in which a carbohydrate binds to another group, which could also be a carbohydrate. A glycosidic bond is found between the two glucose molecules in maltose.
Dehydration synthesis
Dehydration synthesis, or condensation, is the process by which two molecules come together by the loss of a water molecule, such as the forming of maltose from two glucose molecules.
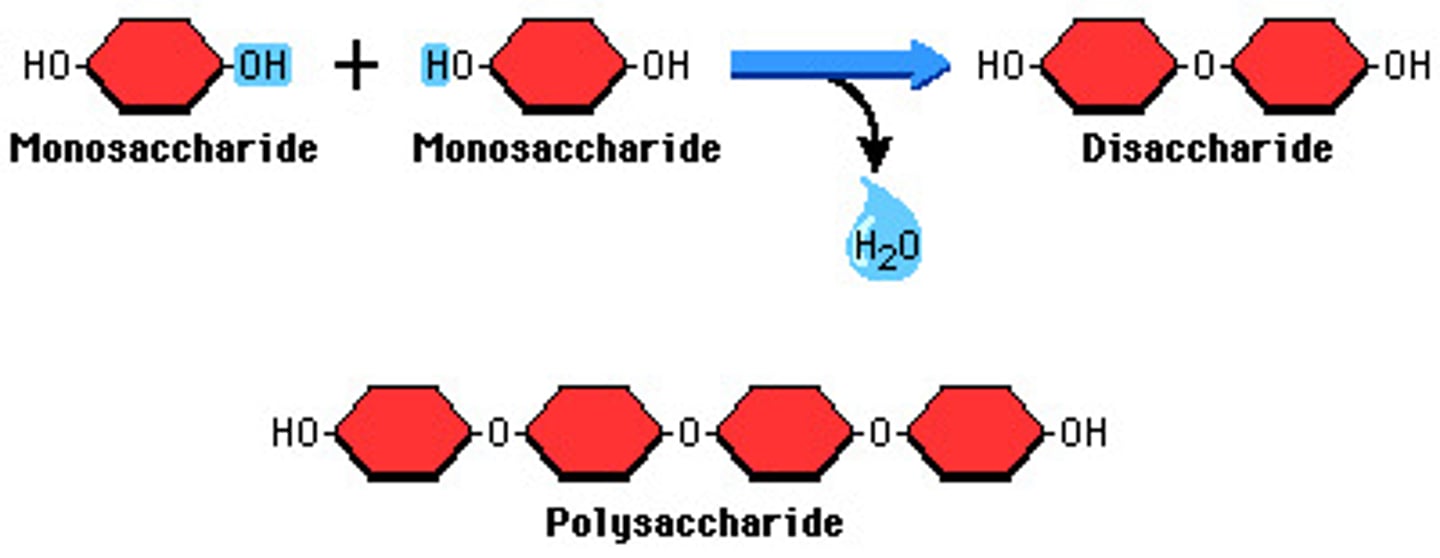
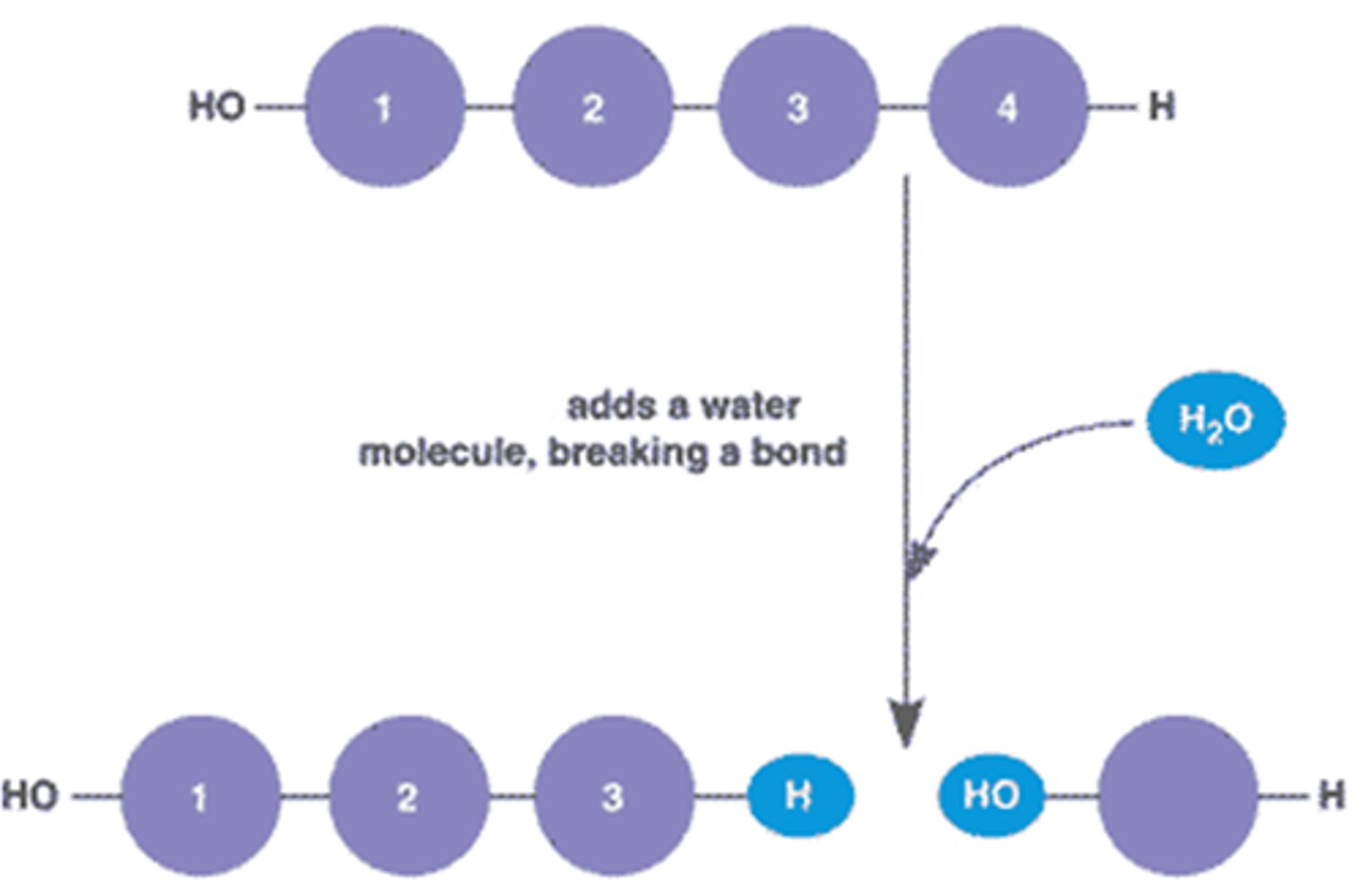
Hydrolysis
Hydrolysis is the opposite process of dehydration synthesis by which molecules are broken up by the addition of a water molecule, such as the formation of two glucose molecules from a maltose.
Polymer
A polymer is a molecule with repeating subunits of the same general type, such as polysaccharides.
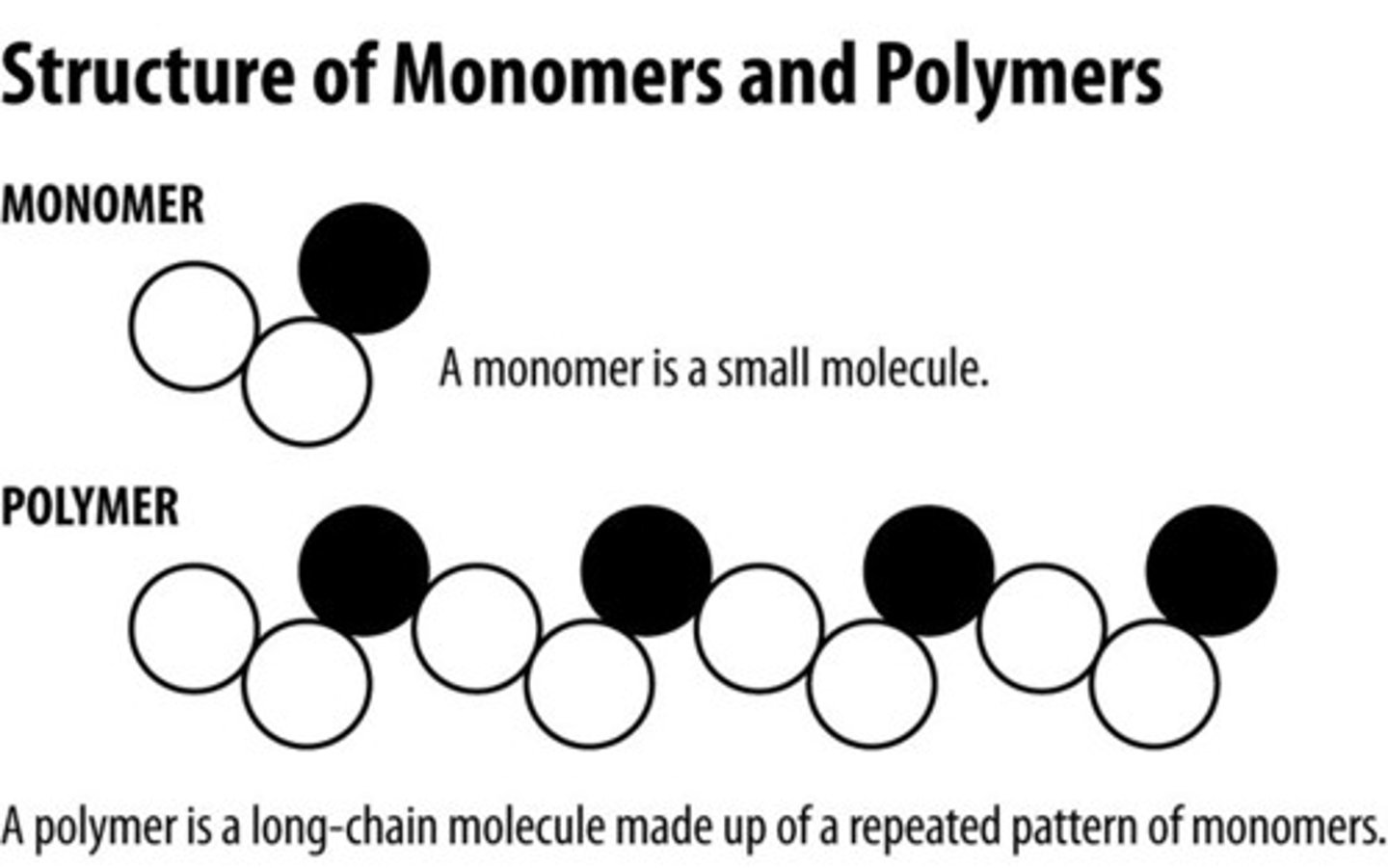
Starch
Starch is a polysaccharide of alpha glucose molecules bound together and is produced by most green plants as an energy store.
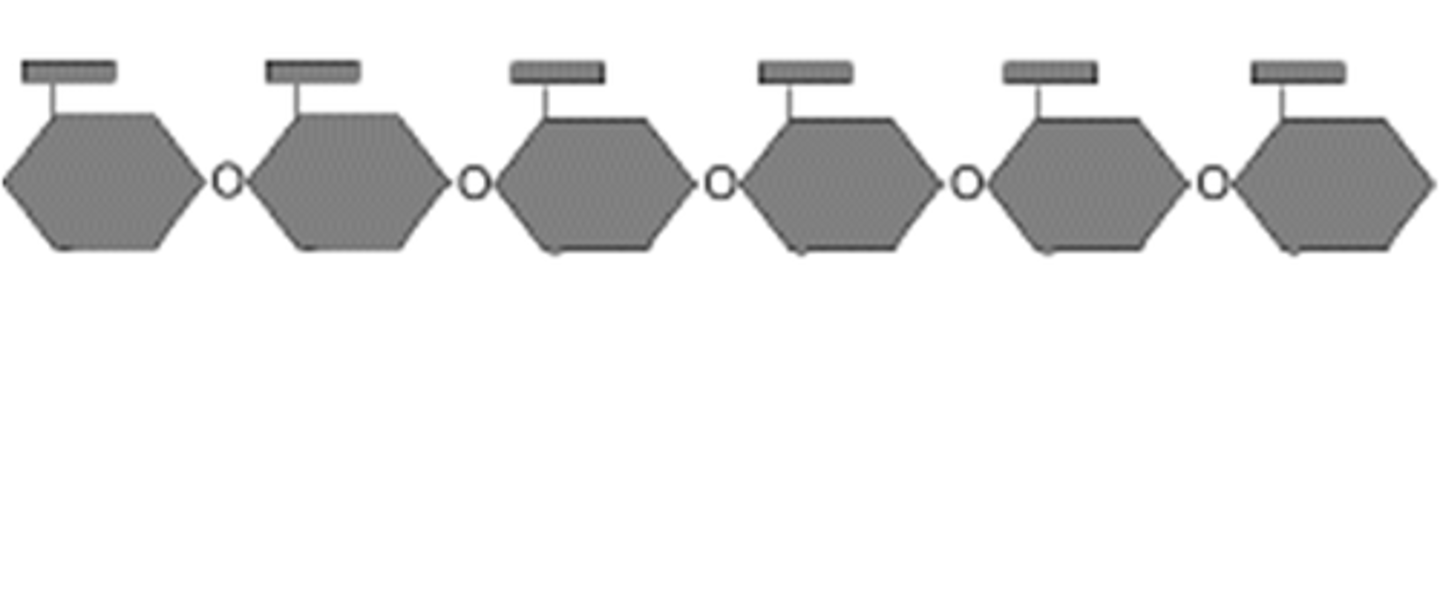
Cellulose
Cellulose is a polysaccharide of beta glucose molecules that is a major part of the cell wall in plants and is used to lend structural support.
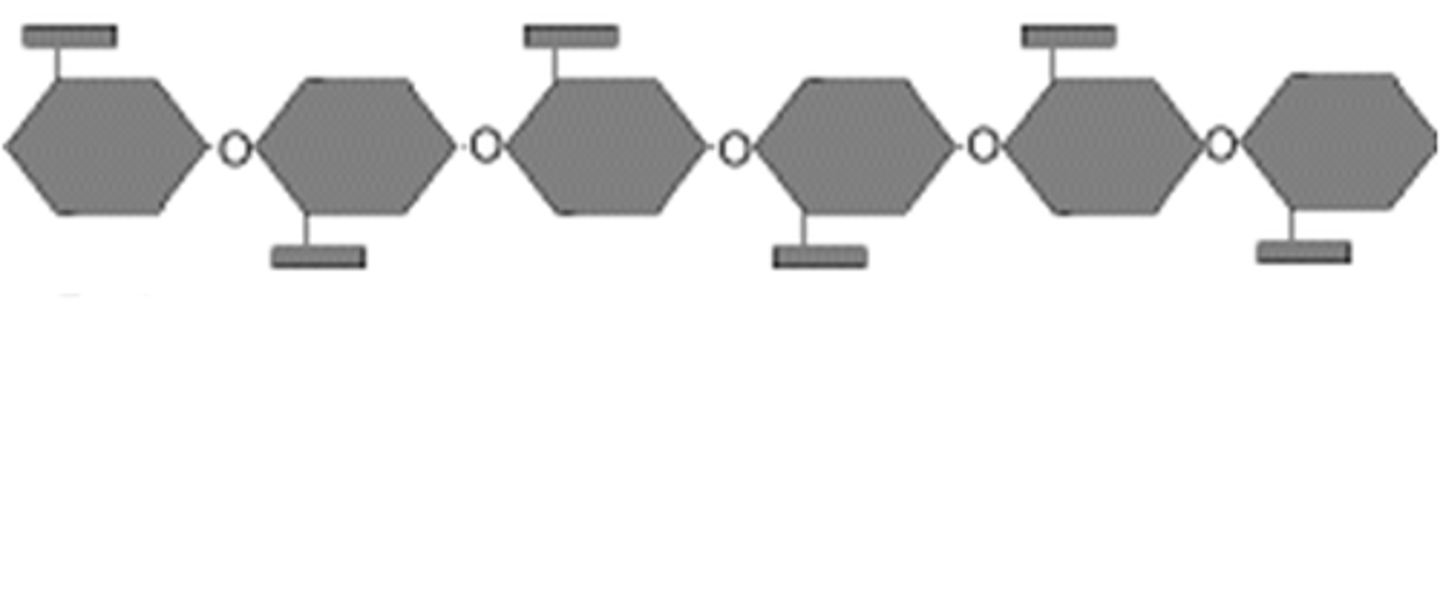
Glycogen
Glycogen is a multi-branch polysaccharide of glucose that is the main storage of glucose in the body.
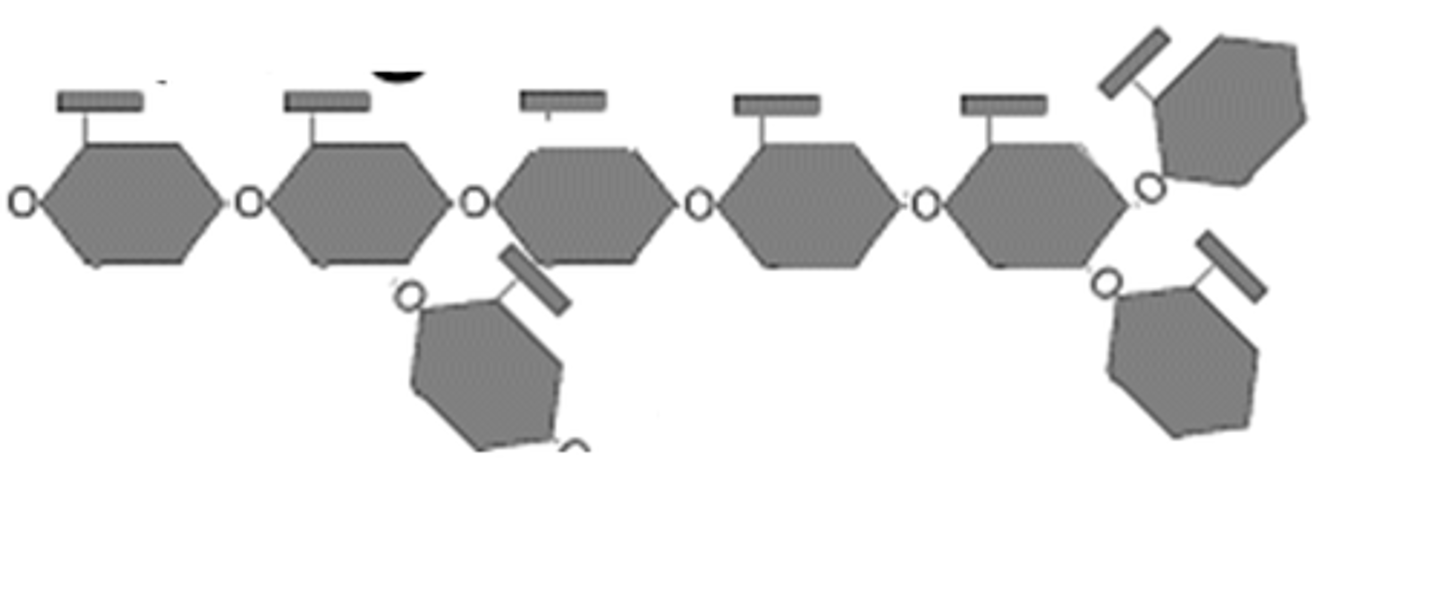
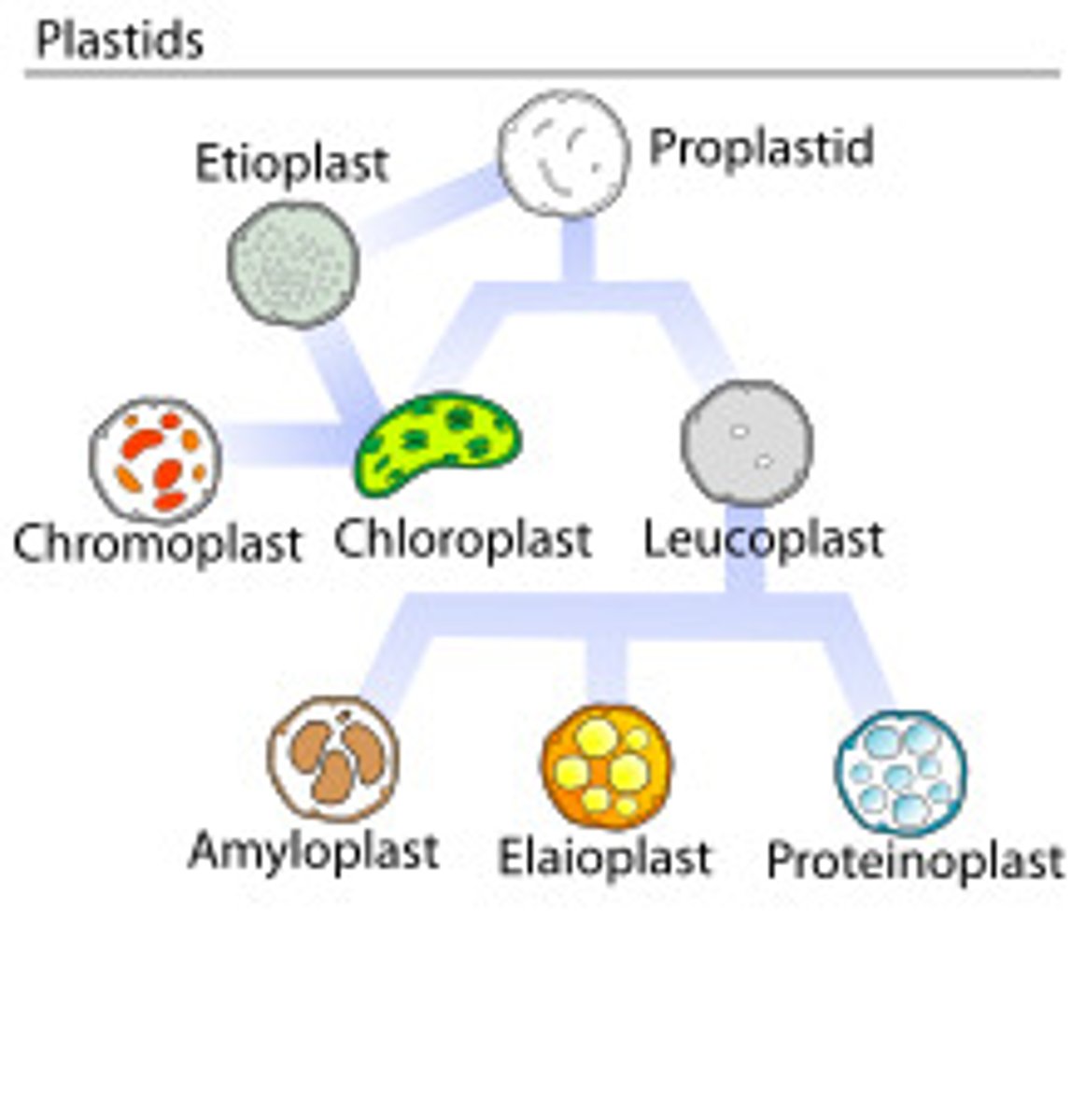
Plastids
Plastids are double membrane bound organelles that temporarily store starch in plants. Plastids include chloroplasts, chromoplasts, and leucoplasts.
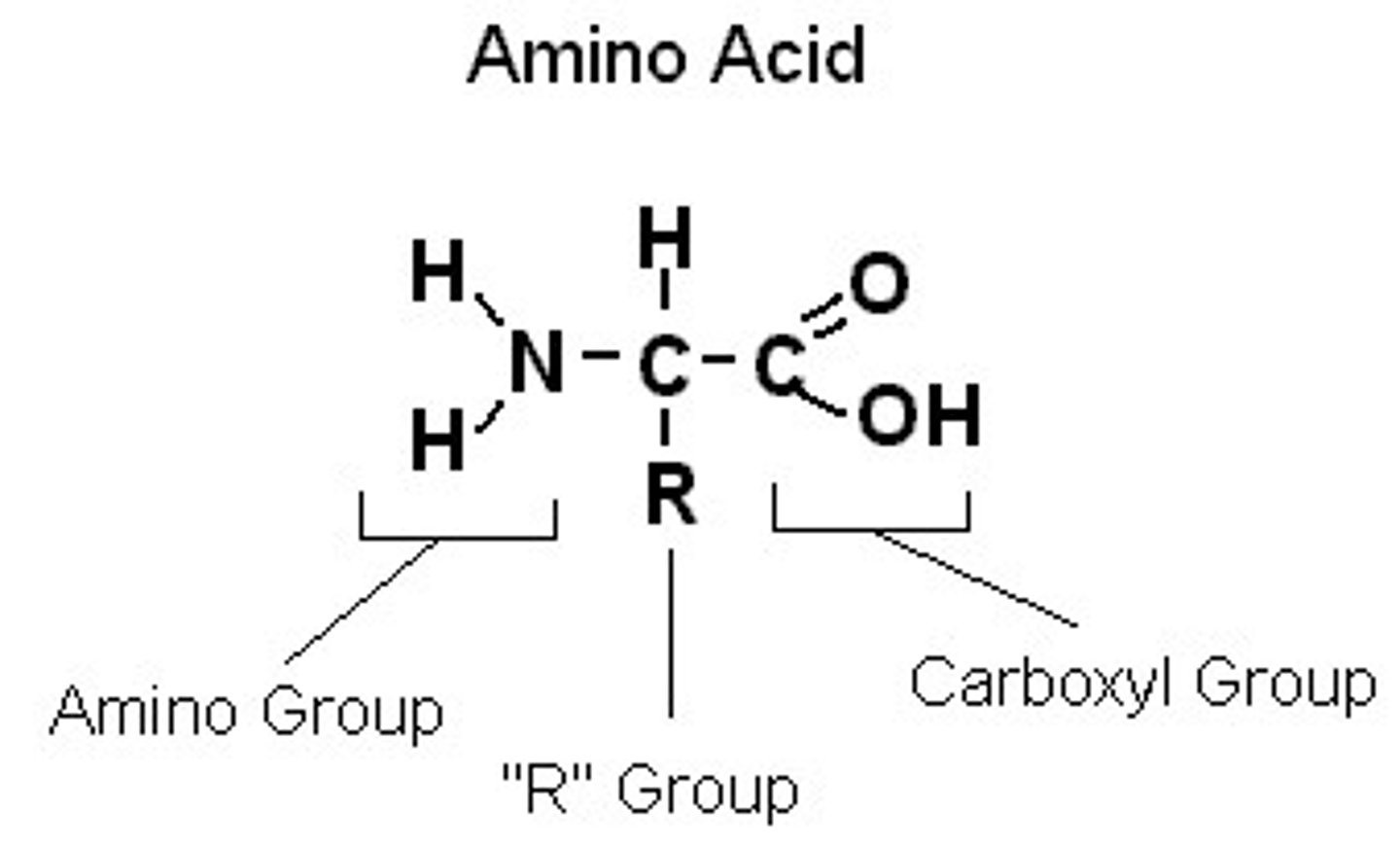
Amino acids
Amino acids are organic molecules that serve as the building blocks of proteins. They contain carbon, hydrogen, oxygen, and nitrogen atoms. Every amino acid has four parts: an amino group, a carboxyl group, a hydrogen, and an R group.
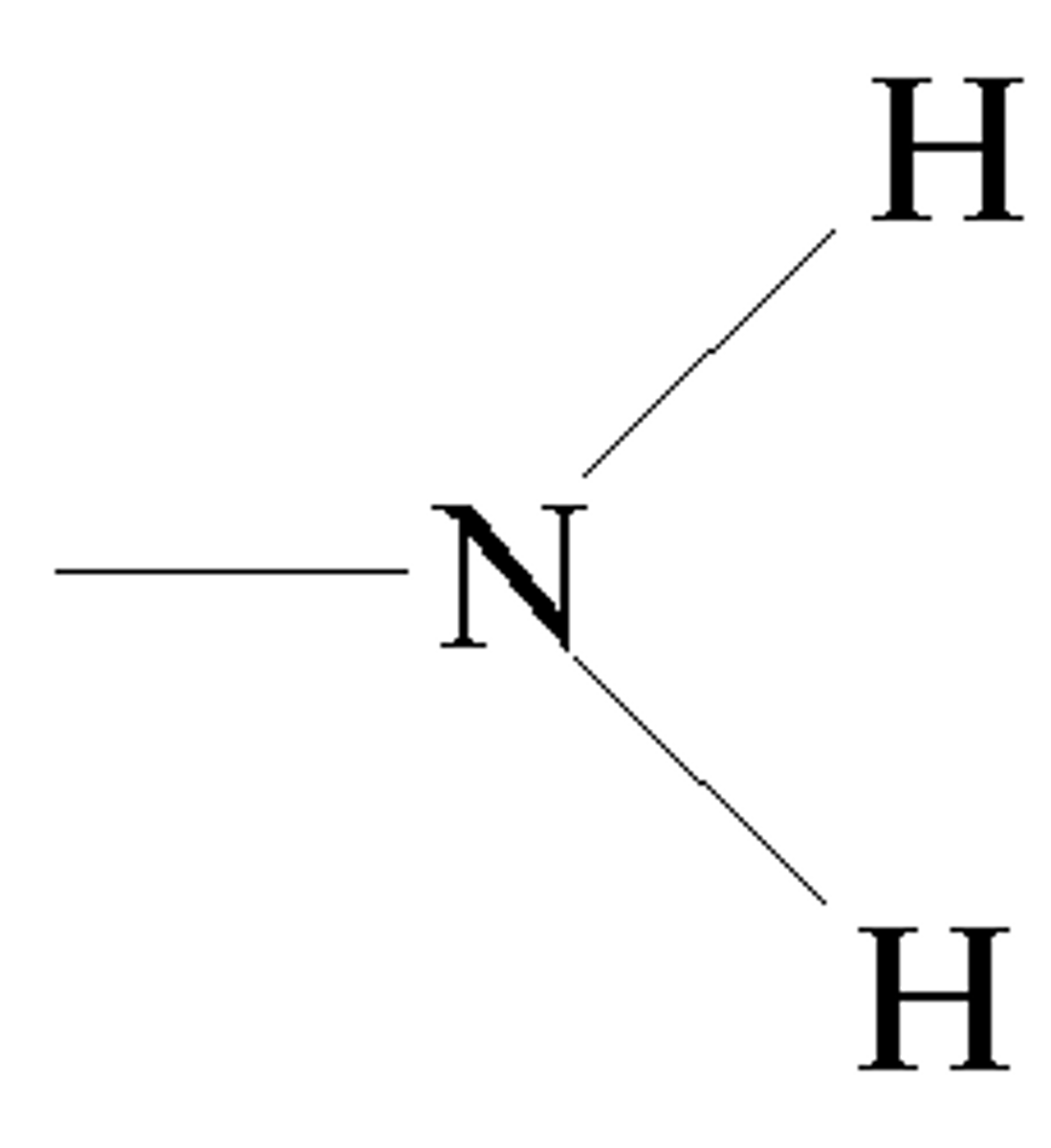
Amino group
An amino group is a functional group and is found in organic compounds known as amines.
Carboxyl group
Carboxyl groups are weak acids that are common in many organic molecules including amino acids and fatty acids.
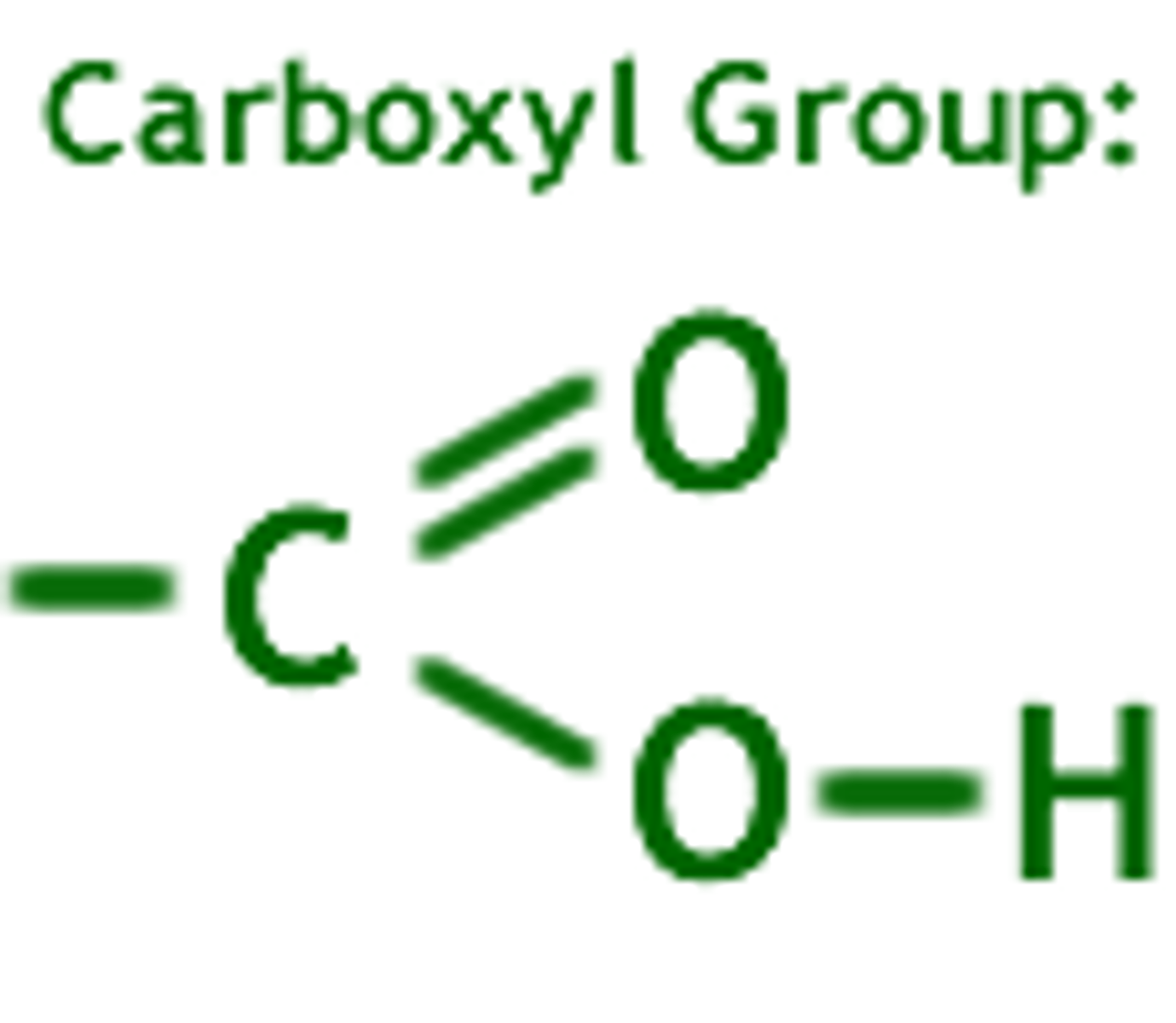
R group
An R-group is any group in which the carbon or hydrogen is attached to the rest of the molecule.
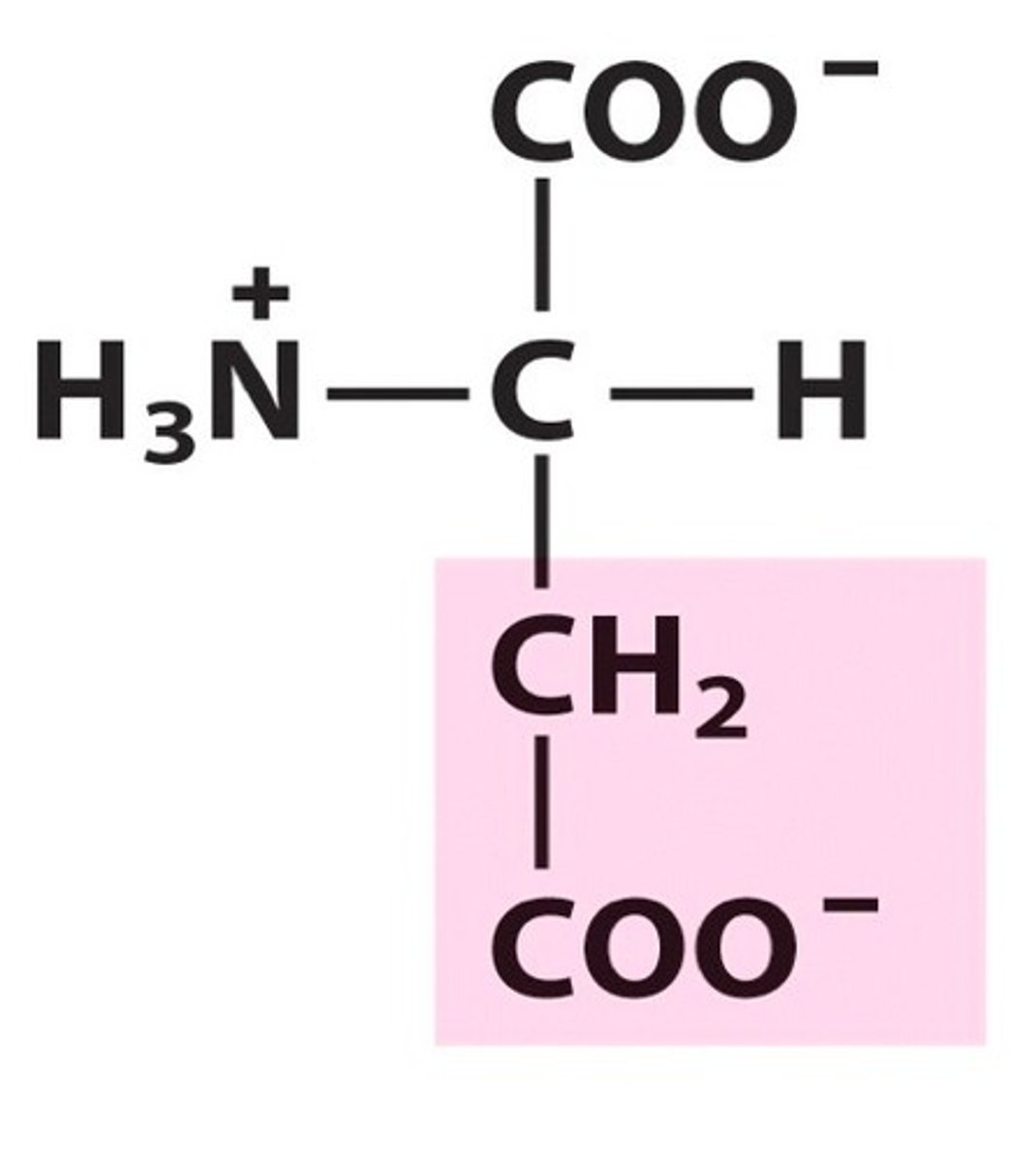
Side chain
Side chain is another name for an R group, and is a group of atoms attached to the main part of a molecule and having a ring or chain structure.
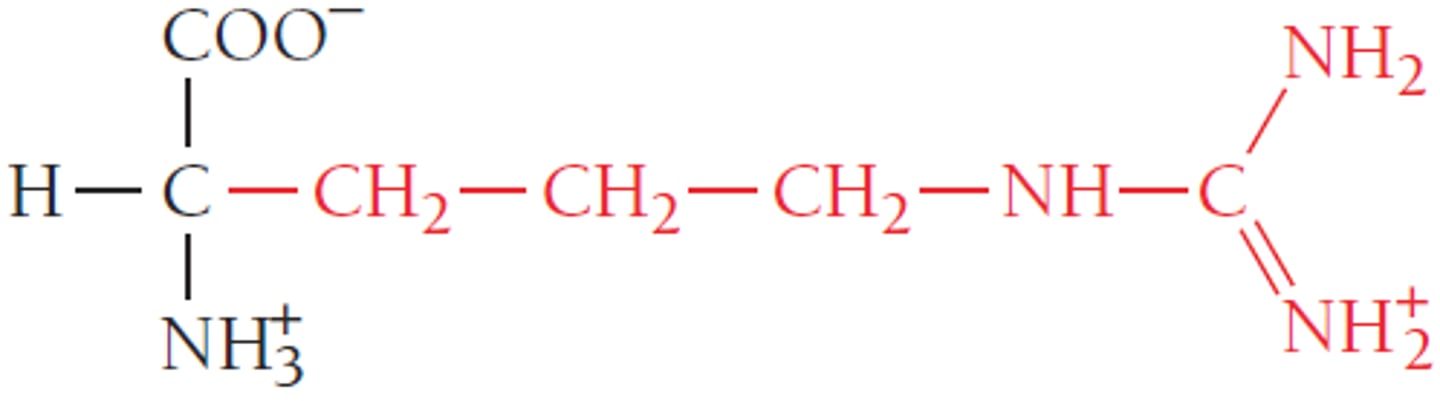
Functional group
A functional group is a distinctive group of atoms that play a large role in determining the chemical behavior of the compound they are a part of. In amino acids, functional groups include the carboxyl group and the amino group.
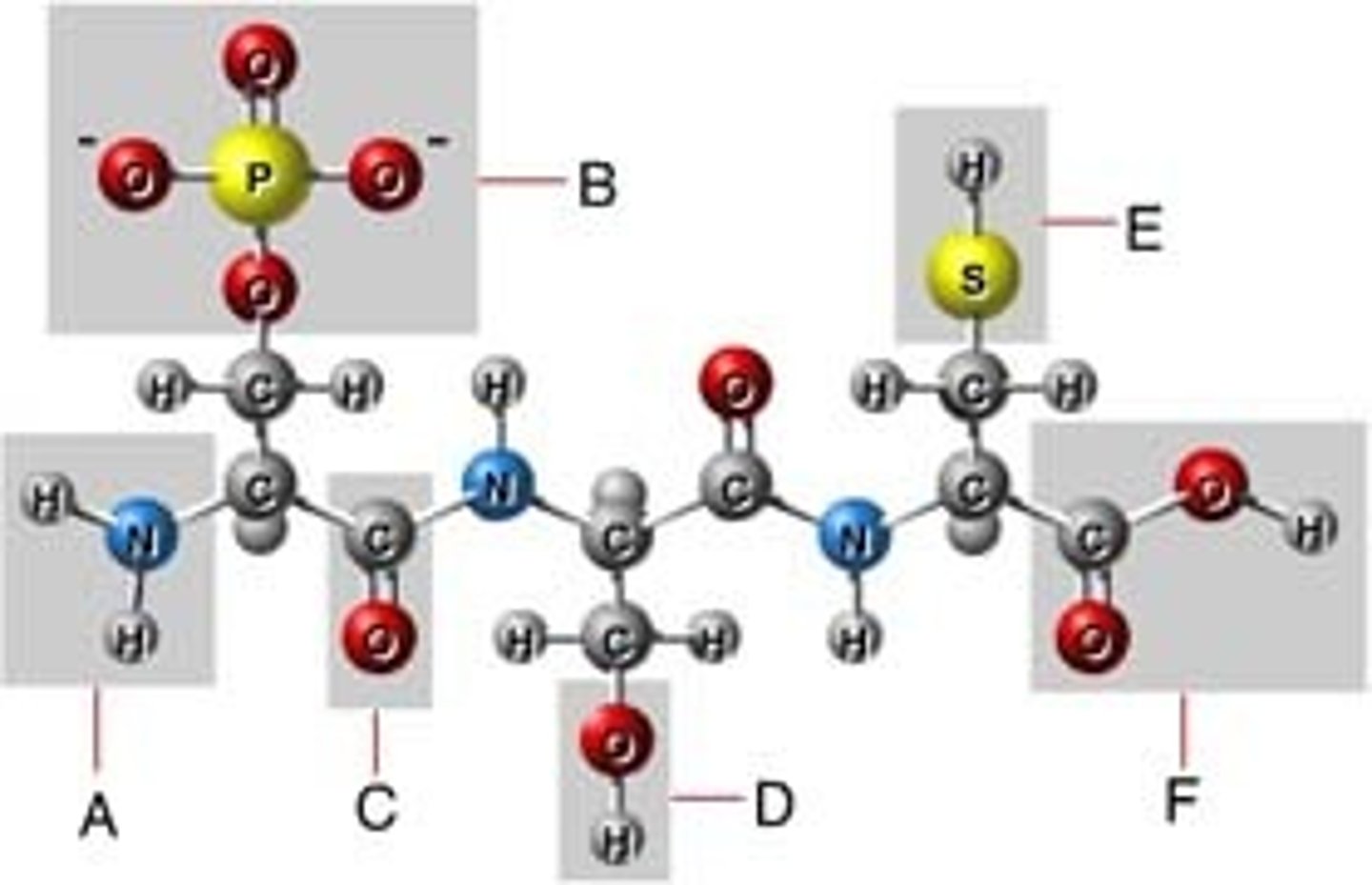
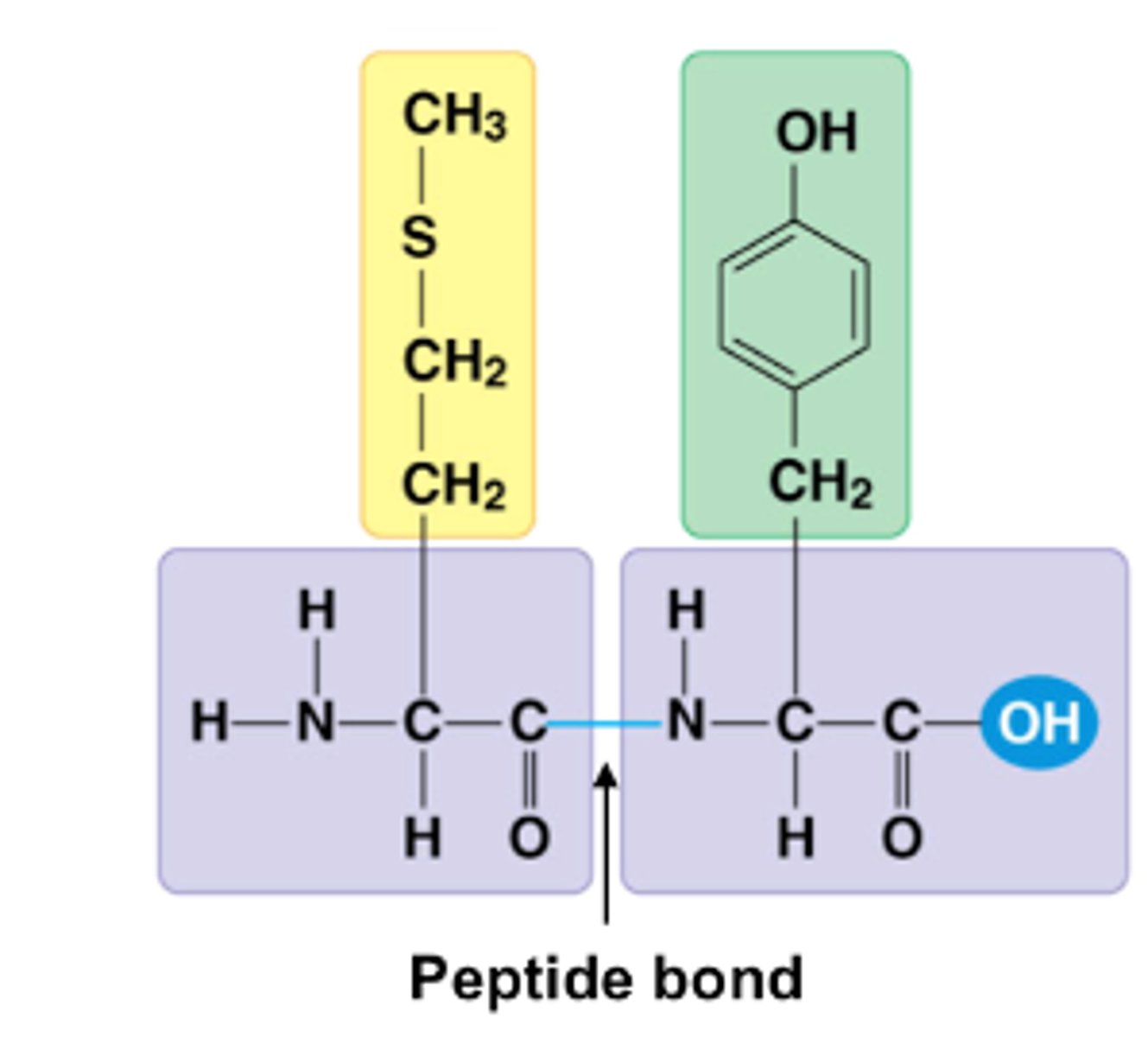
Dipeptide
When two amino acids join they form a dipeptide. In a dipeptide, the carboxyl group of one amino acid combines with the amino group of another amino acid.
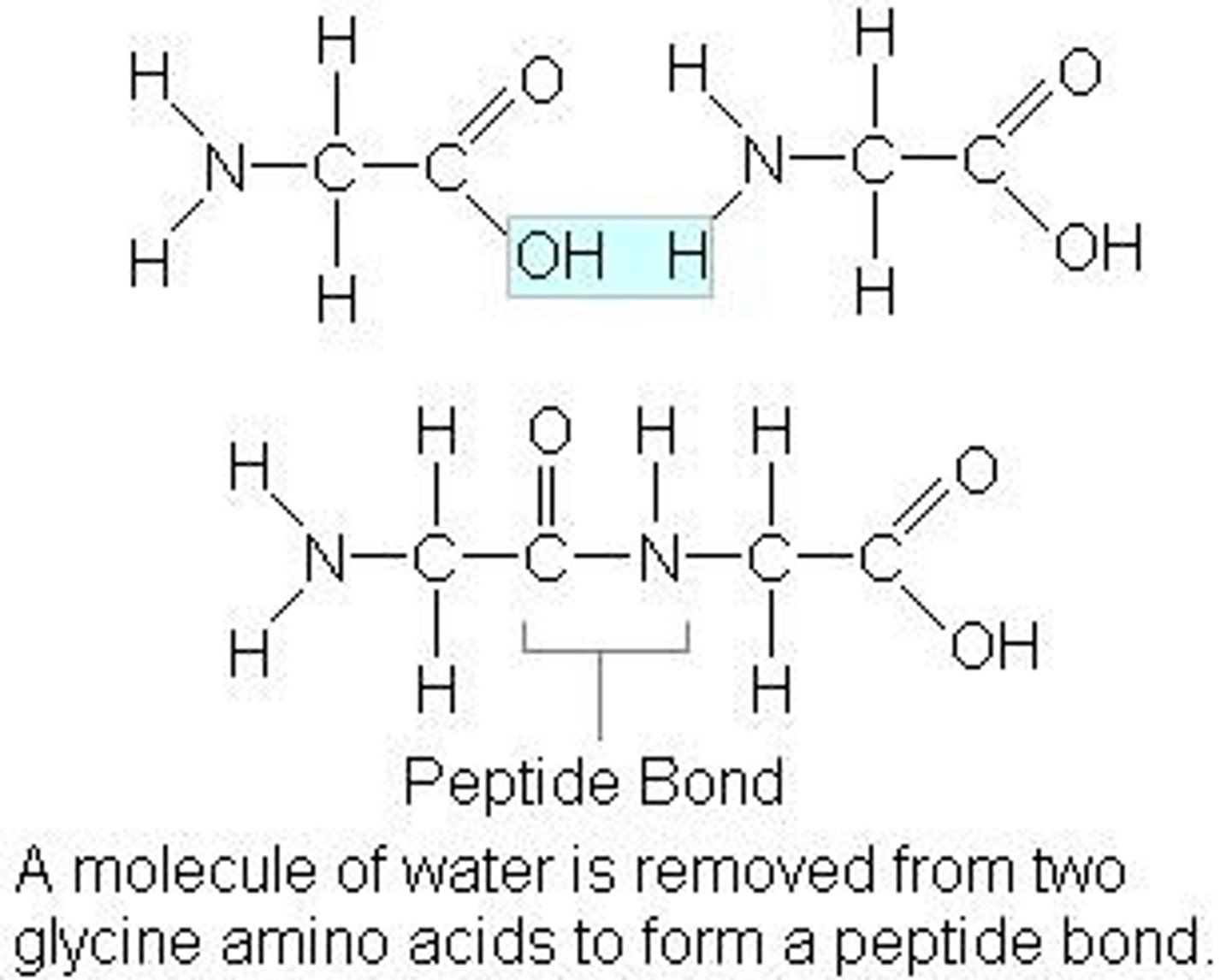
Peptide bond
A peptide bond is the bond between two amino acids.
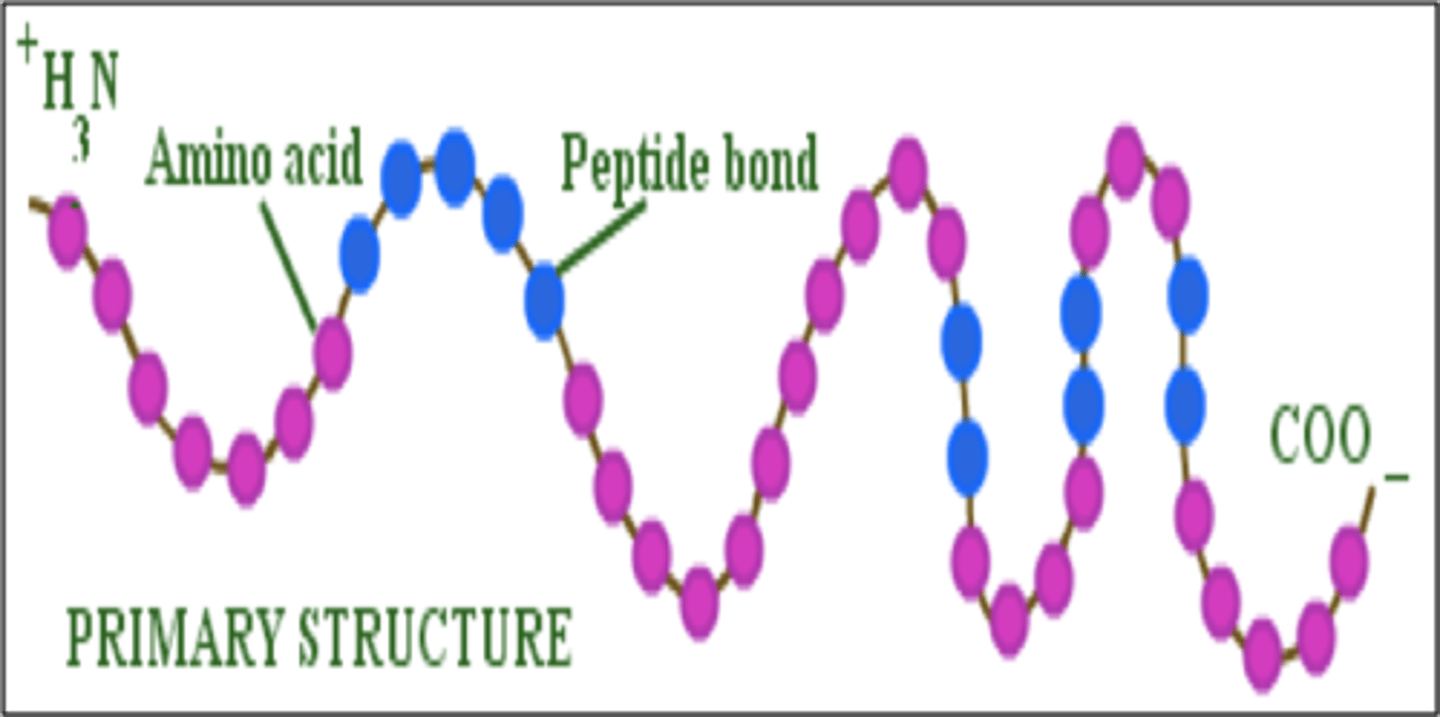
Polypeptide
If a group of amino acids are joined together in a chain, the resulting organic compound is a polypeptide, which is the primary structure of a protein.
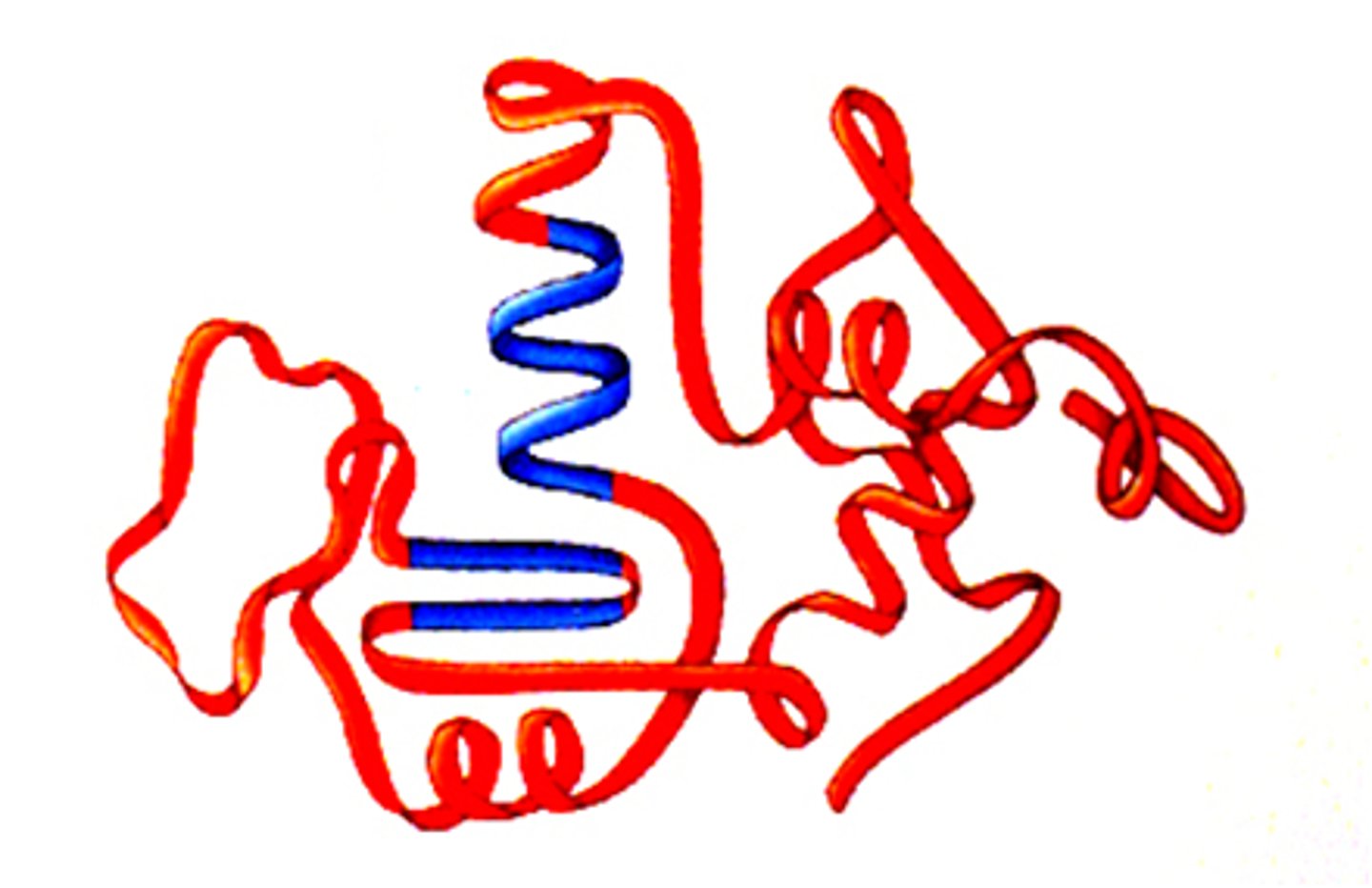
Protein
A protein is a polypeptide, a chain of amino acids, that twists and folds on itself.
Lipid
A lipid is an organic molecule consisting of carbon, hydrogen, and oxygen atoms and includes fats, oils, phospholipids, and steroids. Lipids are important because they function as structural components of cell membranes, sources of insulation, and a means of energy storage.
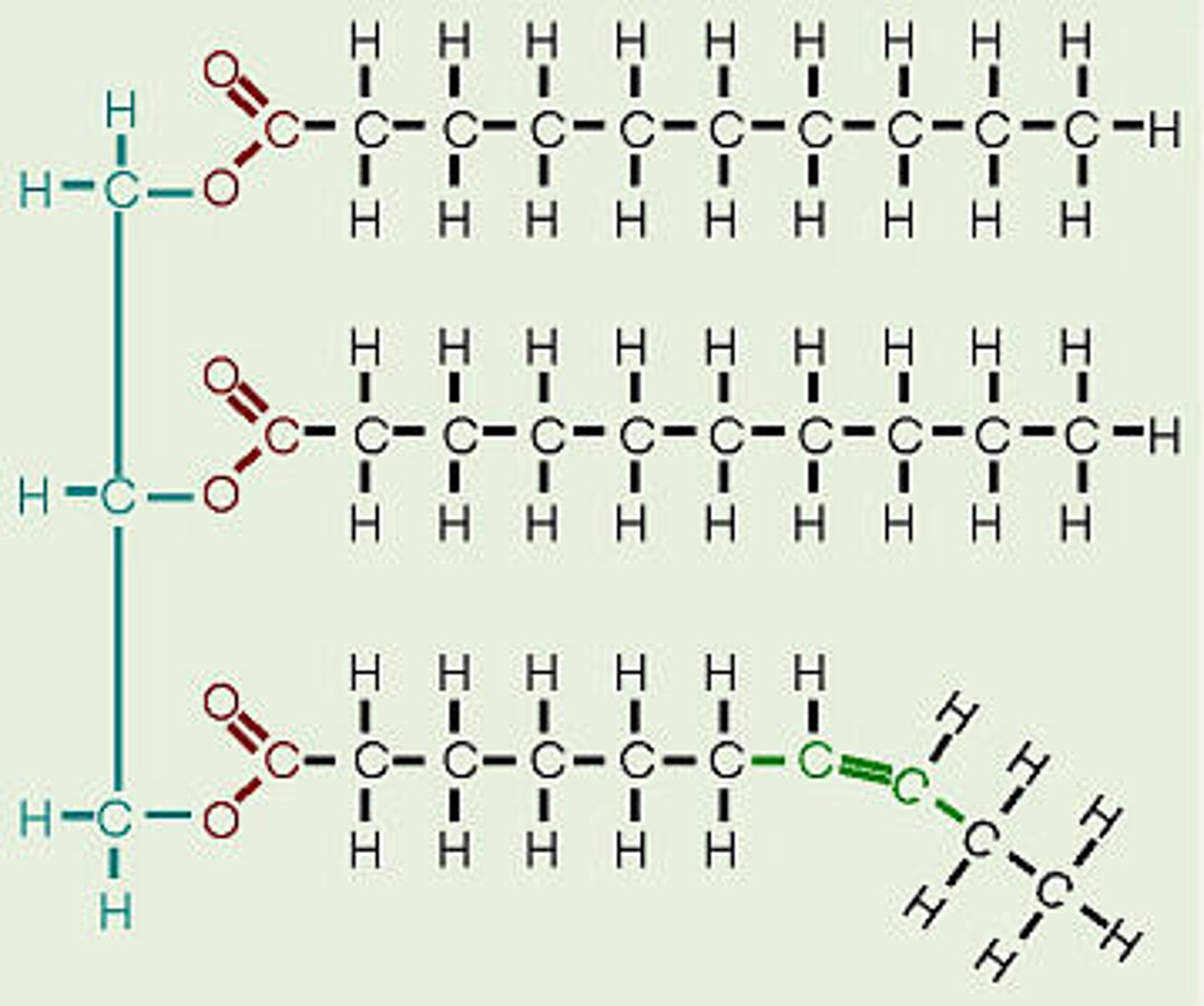
Fats
A neutral fat is the simplest lipid and consists of three fatty acids and one molecule of glycerol, also known as a triglyceride.
Oils
Oils are a type of lipid and are triglycerides that are liquid.

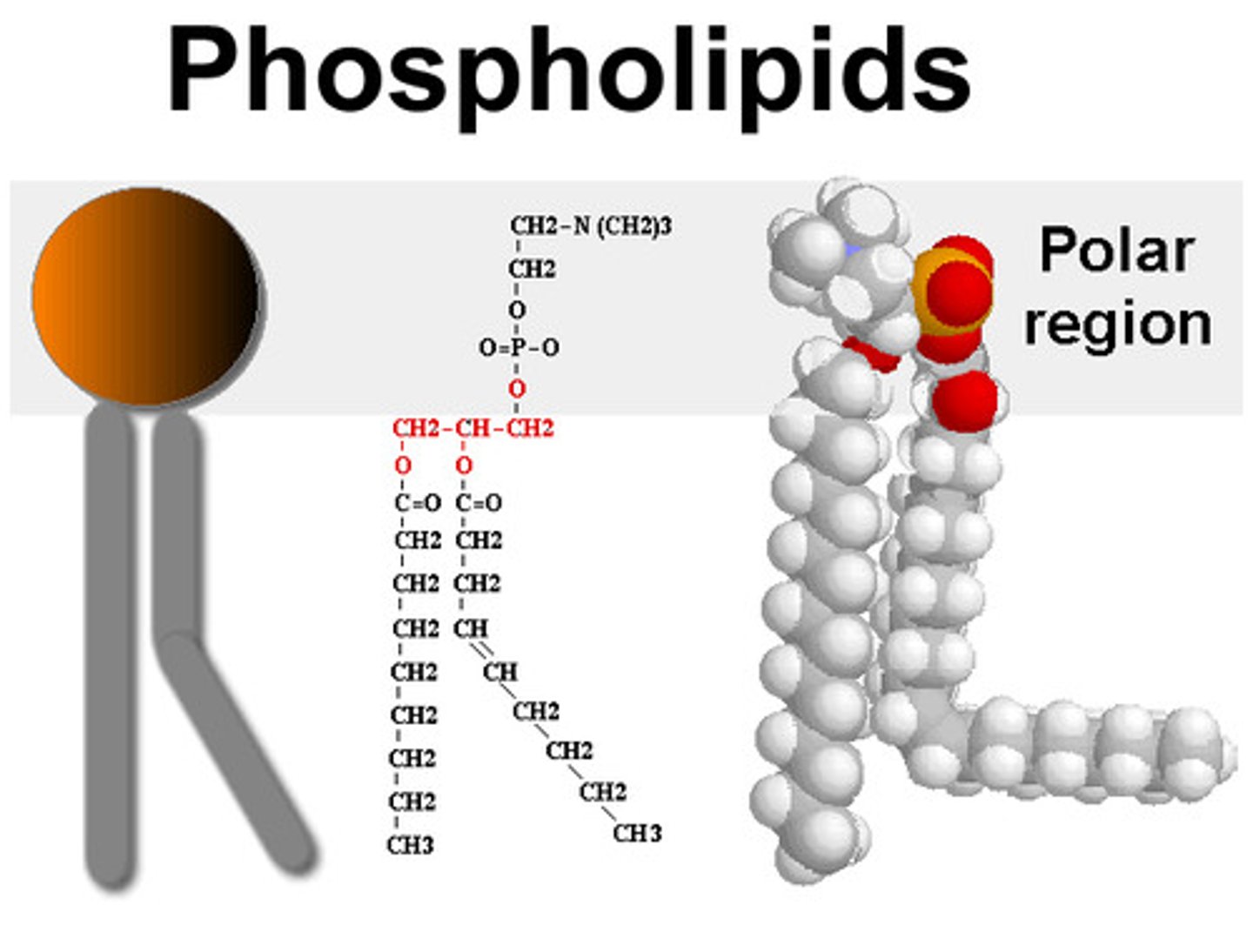
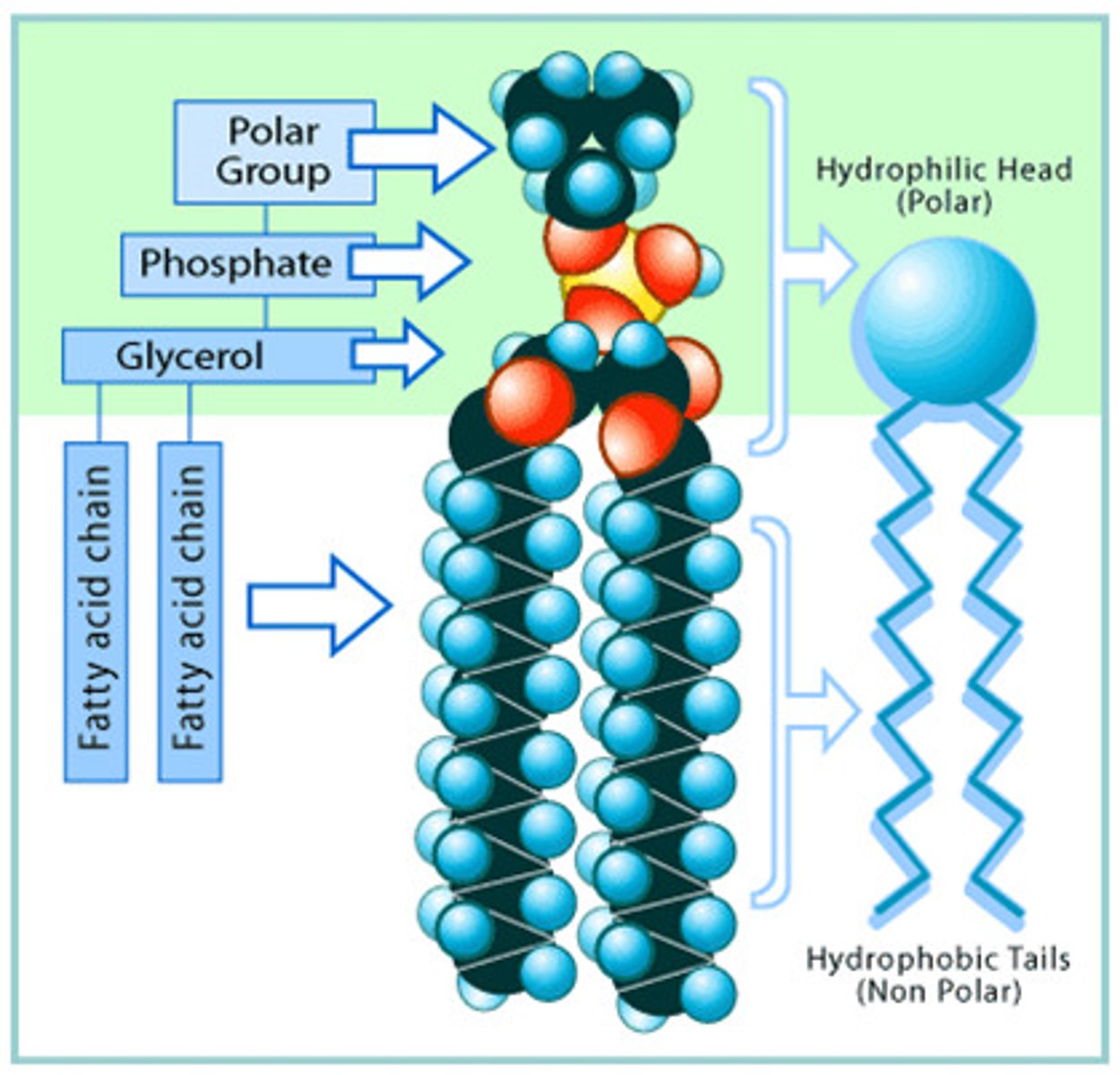
Phospholipid
Phospholipids are a class of lipids that contain two fatty acids tails and one negatively charged phosphate head. They are extremely important in their unique properties with regard to water.
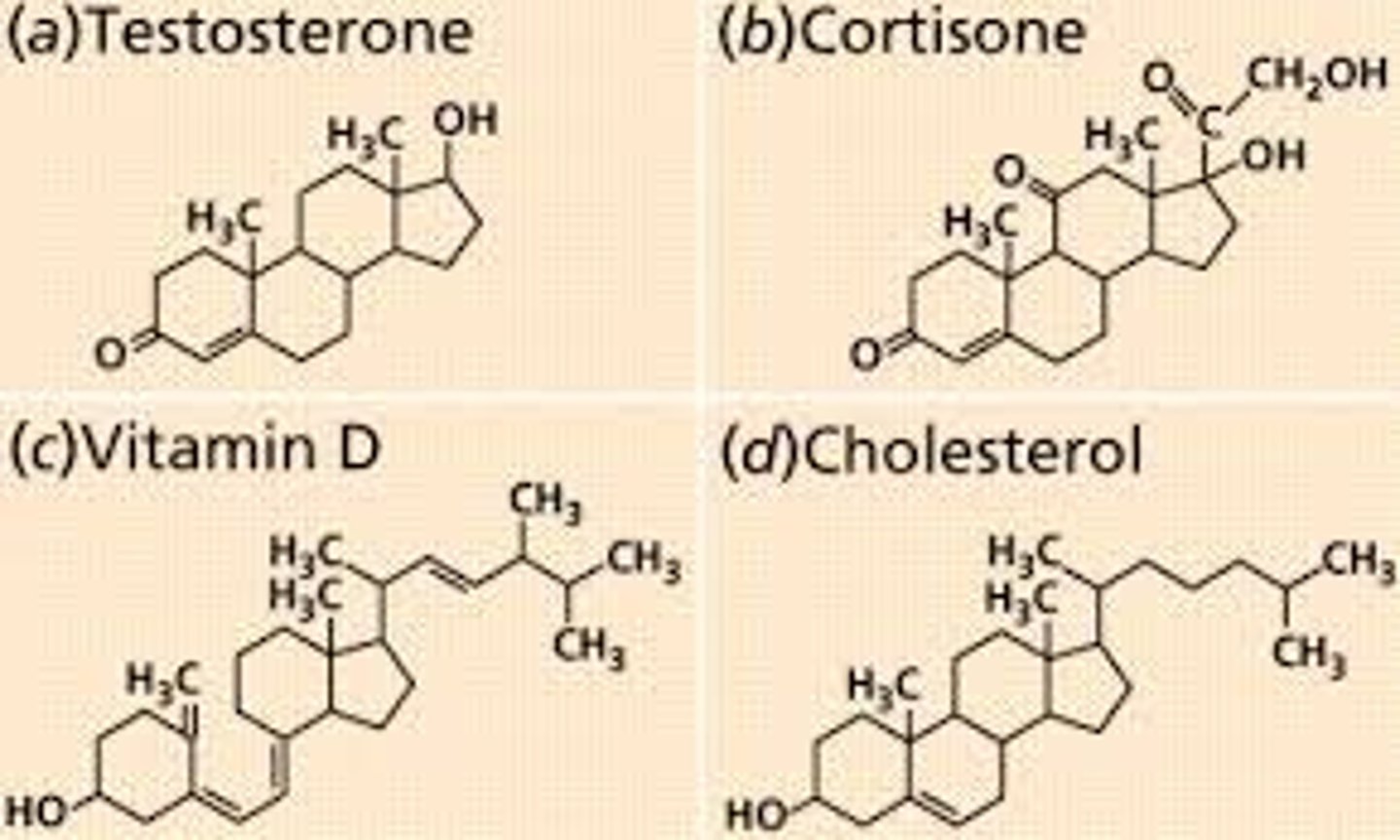
Steriods
Steroids are a class of lipids that have a basic structure of four linked carbon rings and include cholesterol, vitamin D, and a variety of hormones.
Neutral fats
Neutral fats are non polar, uncharged triglycerides that have no acidic or basic groups.
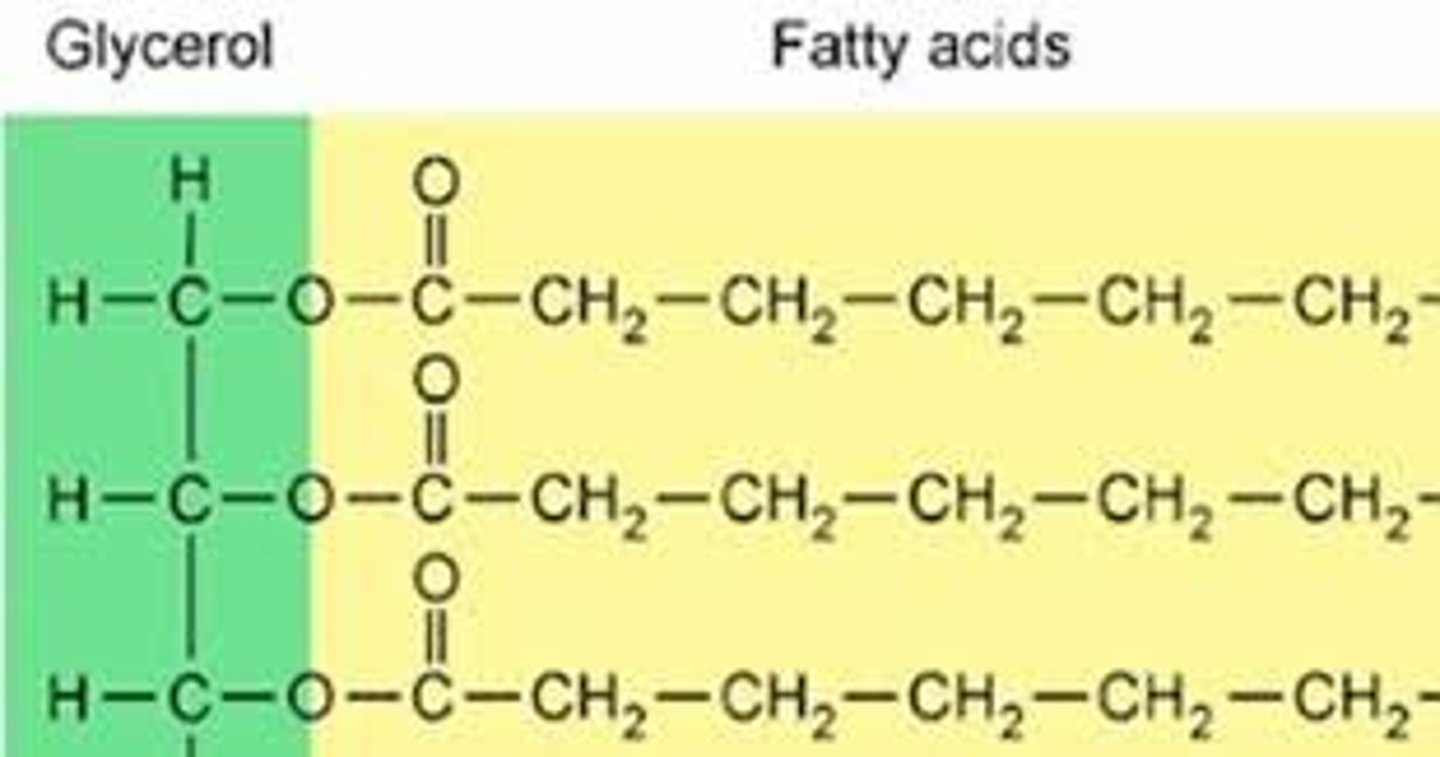
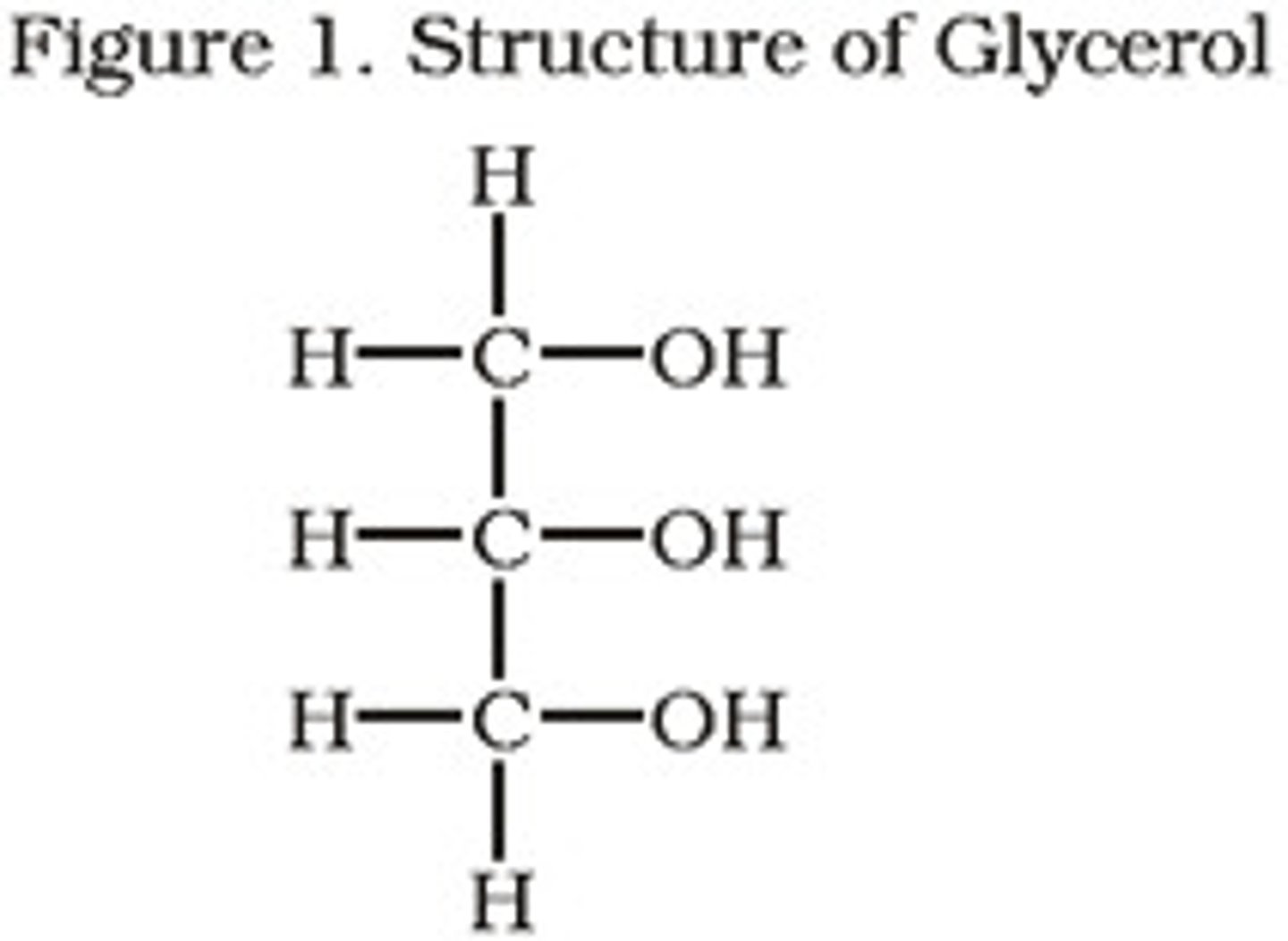
Glycerol
Glycerol is a simple polyol (sugar alcohol) compound that is found in fats.
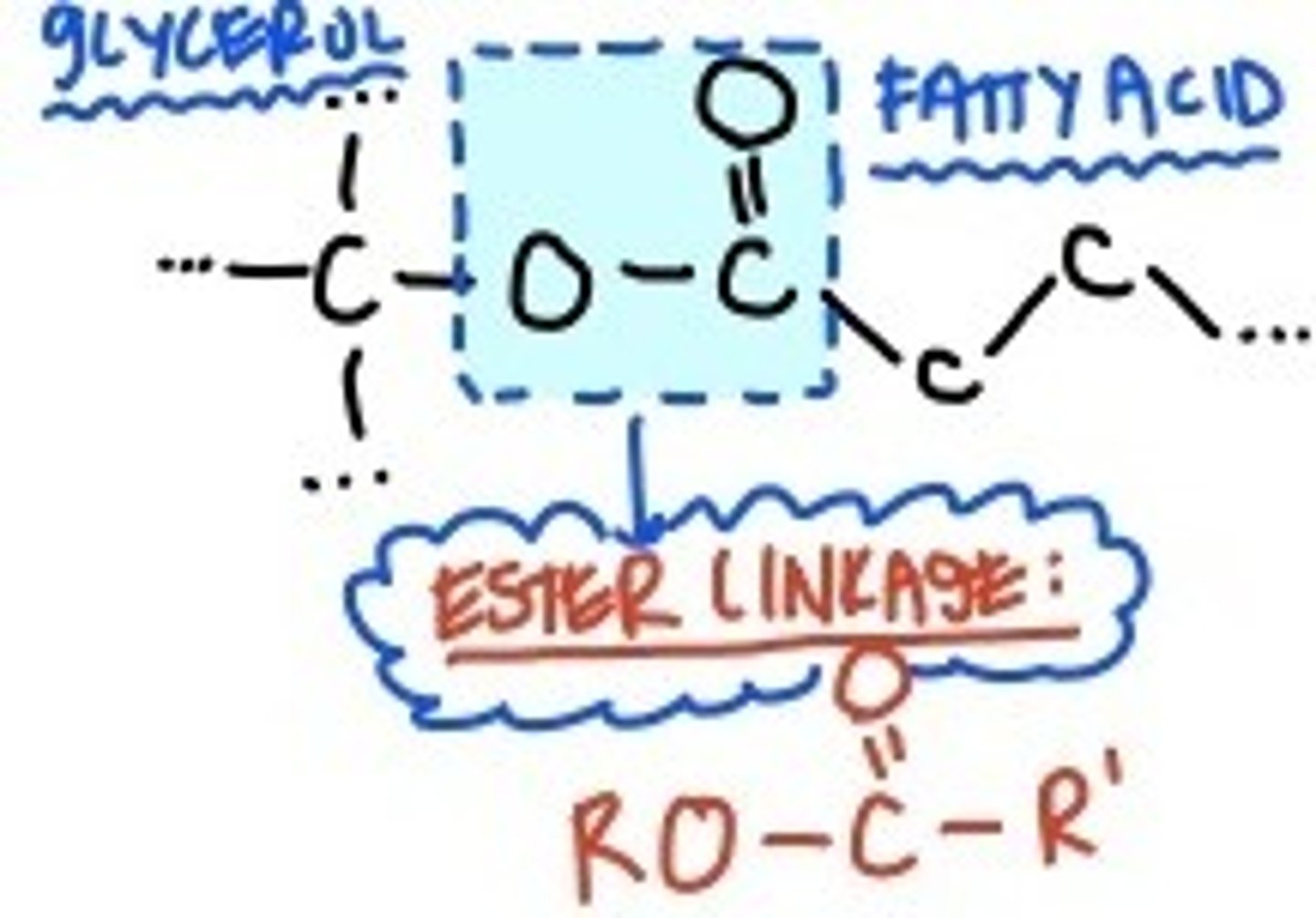
Ester linkage
The linkage formed between the glycerol molecule and the fatty acids in a fat is the ester linkage. This bond is formed through dehydration synthesis.
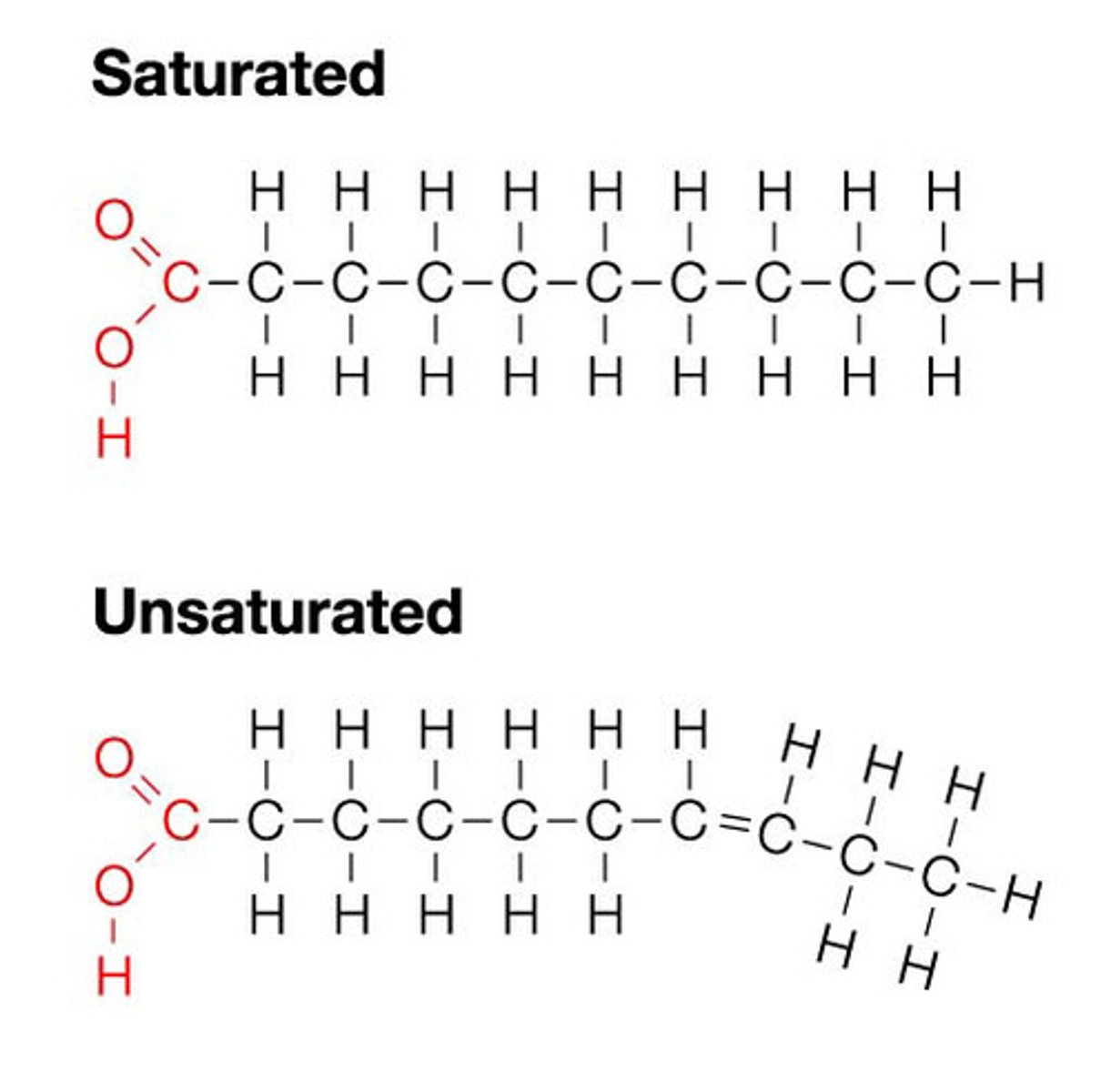
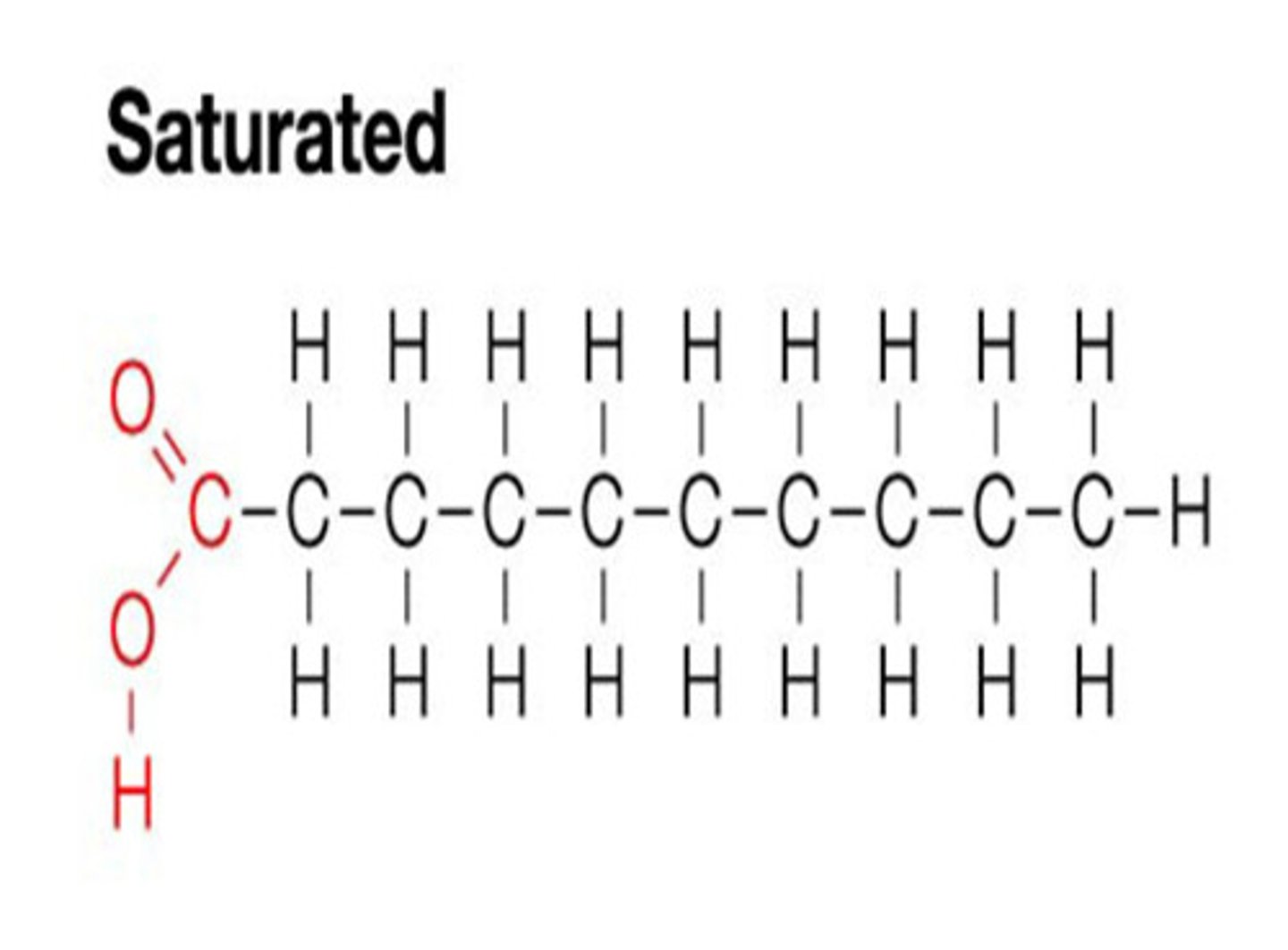
Saturated
If a fatty acid is saturated, it means it has a single covalent bond between each pair of carbon atoms.
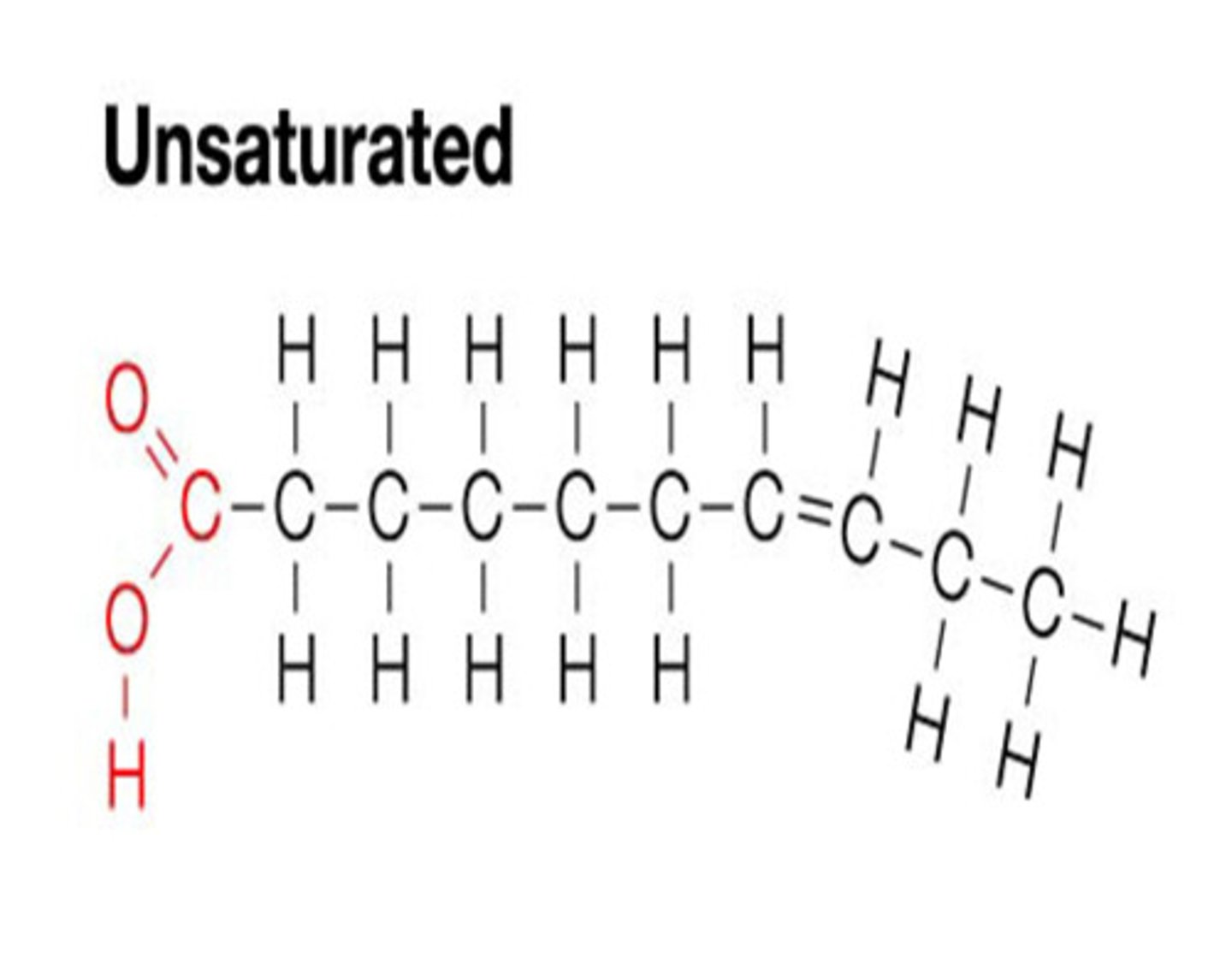
Unsaturated
If a fatty acid is unsaturated it means it has adjacent carbons that are joined by double bonds instead of single bonds.
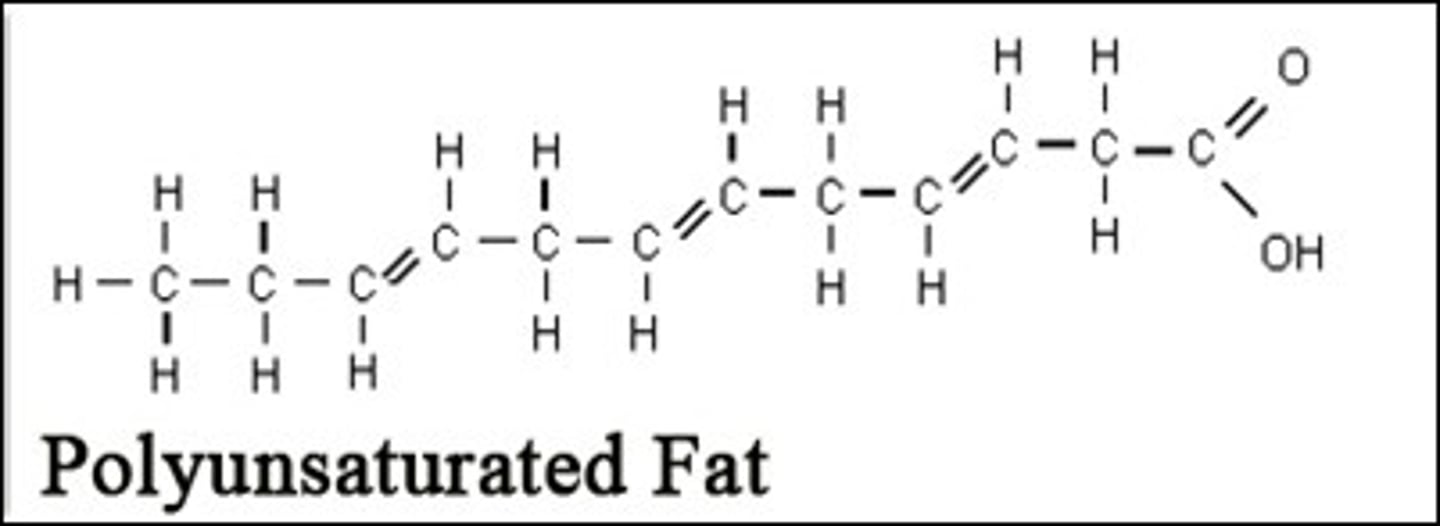
Polyunsaturated
A polyunsaturated fatty acid has many double bonds within the fatty acids.
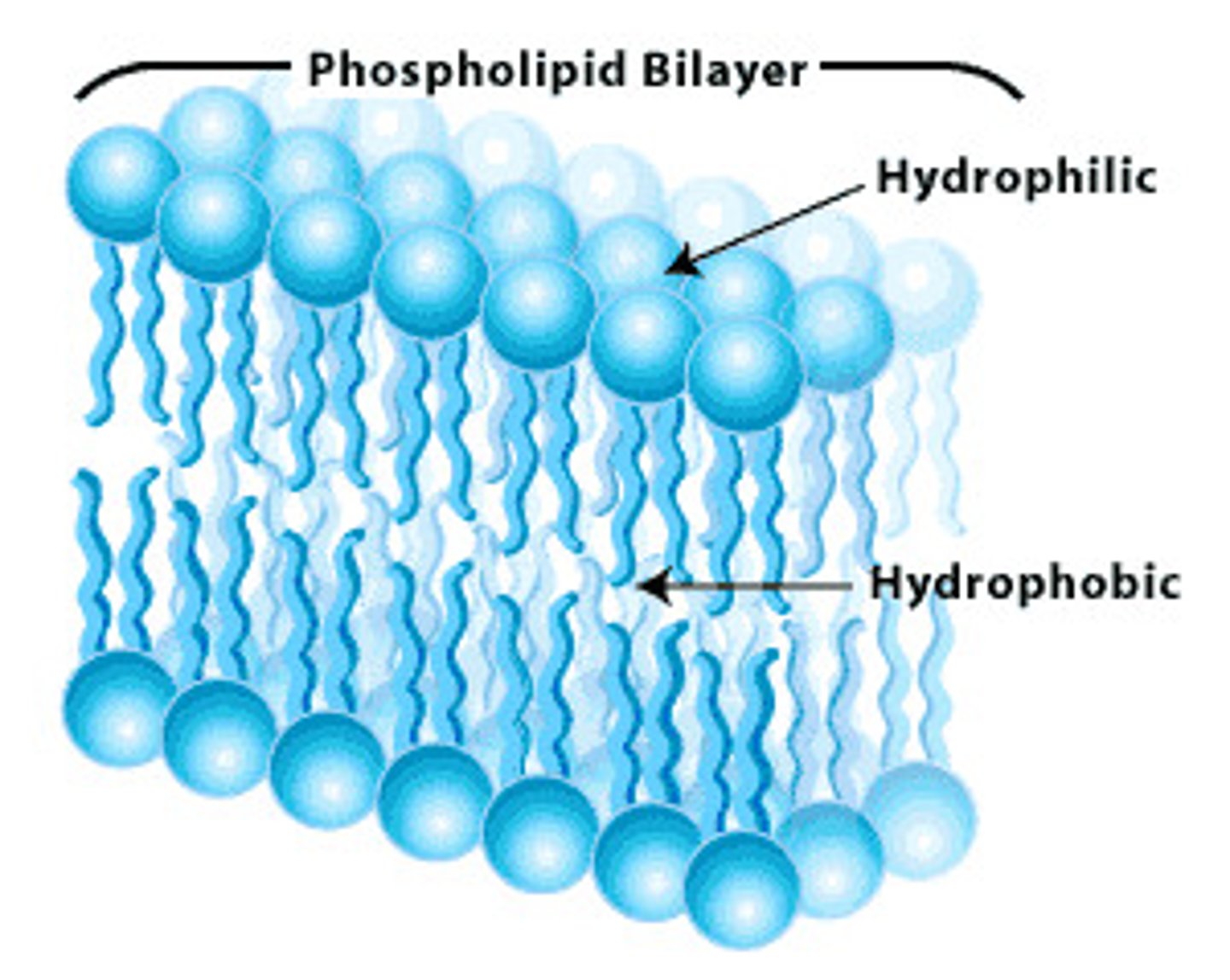
Hydrophobic
A hydrophobic molecule is a molecule that does not mix with water because it is non polar, such as a fatty acids.
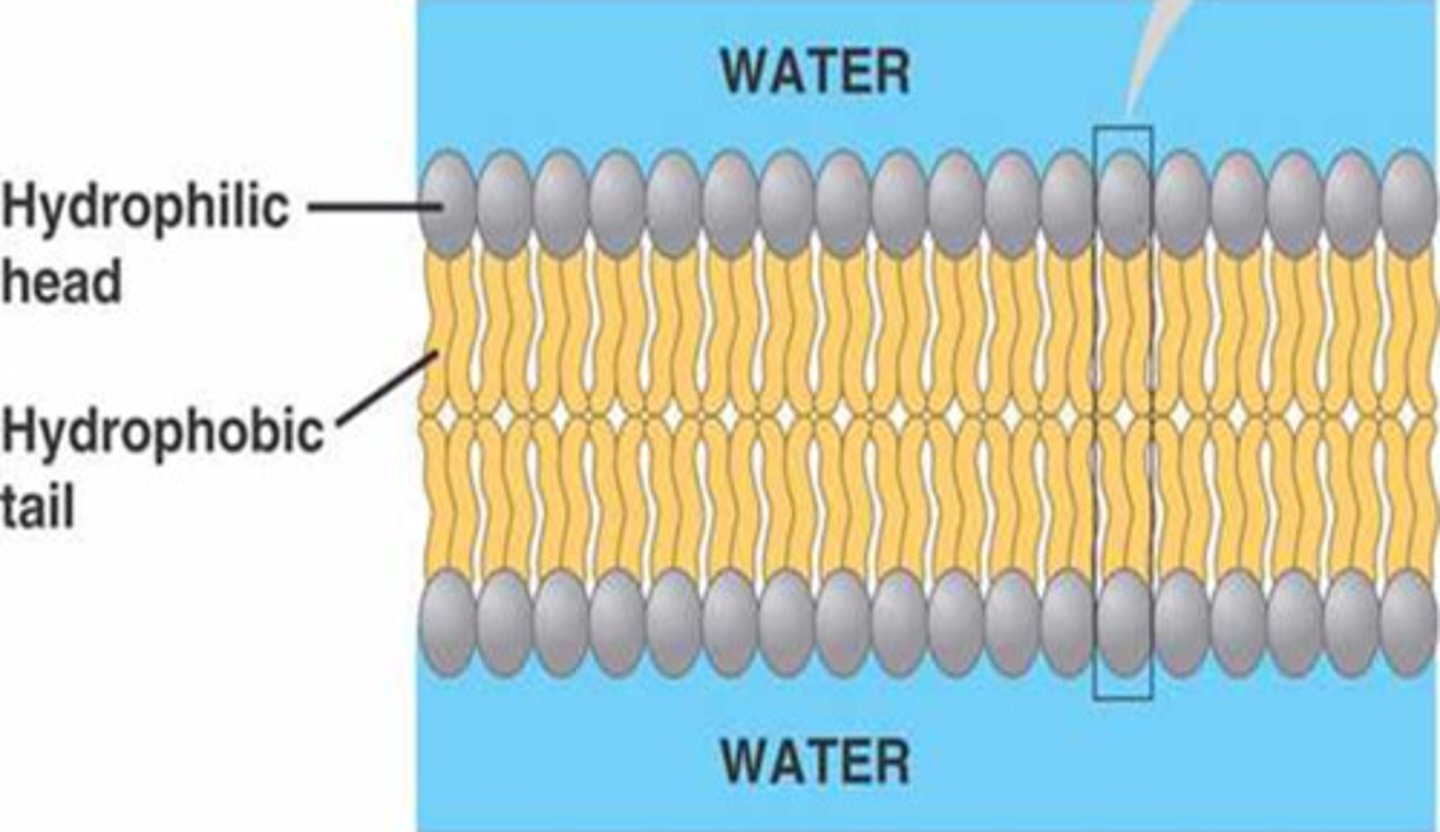
Hydrophilic
A hydrophilic molecule is a molecule that mixes with water because it is polar, such as a phosphate head in a lipid.
Amphipathic
An amphipathic molecule has both a hydrophilic region and a hydrophobic region, such as a phospholipid.
Nucleic acids
Nucleic acids are organic compounds that contain carbon, hydrogen, oxygen, nitrogen, and phosphorus. They are made up of simple units called nucleotides and include deoxyribonucleic acid and ribonucleic acid.
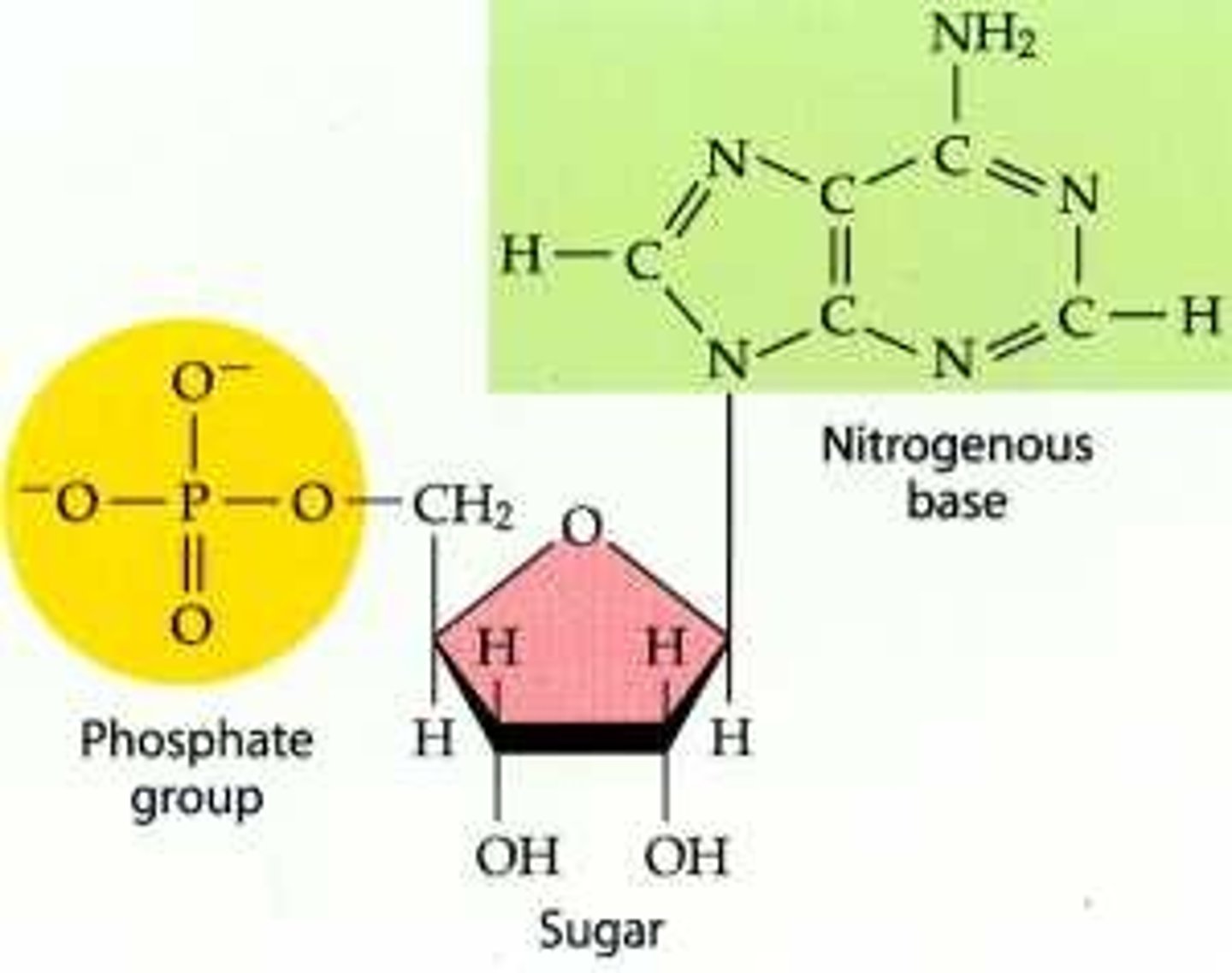
Nucleotides
Nucleotides are simple units that make up nucleic acids. A nucleotide consists of a nitrogen base, a phosphate group, and a five carbon sugar (ribose or deoxyribose).
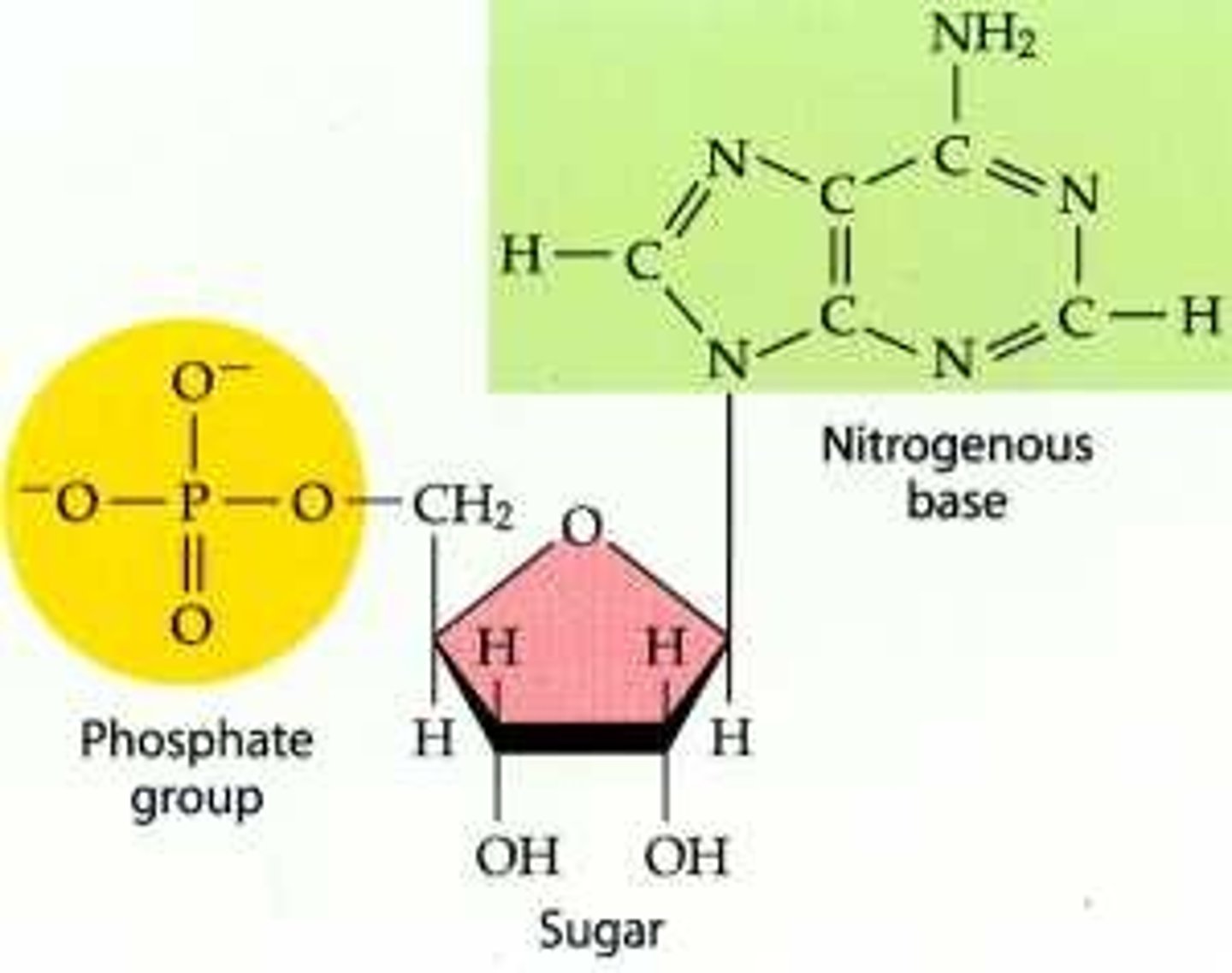
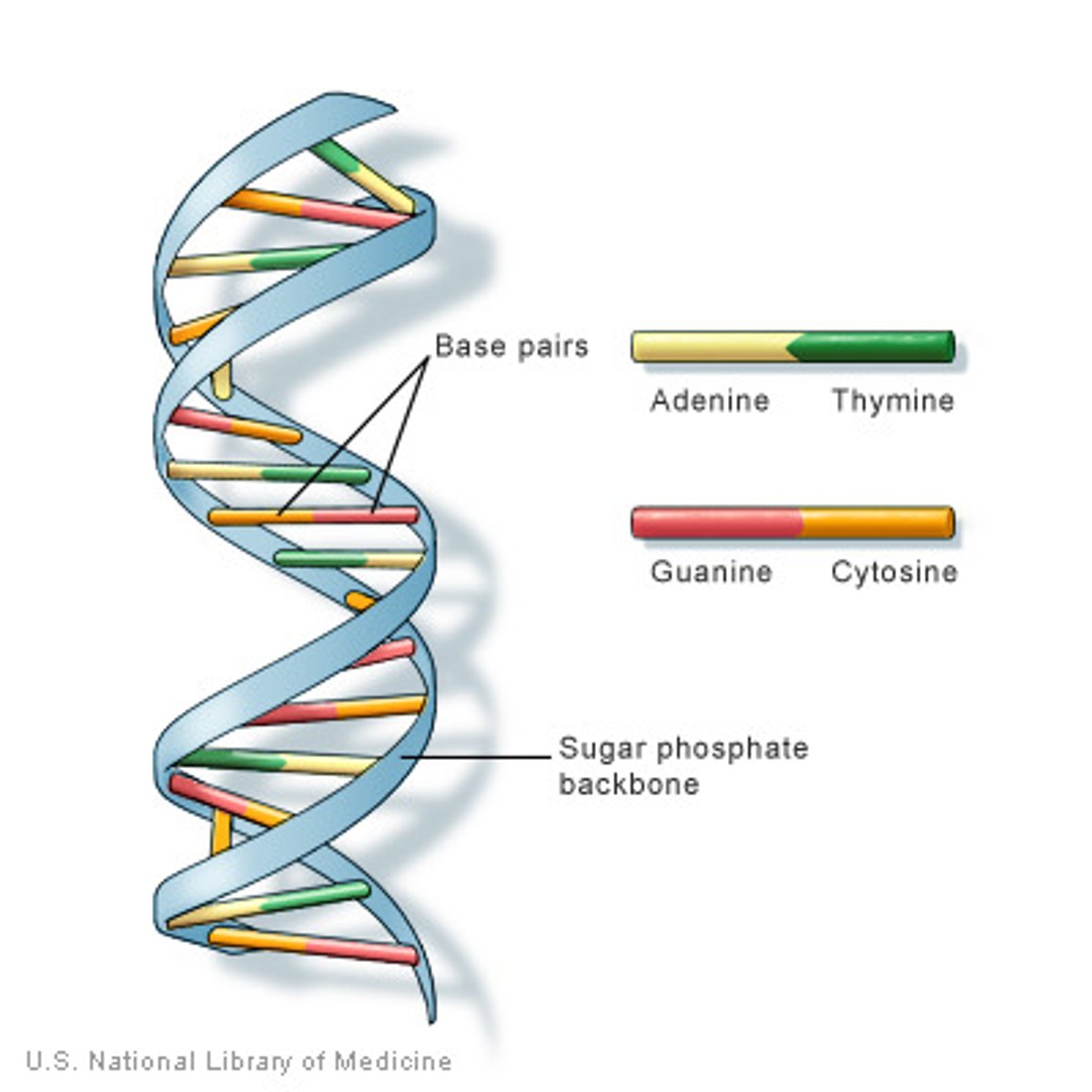
Deoxyribonucleic acid (DNA)
DNA is a nucleic acid that has a deoxyribose sugar, a phosphate group, and a nitrogen base. DNA is important because it contains genes, and it is kept in the nucleus of cells.
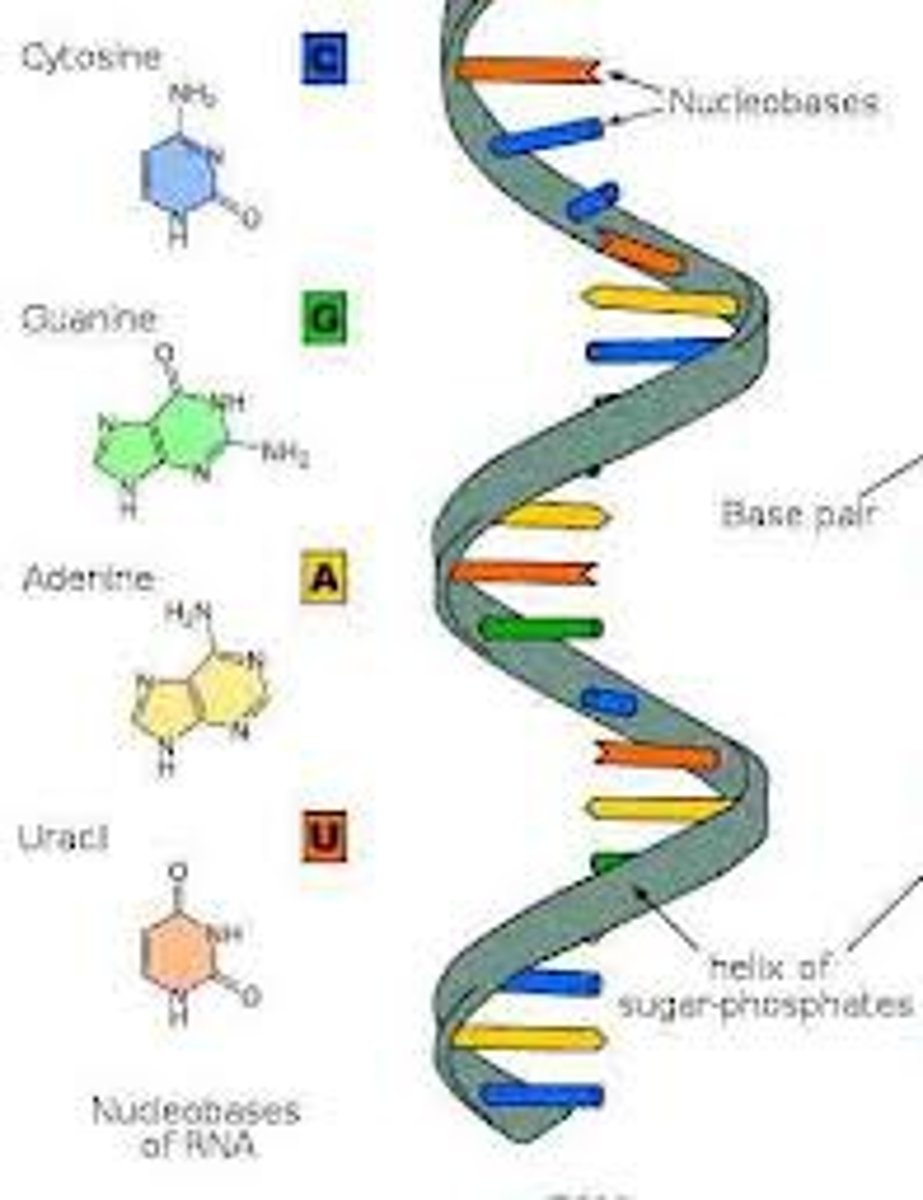
Ribonucleic acid (RNA)
RNA is a nucleic acid that has a ribose sugar, a phosphate group, and a nitrogen base. RNA is important because it has an essential role in protein synthesis.
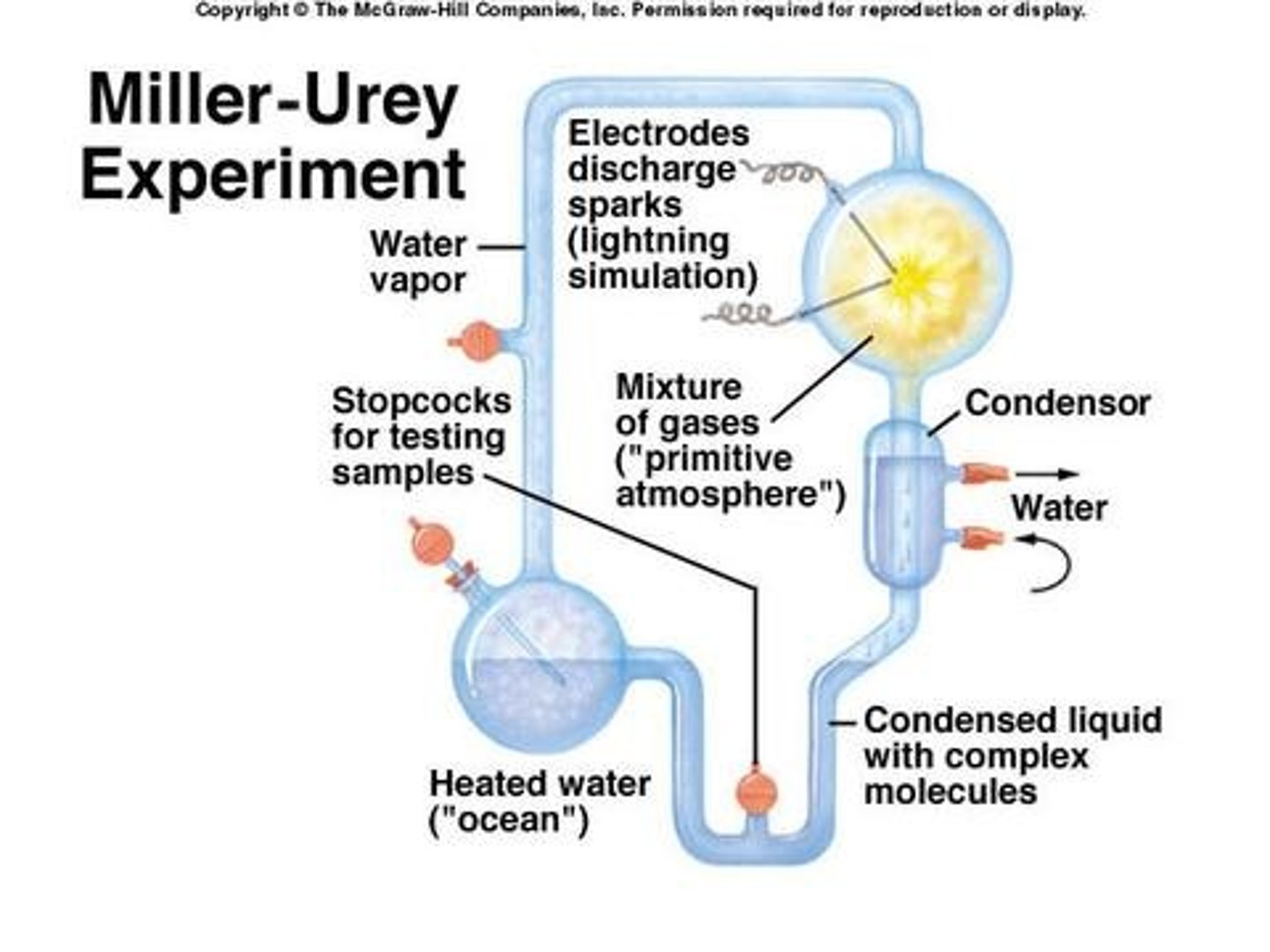
Miller and Urey
Miller and Urey were scientists who, in 1953, simulated the conditions of primitive Earth in a laboratory. They put the gases theorized to be abundant in the early atmosphere into a flask, struck them with electrical charges in order to mimic lighting, and organic compounds similar to amino acids appeared.
Heterotrophs
Heterotrophs are living organism that rely on organic molecules for food, and are also known as consumers.

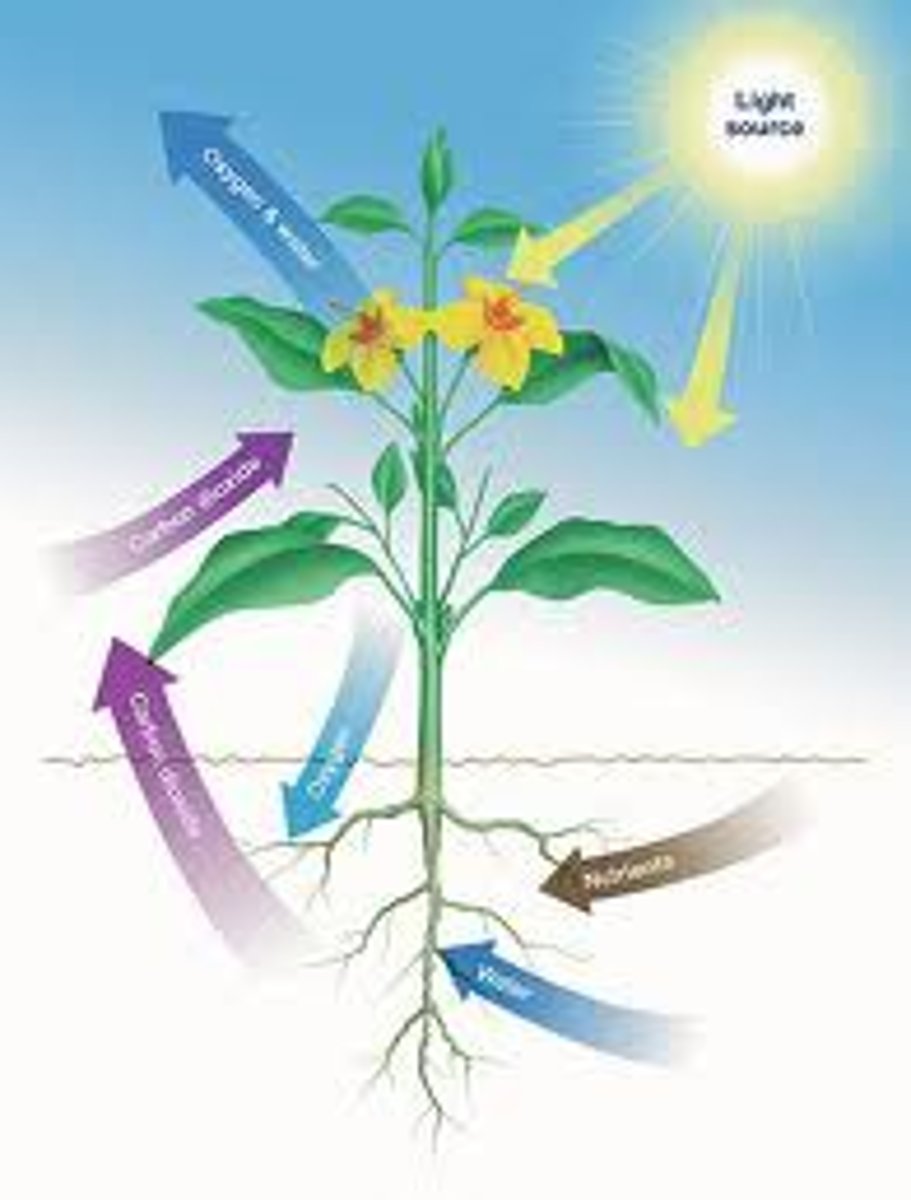
Autotrophs
Autotrophs, or producers, are organism that are able to make their own food using solar or chemical energy.
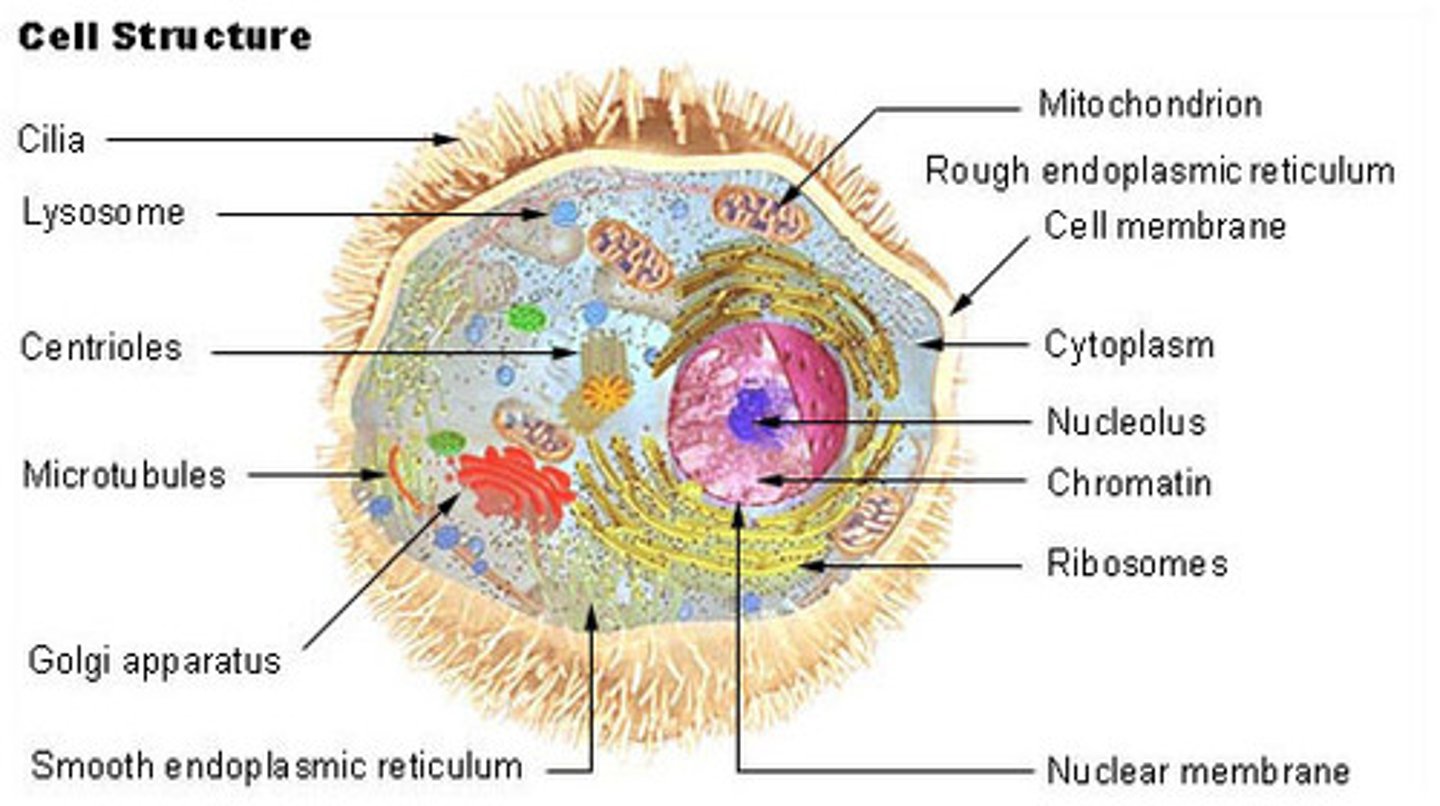
Cells
The cell is the basic unit of structure of function in life and is what all living things are composed of.
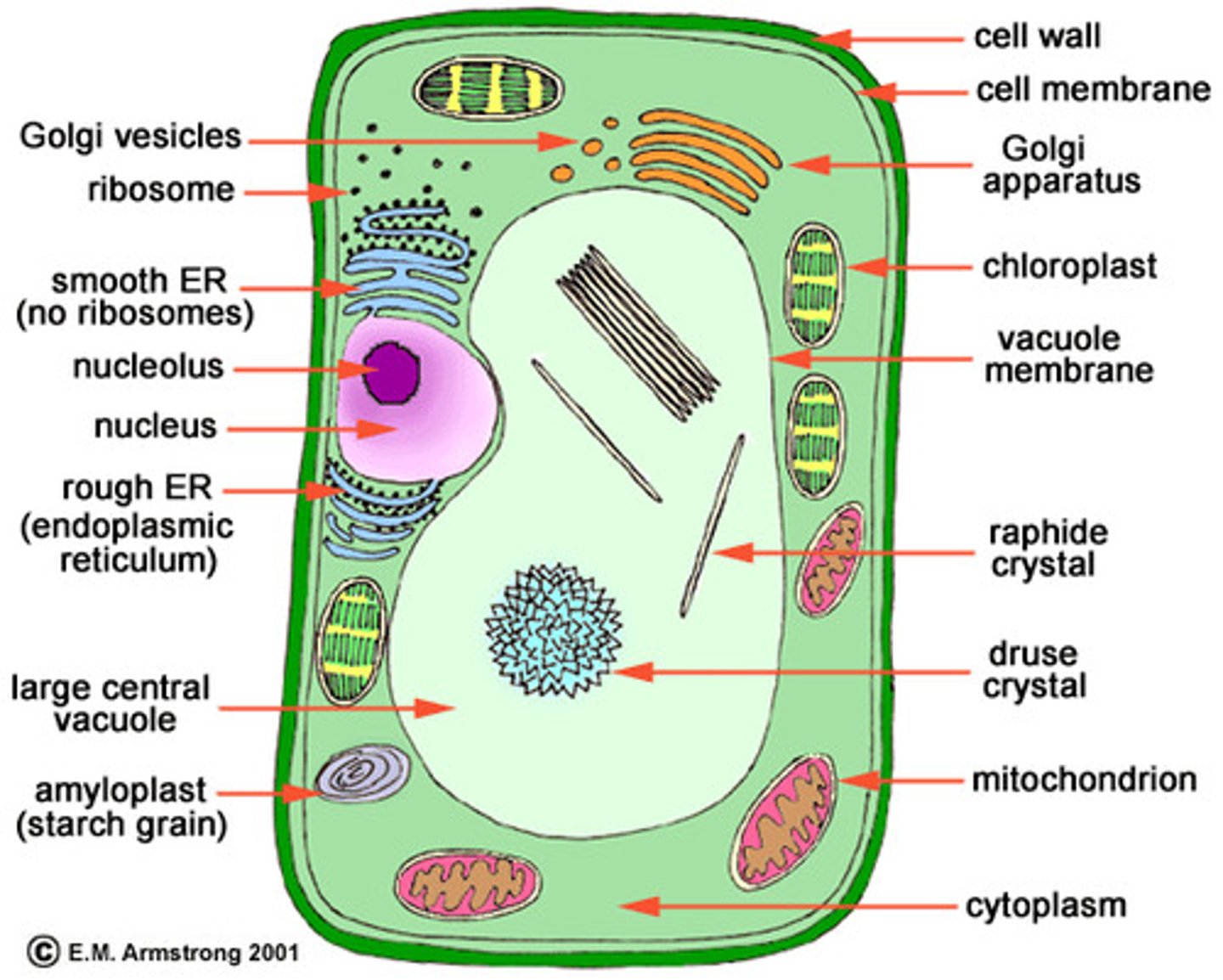
Eukaryotic cells
Eukaryotic cells contain a nucleus and cytoplasm filled with membrane bound organelles. Eukaryotic cells include fungi, protists, plant cells, and animal cells.
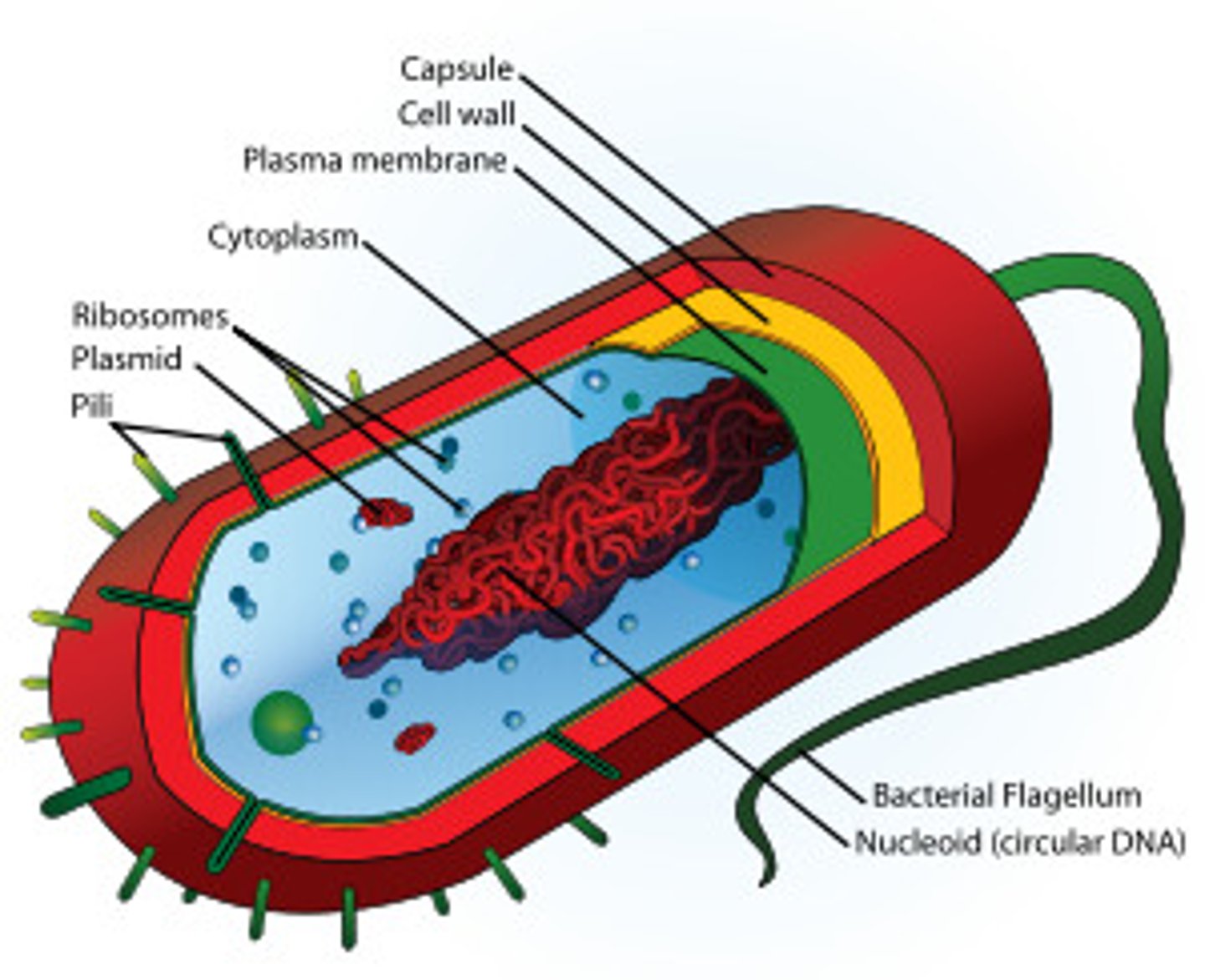
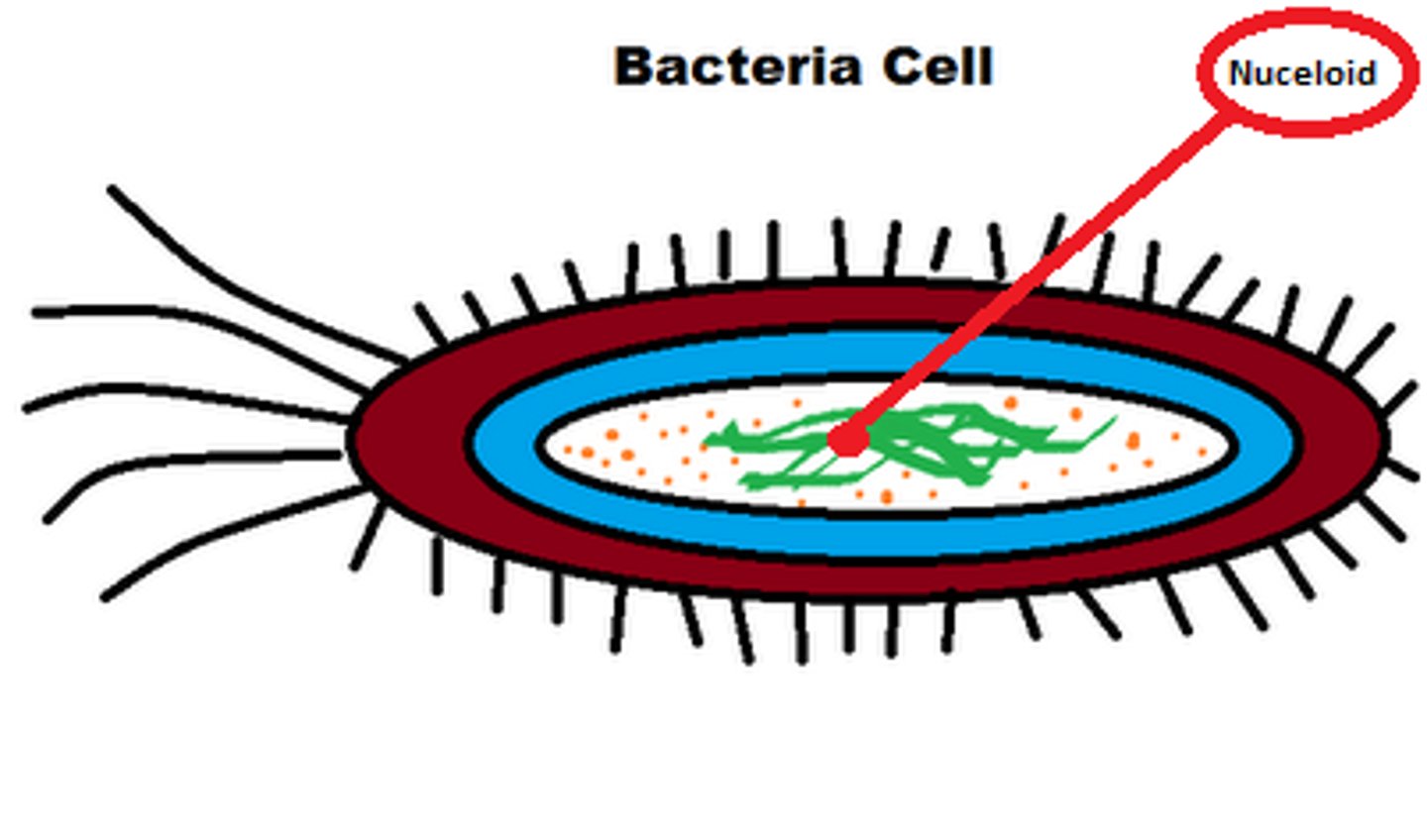
Prokaryotic cells
Prokaryotic cells are much smaller than eukaryotic cells and lack both a nucleus and membrane bound organelles. In a prokaryote, circular DNA lies free in the nucleoid . Most prokaryotes have a cell wall composed of peptidoglycan and may also have ribosomes or flagella.
Cytoplasm
Cytoplasm is a thick solution that fills each cell and contains the organelles. The cytoplasm is composed of water, salt, and proteins and helps contain the organelles and may have important enzymes to break down large molecules.
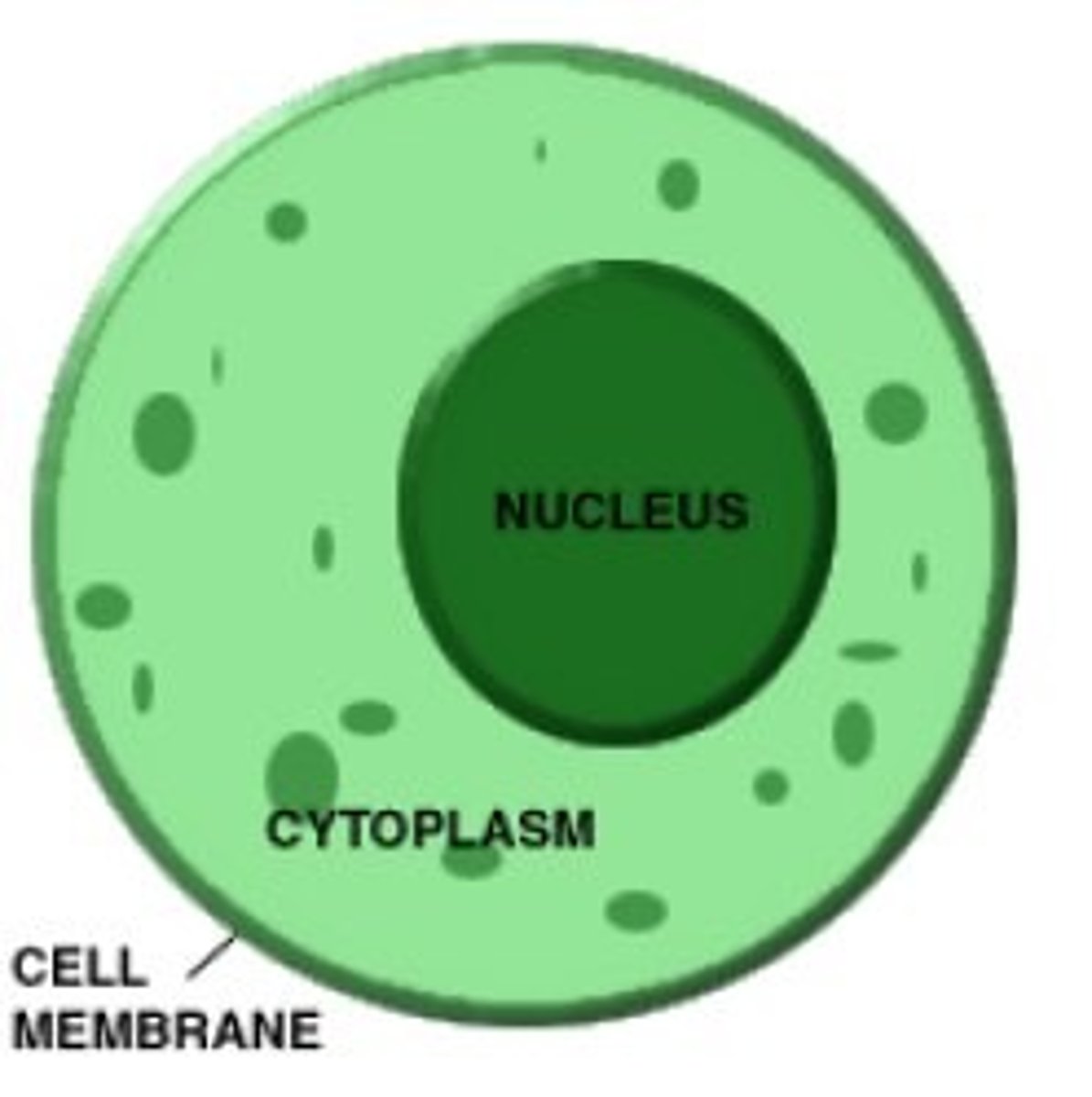
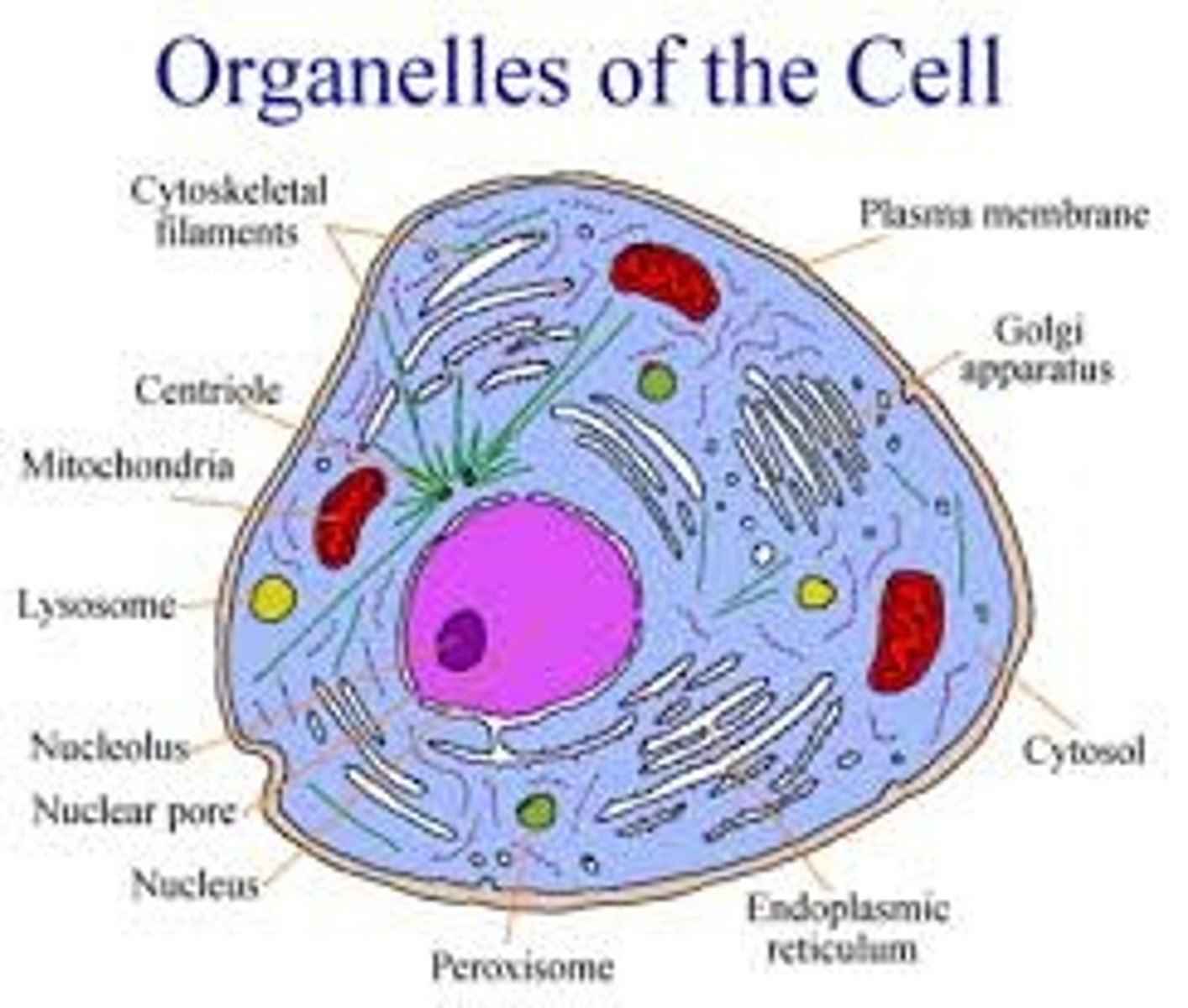
Organelles
Organelles are small units suspended in the cytoplasm which carry out a specific function to help the cell.
Nucleoid
The nucleoid is the area in a prokaryotic cell in which the circular DNA molecule lies free in the cell.
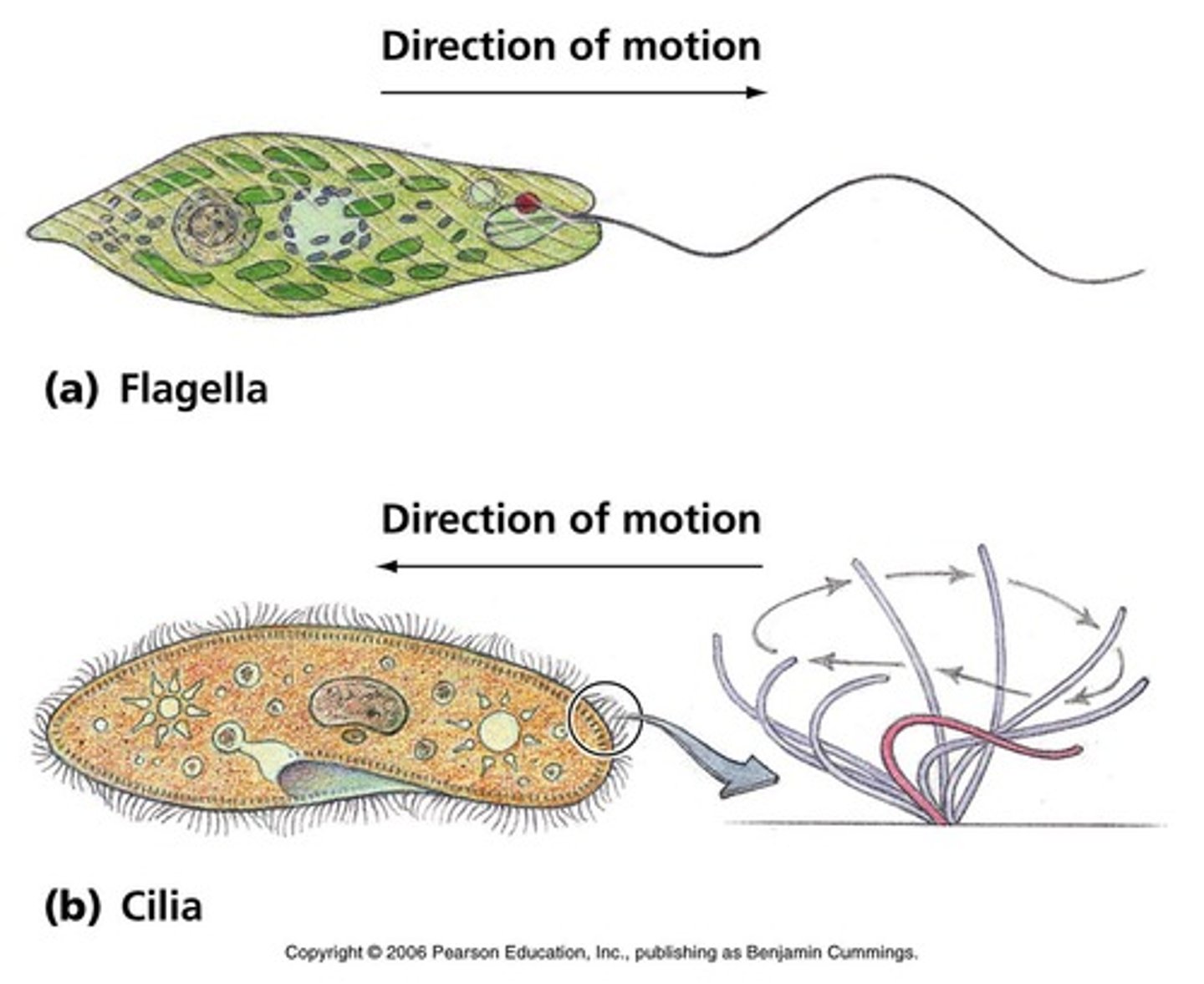
Flagella
Flagella are long projections on a cell used for motility. They are often found on single cell organisms but are sometimes present in larger organisms, such as on sperm.
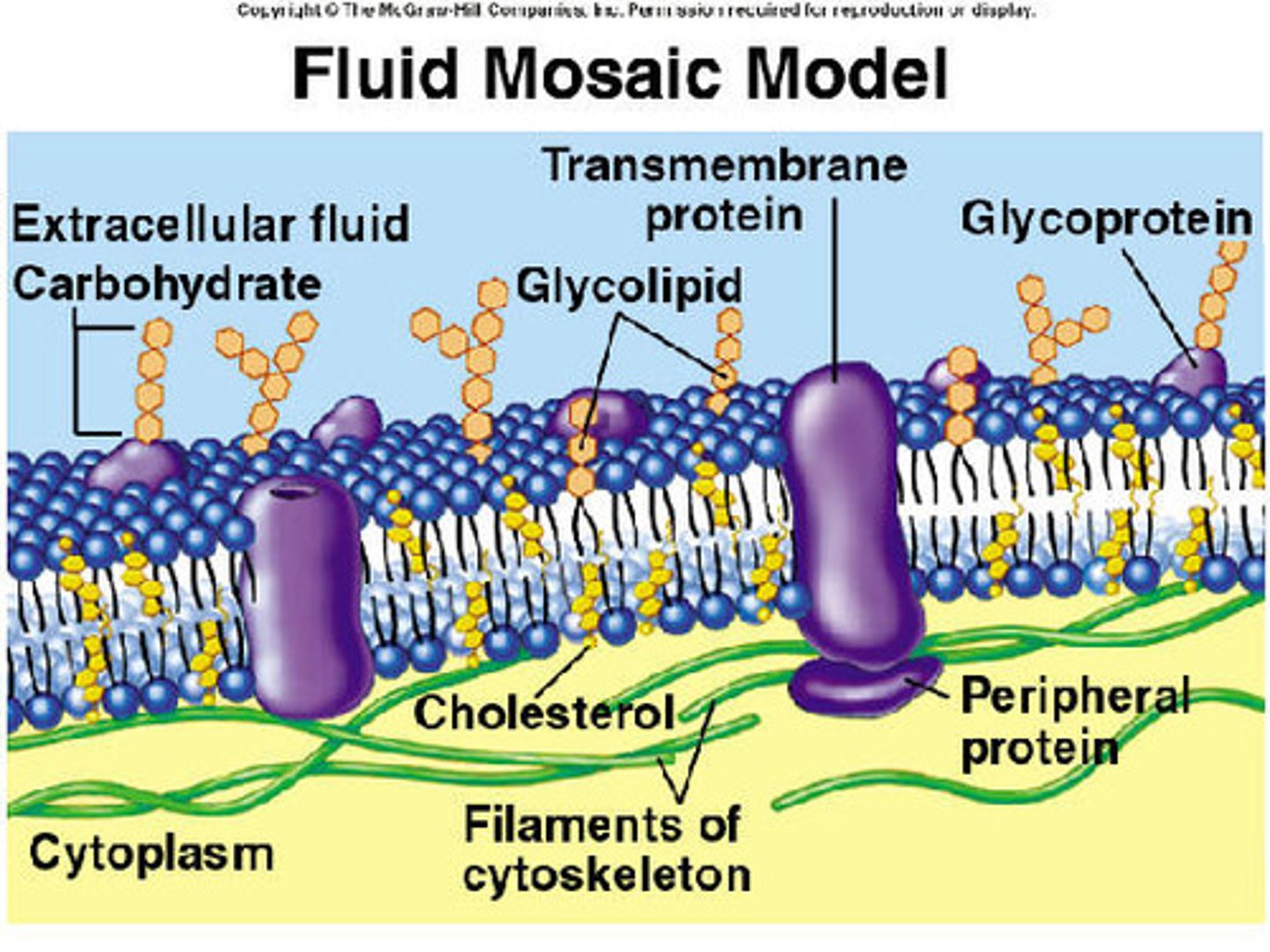
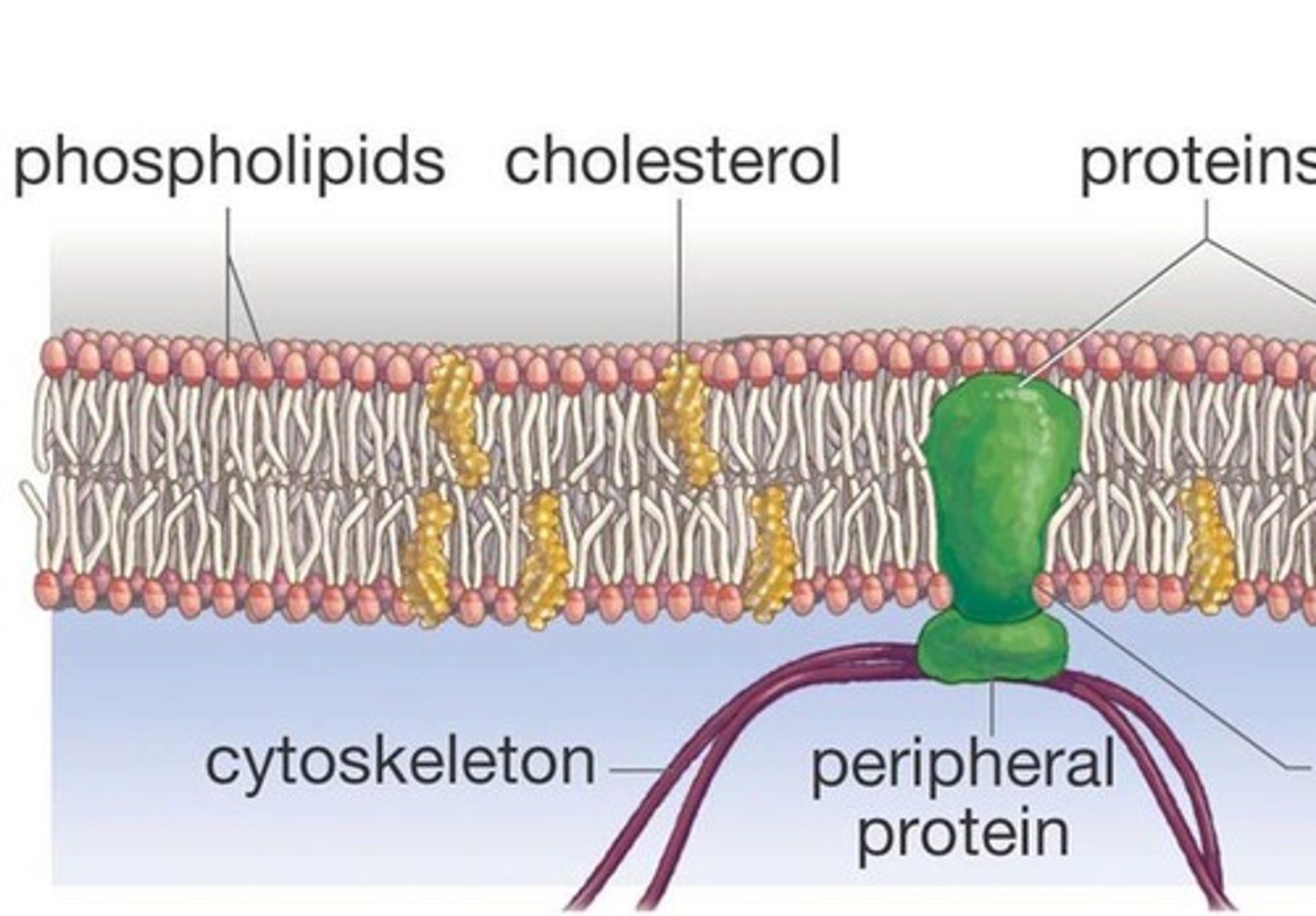
Plasma membrane
The plasma membrane is a double layered structure made up of phospholipids and proteins that serves as an outer envelope for cells. In the membrane, hydrophobic fatty acid tails face inwards and hydrophilic phosphate heads face outwards. The membrane is semi-permeable and regulated the movement of substances in and out of the cell.
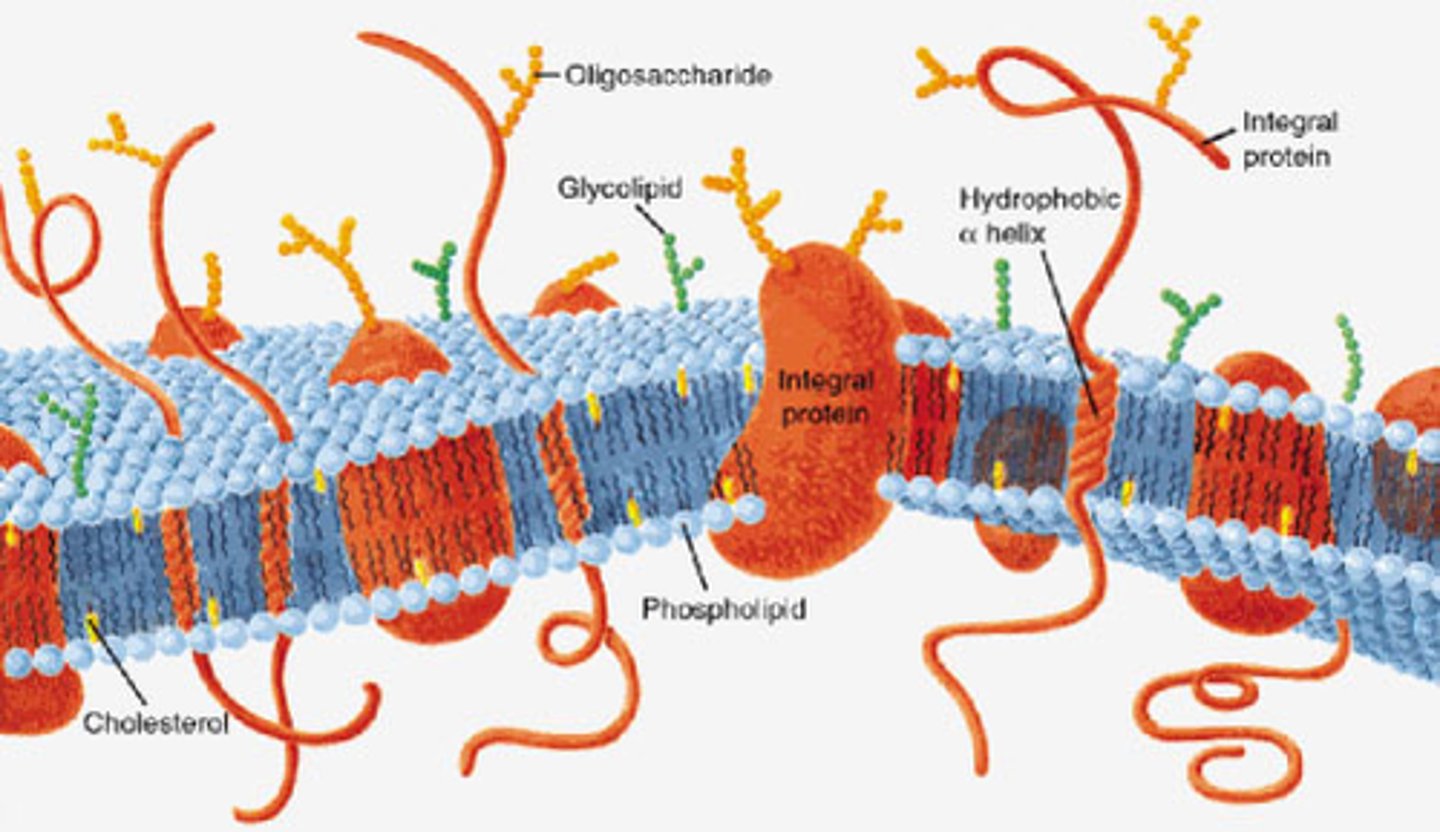
Peripheral proteins
Peripheral proteins are proteins that are loosely associated with the lipid bilayer and only temporarily attach to the membrane. They are located on the inner or outer surface of the membrane.
Integral proteins
Integral proteins are firmly bound to the plasma membrane and are amphipathic.

Transmembrane proteins
Transmembrane proteins are integral proteins that do not extend all the way through the membrane.

Fluid-mosaic model
The fluid-mosaic model refers to the arrangement of phospholipids and proteins in the plasma membrane of cells.
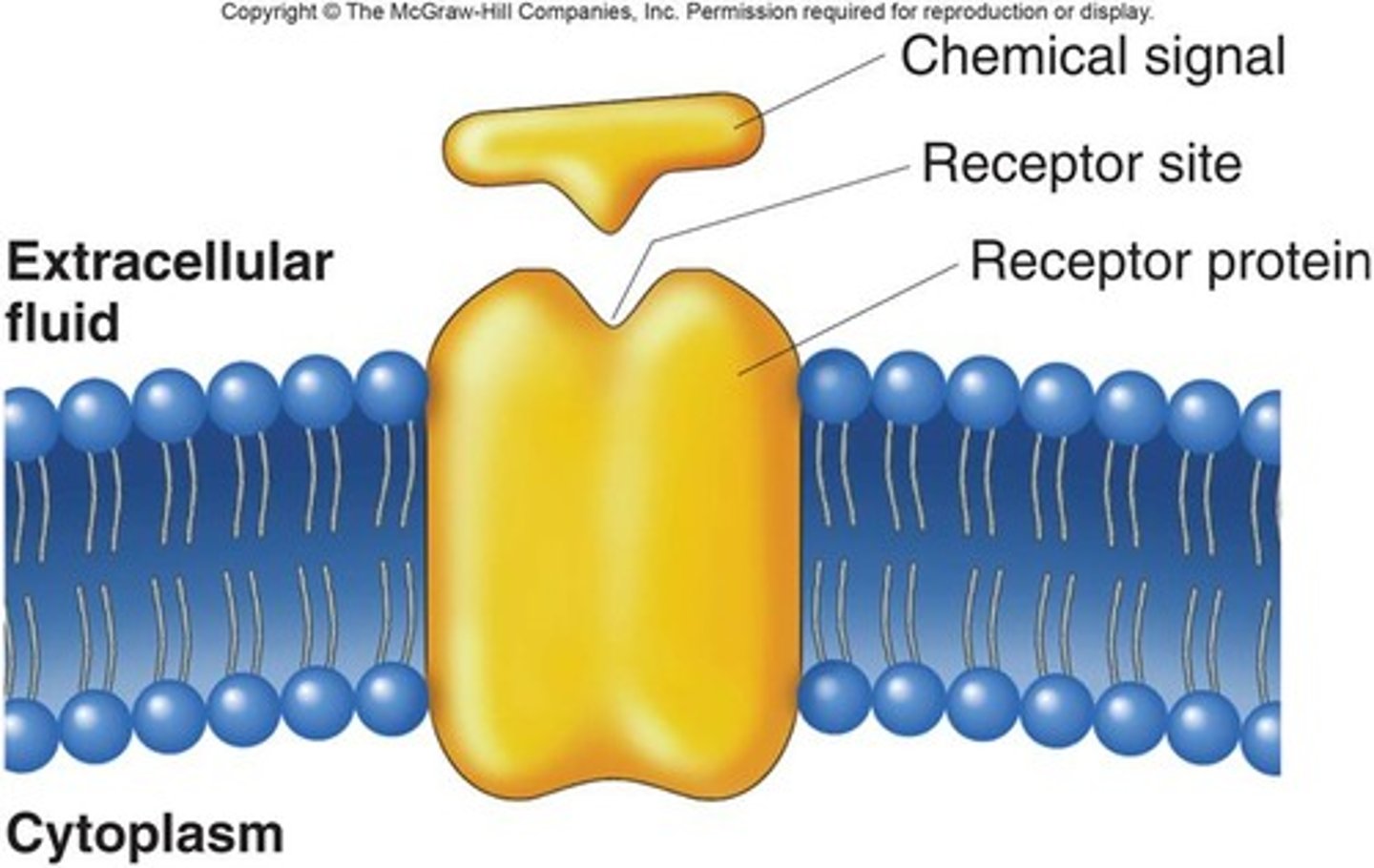
Receptor proteins
Receptor proteins are membrane proteins that serve as docking sites for proteins of the extracellular matrix or hormones.
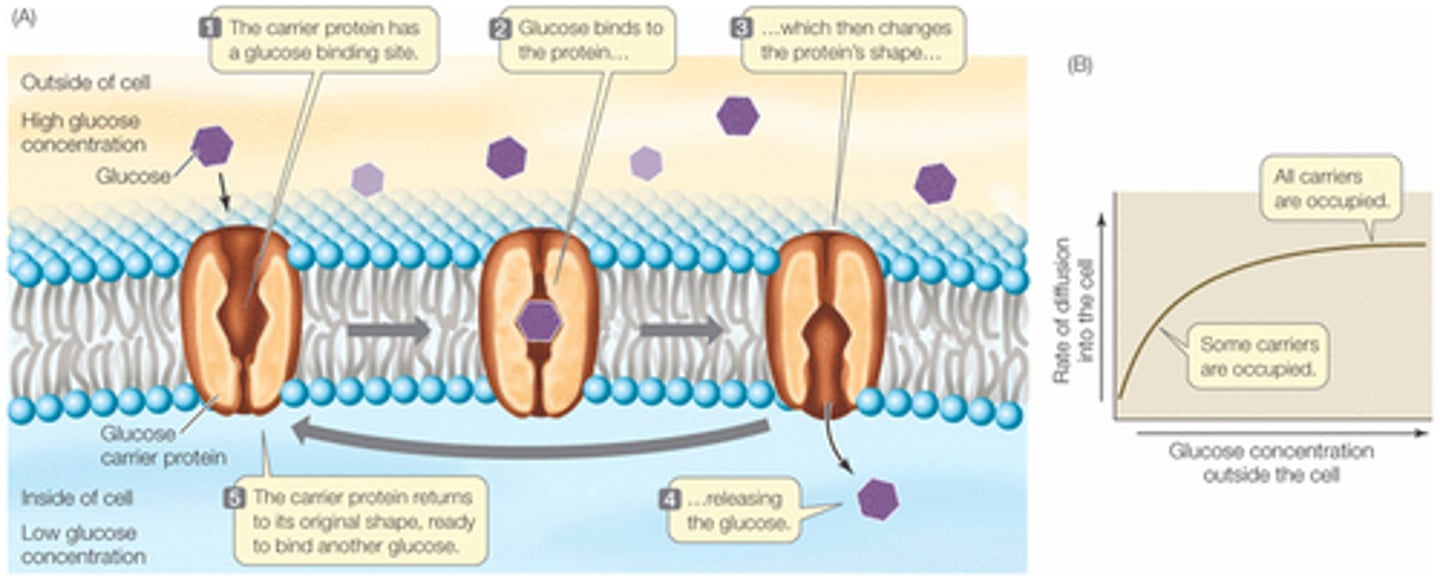
Transport proteins
Transport proteins are membrane proteins that form pumps that use ATP to actively transport solutes across the membrane.
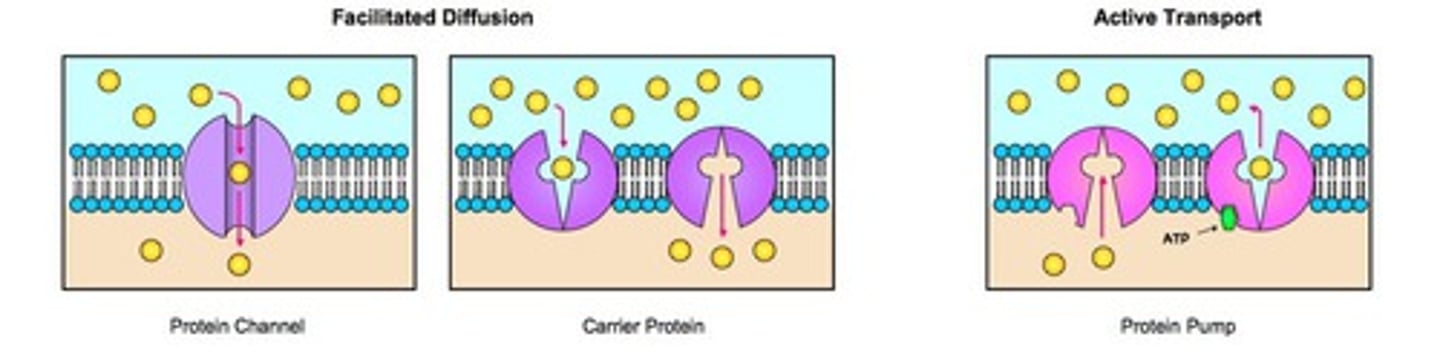
Channel proteins
Channel proteins are membrane proteins that form channels that selectively allow the passage of certain ions or molecules.
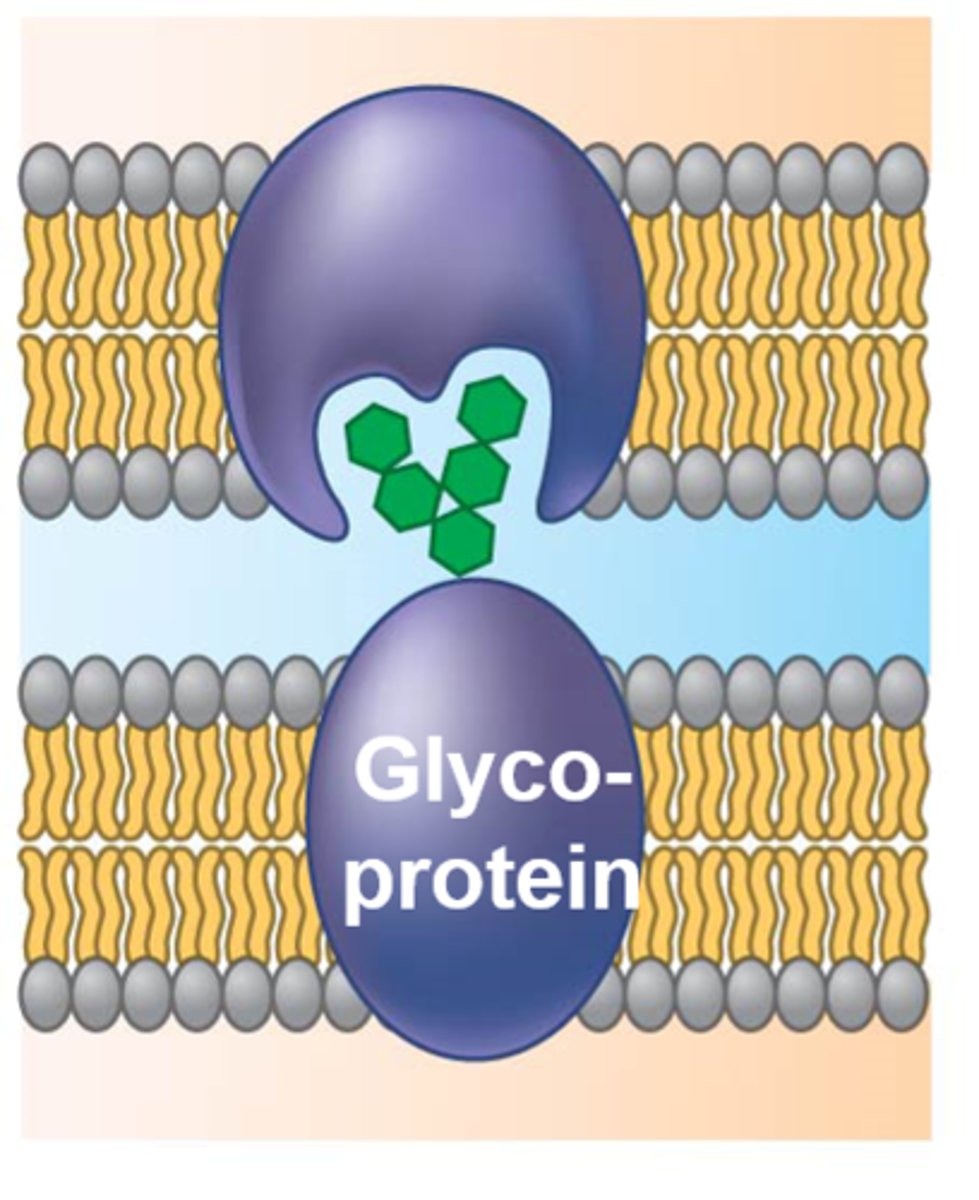
Recognition and adhesion proteins
Recognition and adhesion proteins, such as glycoproteins, are exposed on the extracellular surface and play a role in cell recognition and adhesion
Carbohydrate side chains
A carbohydrate side chain is attached to the surface of some proteins found only on the outer surface of the plasma membrane.
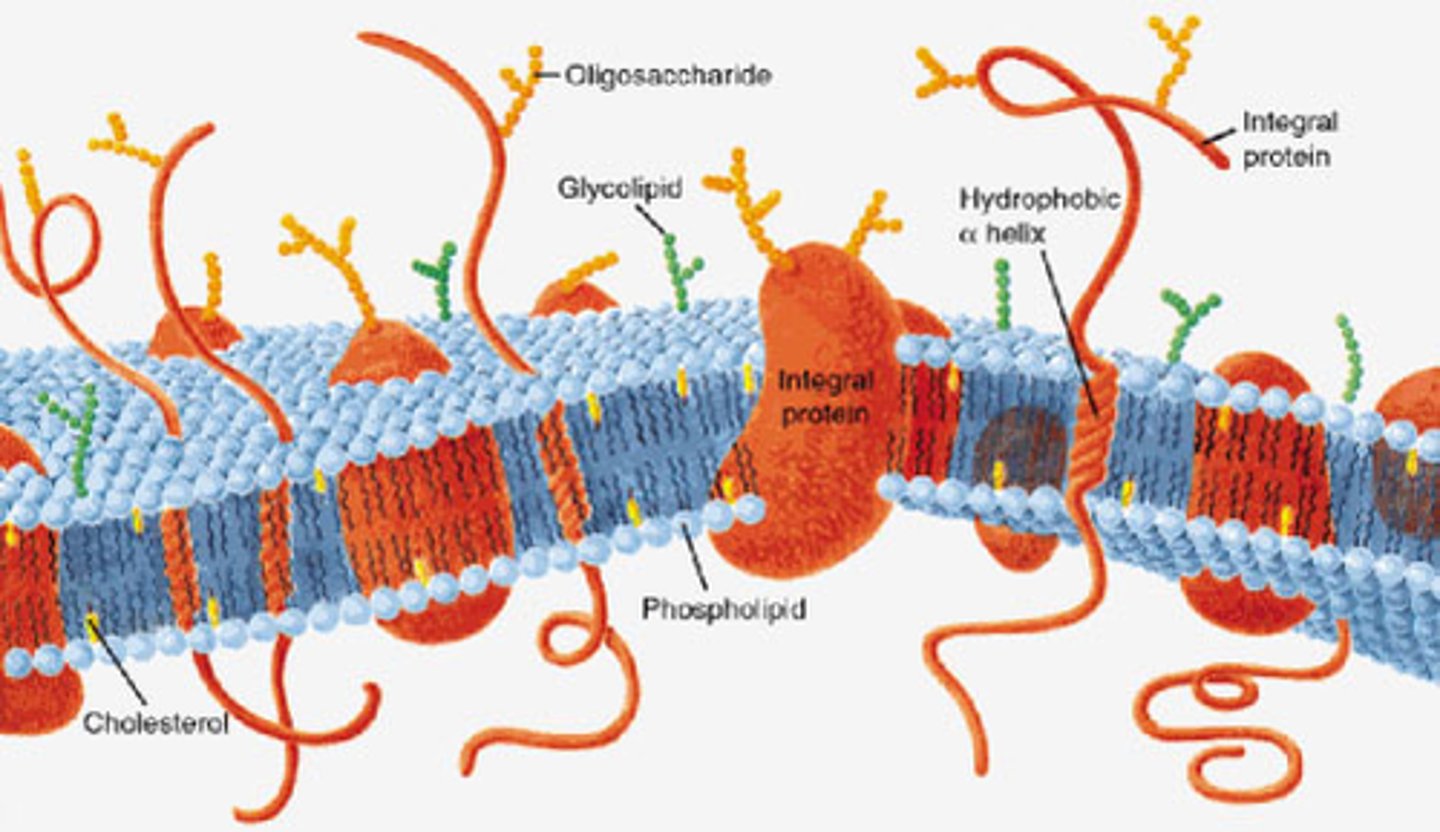
Cholesterol
Cholesterol is a steroid that is found in the phospholipid bilayer because it helps stabilize membrane fluidity in animal cells.
Phospholipid bilayer
The phospholipid bilayer is the double layered plasma membrane that surround cells.
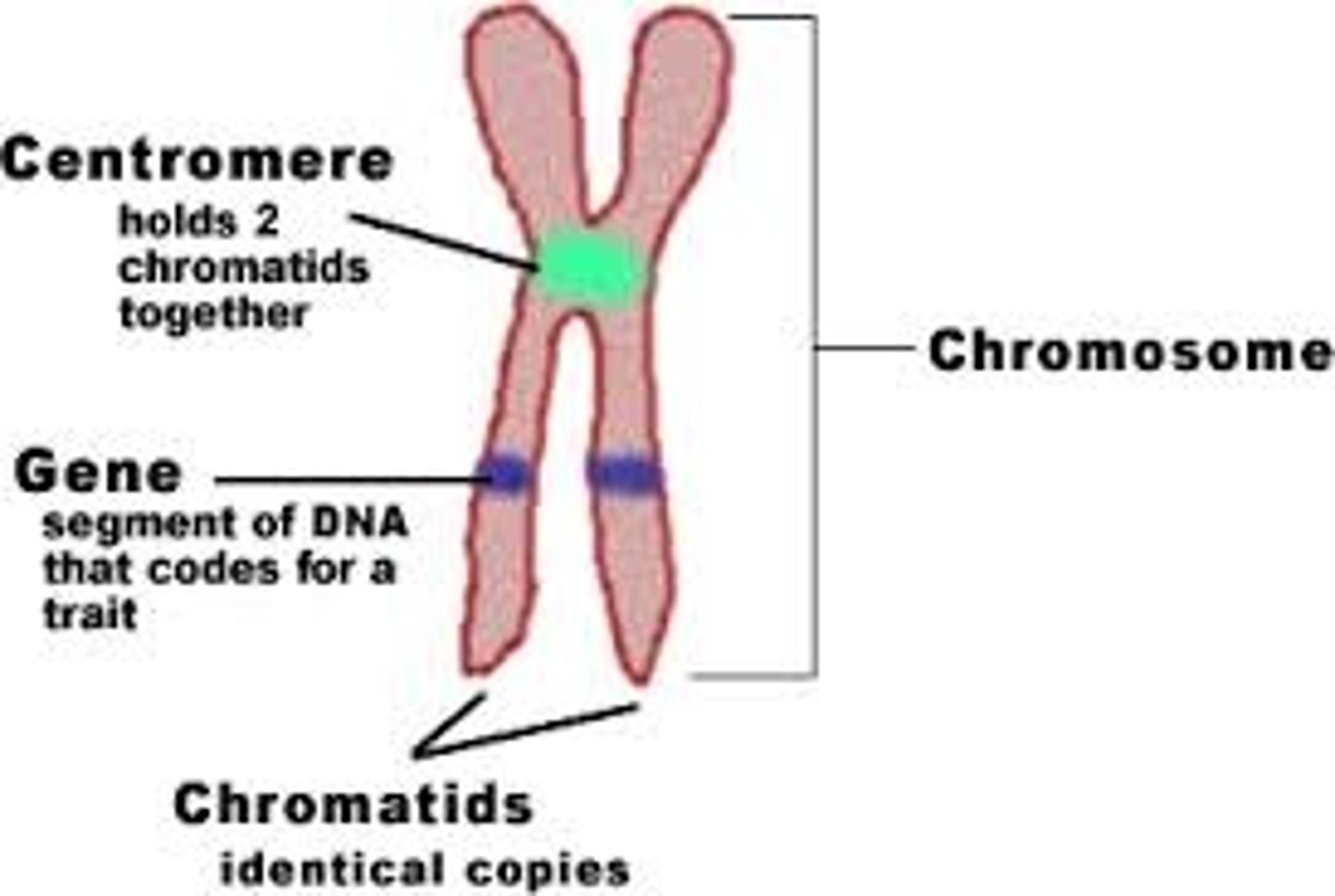
Chromosomes
DNA is organized into large structures called chromosomes in the nucleus.
Nucleolus
The nucleolus is the most visible structure within a cell's nucleus and is where rRNA is made and ribosomes are assembled.
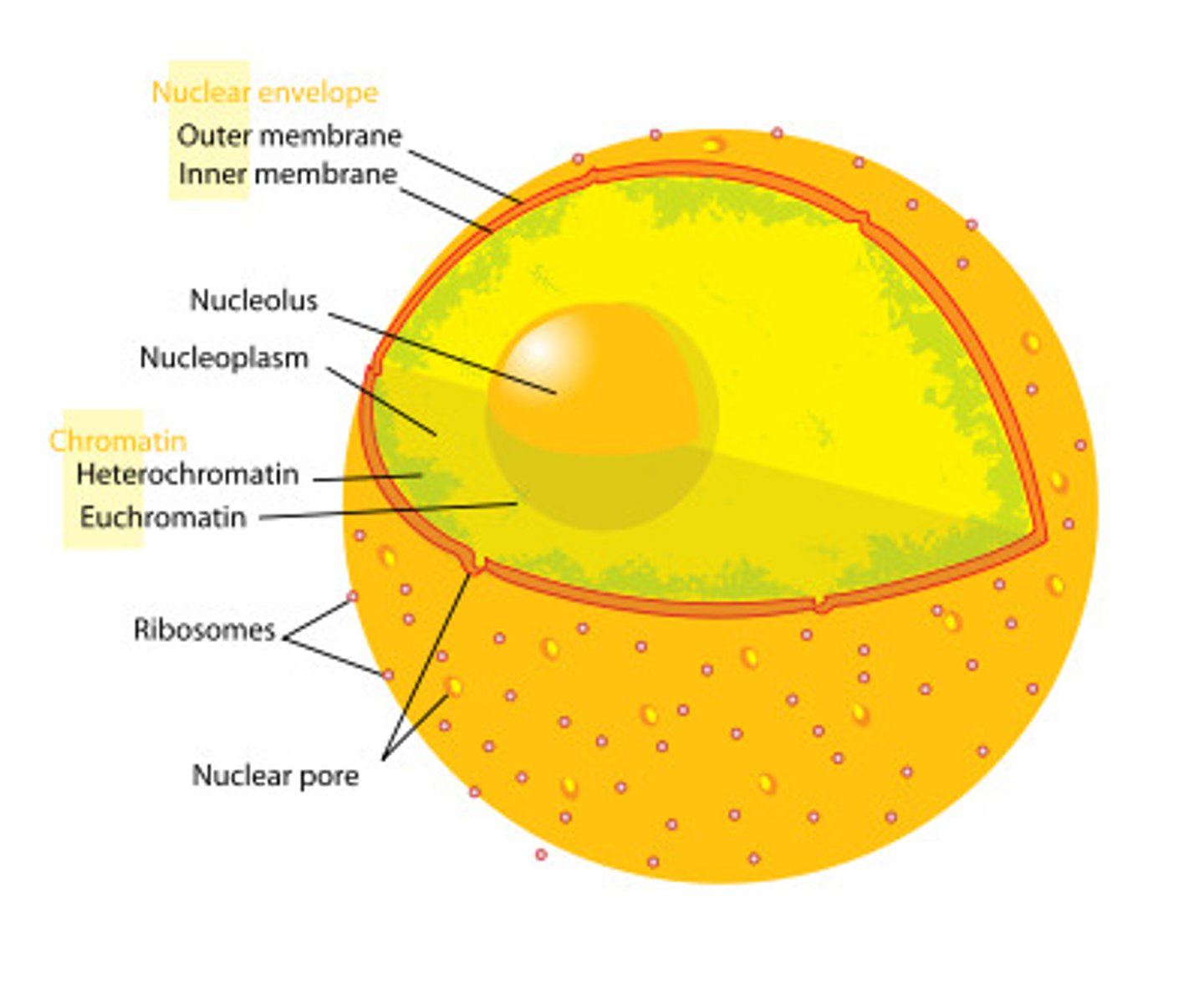
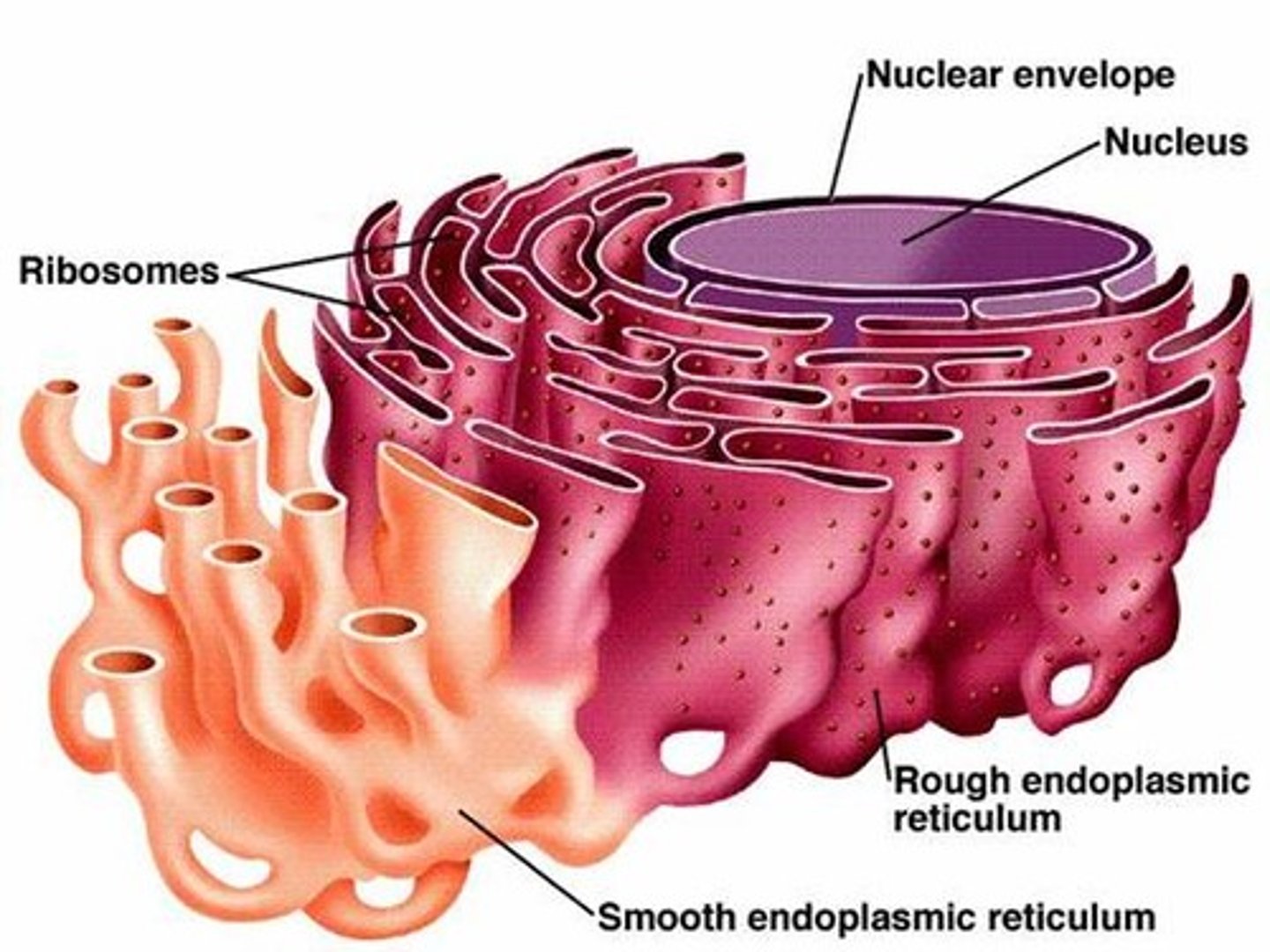
Ribosomes
Ribosomes are organelles that are the sites of protein synthesis - they manufacture all the proteins required by the cell or secreted by the cell. Ribosomes are round structures composed of RNA and proteins and can be either free floating or attached to the endoplasmic reticulum.
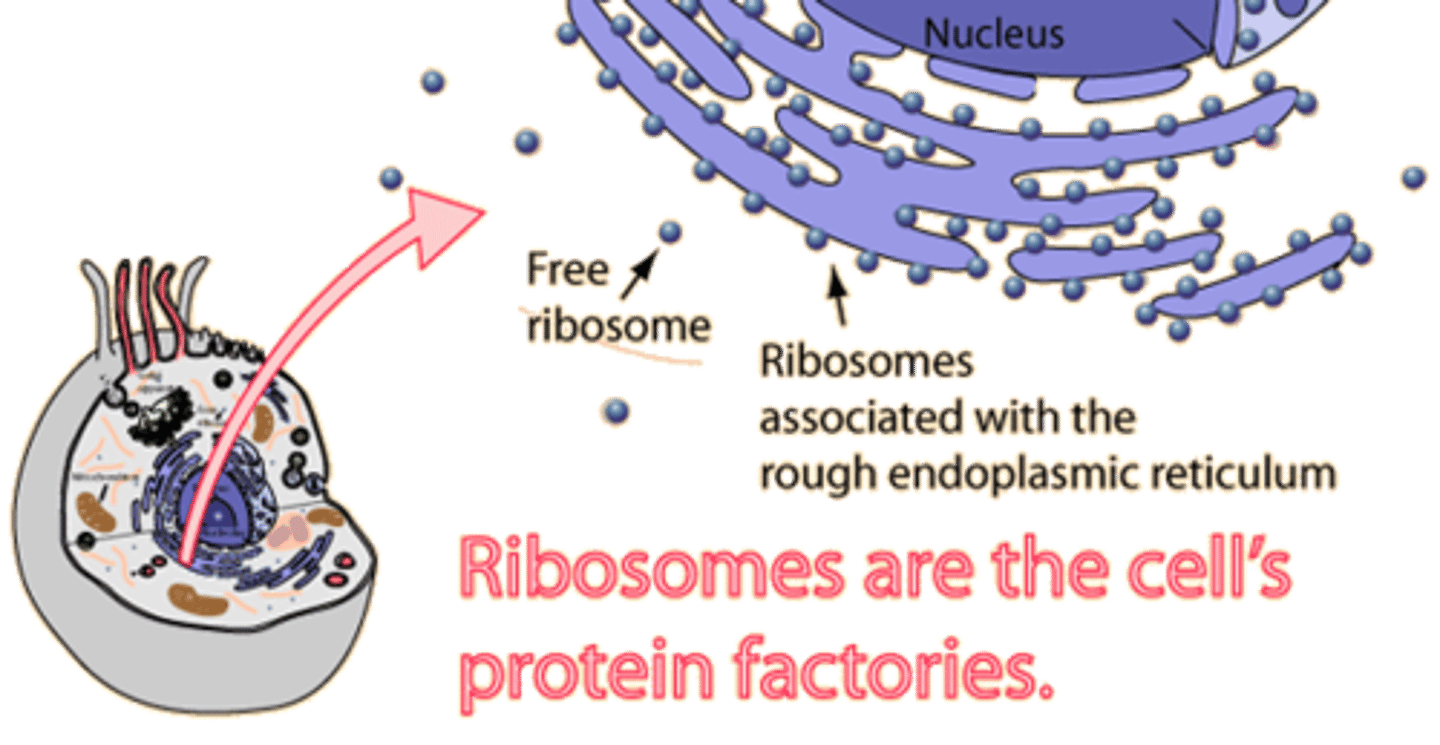
Endoplasmic reticulum (ER)
The endoplasmic reticulum is a continuos channel that extends into many regions of the cytoplasm. The rough ER contains many ribosomes on its surface and generates proteins, which are then trafficked to or across the plasma membrane. The smooth ER lacks ribosomes and makes lipids, hormones, and steroids, and breaks down toxic chemicals.