OCR A 5.1.1 How Fast
1/21
Earn XP
Description and Tags
(incomplete) without PAG
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
22 Terms
how to find rate from change and time graph
-gradient of line of best fit
when curved:
-draw tangent to point where asked for
rate equation linked with conc of substances

rate unit
moldm^-3
how to find order of reaction
add up powers
zero order
change in conc no effect on rate
1st order

2nd order

rate constant (k)
-allows to equate rate and conc
-fixed at particular temp
-if temp change so does k
-larger k is faster RoR
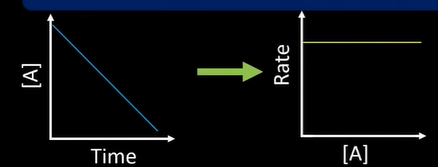
what order
0: as conc increases rate doesnt
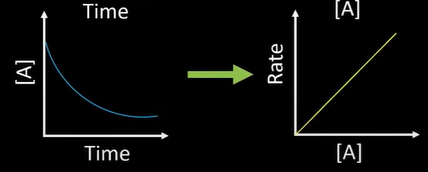
what order
-1st: straight line thru origin
-first graph: Prove that half life exists
-as conc changes rate changes equally
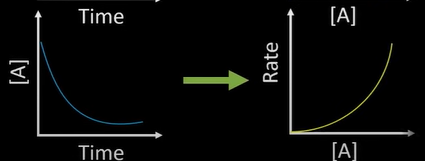
what order
-2nd
-rate changes in unequal amounts
-changing conc changes rate sqrd
half life
time taken for half of reactants to be used up
how to calc k from conc time graph
-find half life
k= ln2/halflife
units= s^-1
rate determining step
-slowest step in multi step reaction
-this determines whole reaction rate
-reactants that effect RoR must be in this step
-catalysts can be in too
which reactants effect rate
reactants in rate equation
arrhenius equation
-links Ea and temp to rate constant

as Ea decreases what happens to k
k increases
as temp increases what happens to k
k increases
arrhenius eqtaution to find Ea

arrhenius plot axis label
-x: 1/t
-y: lnk
arrhenius plot find Ea
-draw lob
-gradient = -Ea/R
-Ea = 8.31 x gradient
arrhenius plot find Arrhenius consatnt
-sub any coordinate and gradient into equation