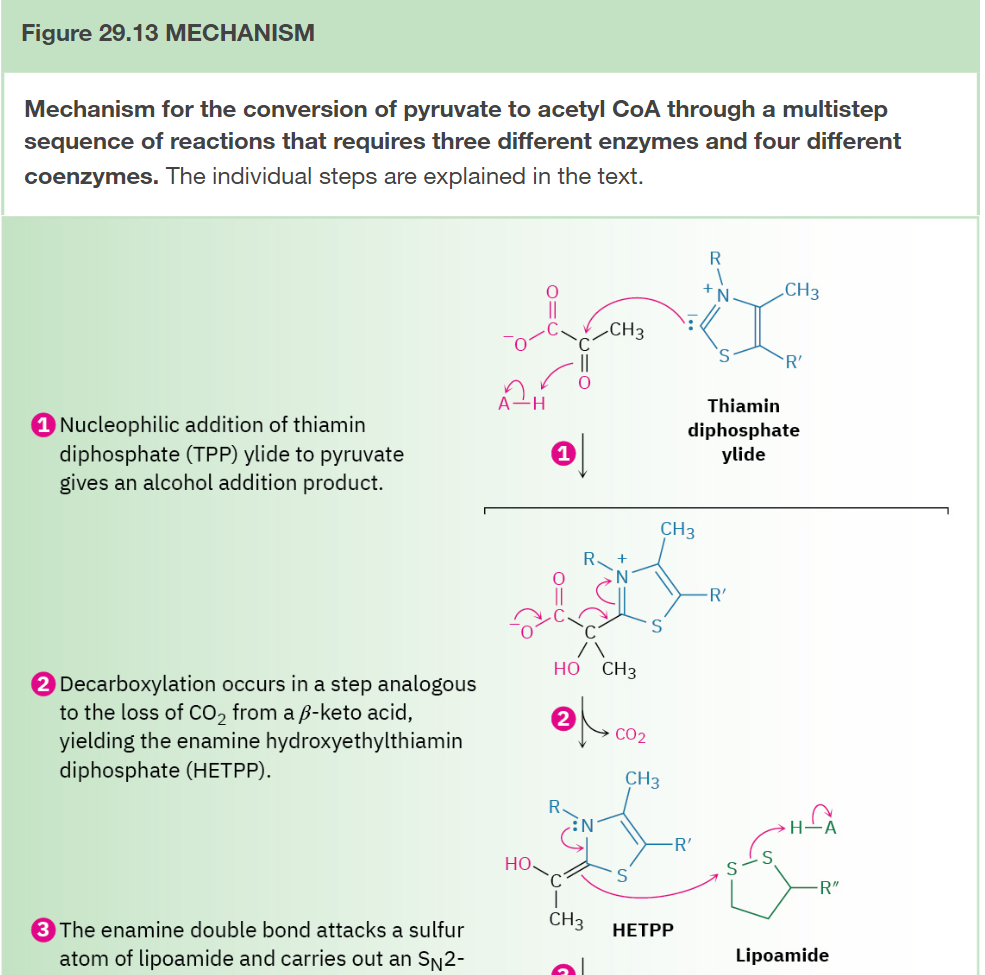
Conversion of Pyruvate to Acetyl CoA
1/5
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
6 Terms
Summary
Step 1: Addition of Thiamin Diphosphate
The conversion of pyruvate to acetyl CoA begins by reaction of pyruvate with thiamin diphosphate, a derivative of vitamin B1. Formerly called thiamin pyrophosphate, thiamin diphosphate is usually abbreviated as TPP. The spelling thiamine is also correct and frequently used.
The key structural element in thiamin diphosphate is the thiazolium ring—a five-membered, unsaturated heterocycle containing a sulfur atom and a positively charged nitrogen atom. The thiazolium ring is weakly acidic, with a pKa of approximately 18 for the ring hydrogen between N and S. Bases can therefore deprotonate thiamin diphosphate, leading to formation of an ylide much like the phosphonium ylides used in Wittig reactions (Section 19.11). As in the Wittig reaction, the TPP ylide is a nucleophile and adds to the ketone carbonyl group of pyruvate to yield an alcohol addition product.
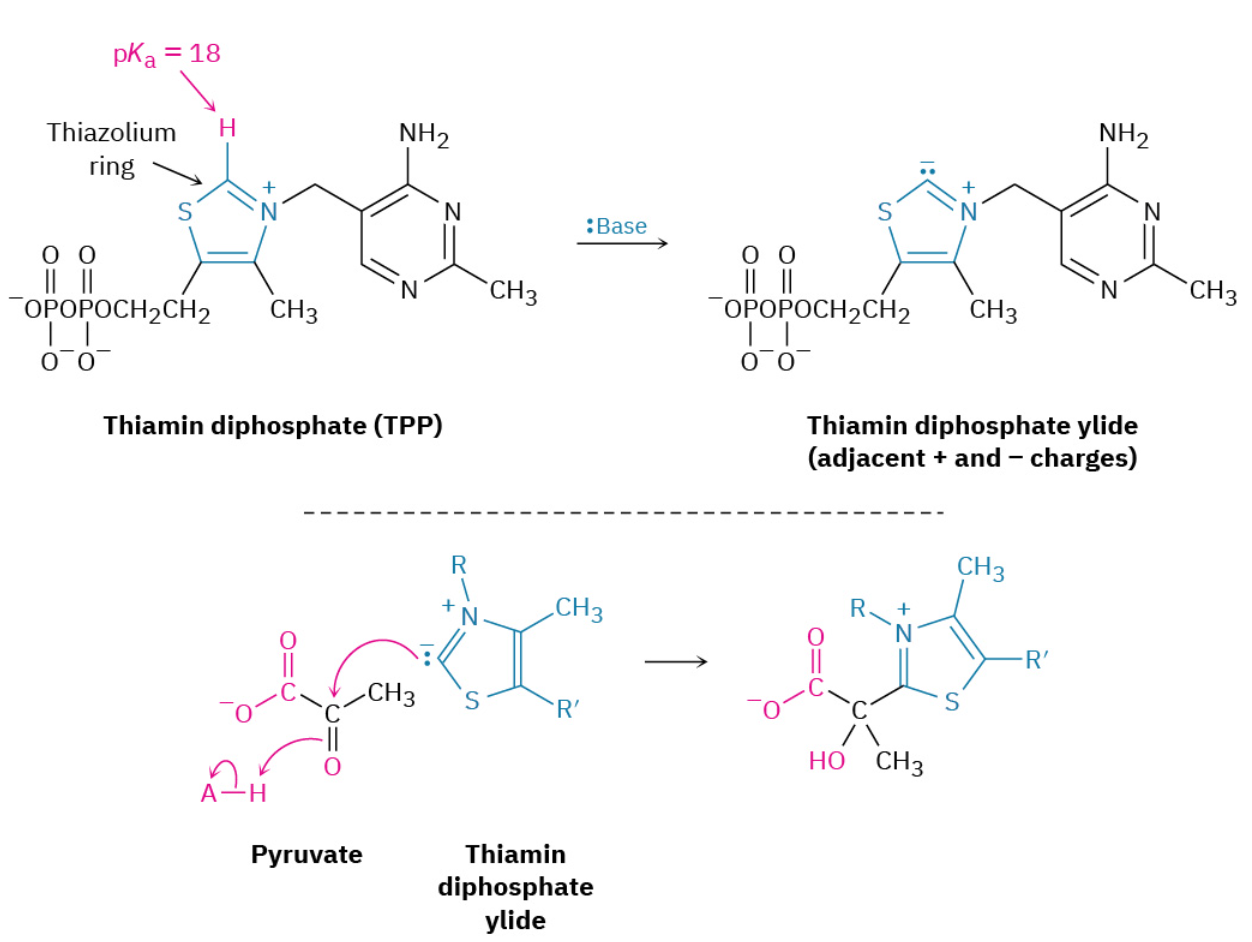
Step 2: Decarboxylation
The TPP addition product, which contains an iminium ion β to a carboxylate anion, undergoes decarboxylation in much the same way that a β-keto acid decarboxylates in the acetoacetic ester synthesis (Section 22.7). The C═N+C═N+ bond of the pyruvate addition product acts like the C═OC═O bond of a β-keto acid to accept electrons as CO2 leaves, giving hydroxyethylthiamin diphosphate (HETPP).
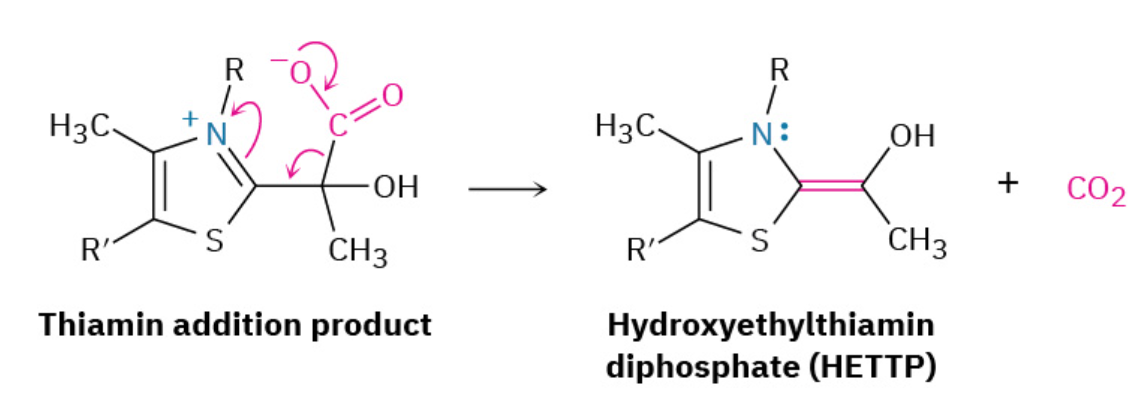
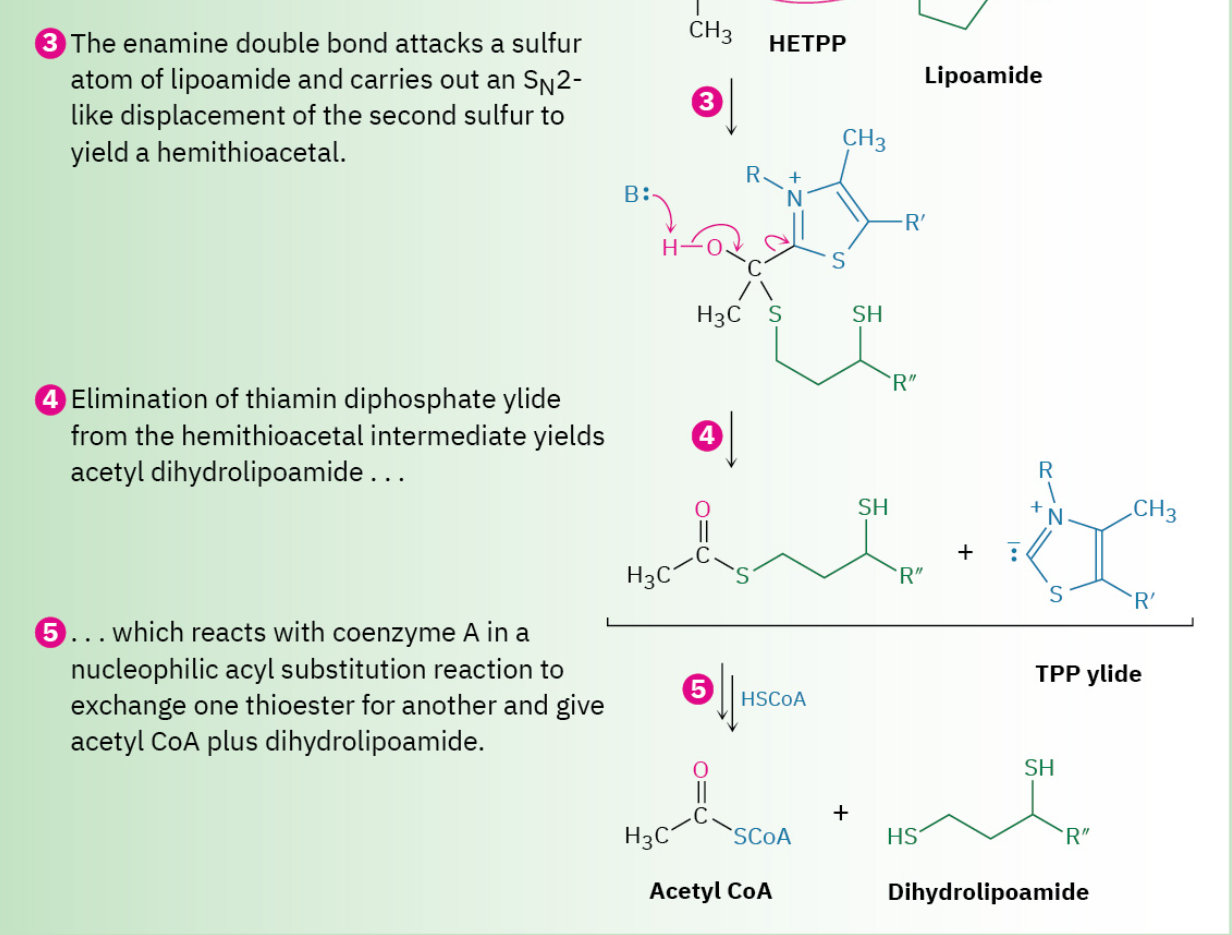
Step 3: Reaction with Lipoamide
Hydroxyethylthiamin diphosphate is an enamine (R2N–C═CR2N─C═C), which, like all enamines, is nucleophilic (Section 23.11). It therefore reacts with the enzyme-bound disulfide lipoamide by nucleophilic attack on a sulfur atom, displacing the second sulfur in an SN2-like process.
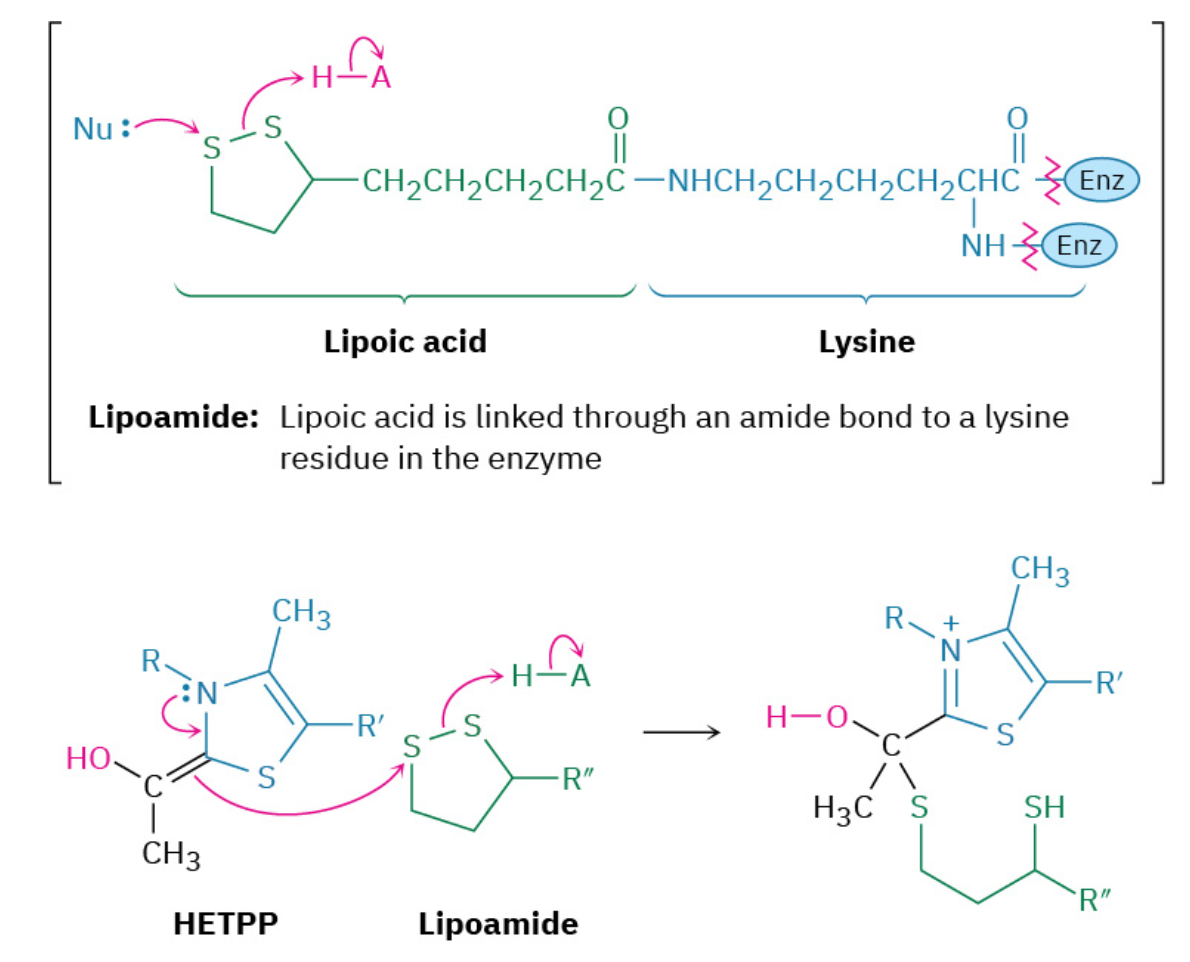
Step 4: Elimination of Thiamin Diphosphate
The product of the HETPP reaction with lipoamide is a hemithioacetal, which eliminates thiamin diphosphate ylide. This elimination is the reverse of the ketone addition in step 1 and generates acetyl dihydrolipoamide.
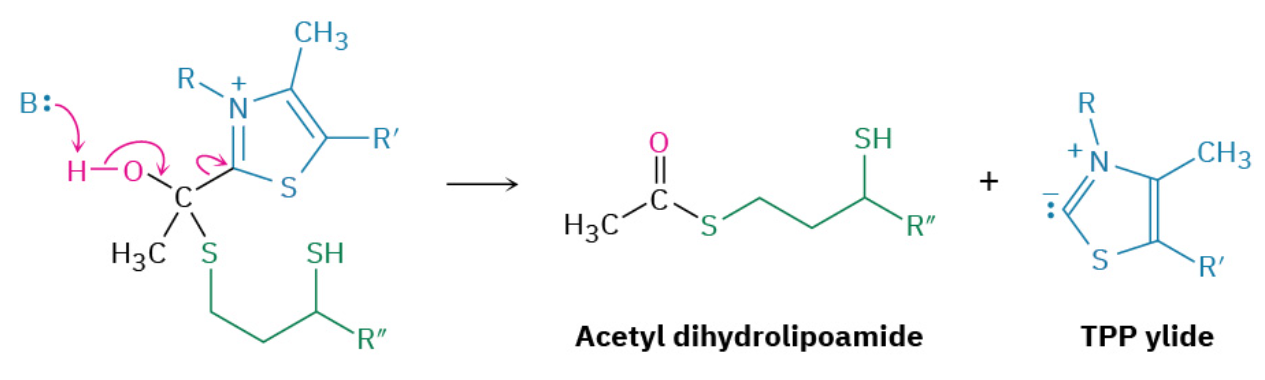
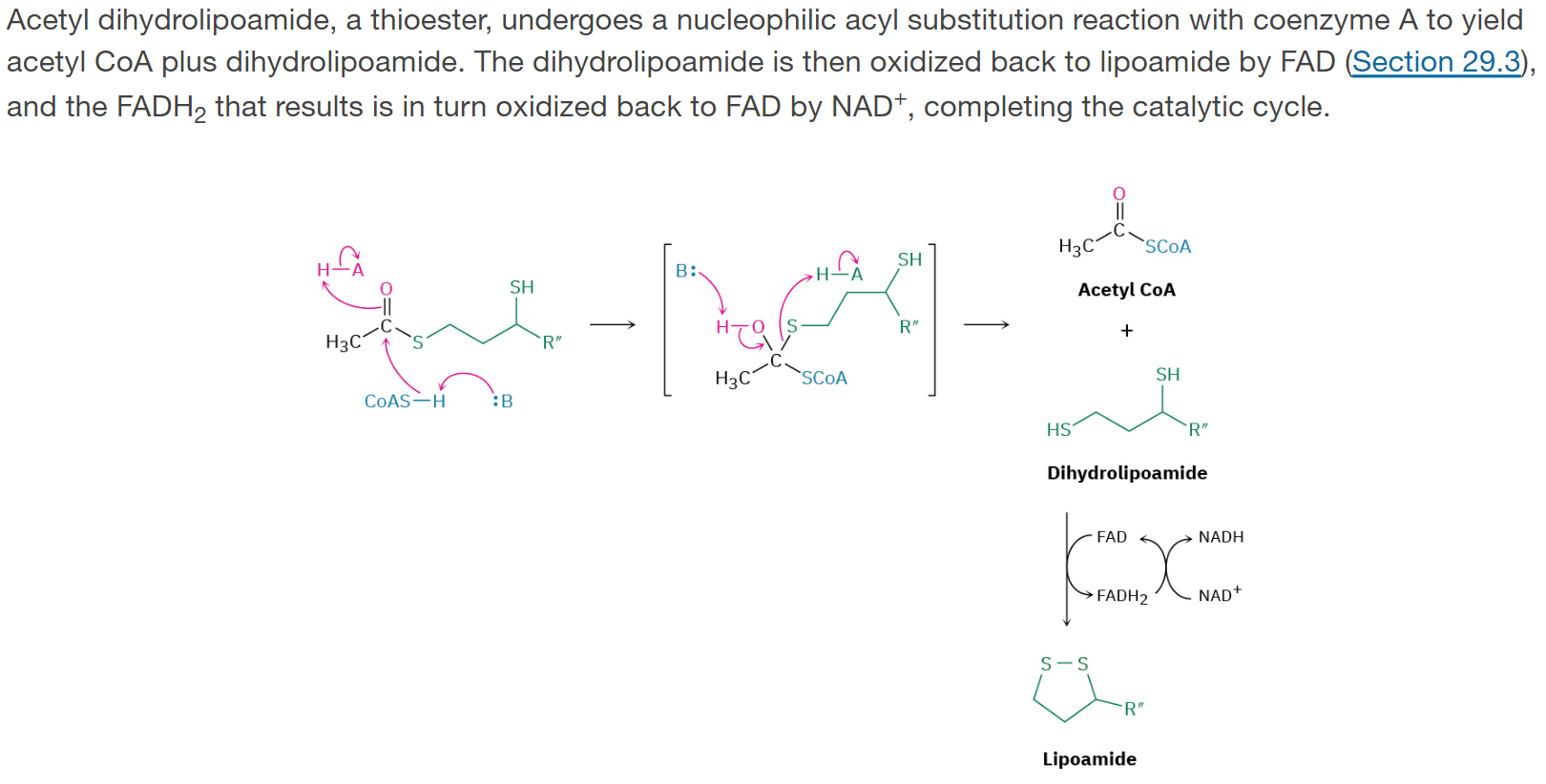
Step 5: Acyl Transfer
Acetyl dihydrolipoamide, a thioester, undergoes a nucleophilic acyl substitution reaction with coenzyme A to yield acetyl CoA plus dihydrolipoamide. The dihydrolipoamide is then oxidized back to lipoamide by FAD, and the FADH2 that results is in turn oxidized back to FAD by NAD+, completing the catalytic cycle.