Gypsum in Dentistry: Types, Reactions, and Applications
1/77
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
78 Terms
GYPSUM
CaSO4•2H2O; calcium sulfate dihydrate
Calcination
The process of heating a solid material to drive off volatile chemically combined components such as water and carbon dioxide.
Calcining
Grounding calcium sulfate dihydrate to a powder and heated in an oven to dry.
Cast
A reproduction of the shape and surface of a structure made in an impression of the surface.
Dental Plaster (Plaster of Paris)
The beta form of calcium sulfate hemihydrate (CaSO4• ½ H2O).
Dental stone
The alpha form of calcium sulfate hemihydrate (CaSO4• ½ H2O).
Die
A reproduction of a prepared tooth made from a gypsum product, epoxy resin, a metal, or a refractory material.
Gypsum-based investment
A refractory material consisting of silica and gypsum as a binder used to produce a mold for the metal casting process.
Hygroscopic setting expansion
The expansion that occurs when gypsum or a gypsum-bonded investment sets while immersed in water (usually heated to approximately 38 °C [100 °F]).
![<p>The expansion that occurs when gypsum or a gypsum-bonded investment sets while immersed in water (usually heated to approximately 38 °C [100 °F]).</p>](https://knowt-user-attachments.s3.amazonaws.com/18779cda-a522-42e4-8b82-005bd3dfc1ca.jpg)
Model
A positive likeness of an object.
Normal setting expansion
The expansion that occurs when gypsum or a gypsum-bonded investment sets in ambient air.
Exothermic reaction
Where heat evolves as the gypsum sets.
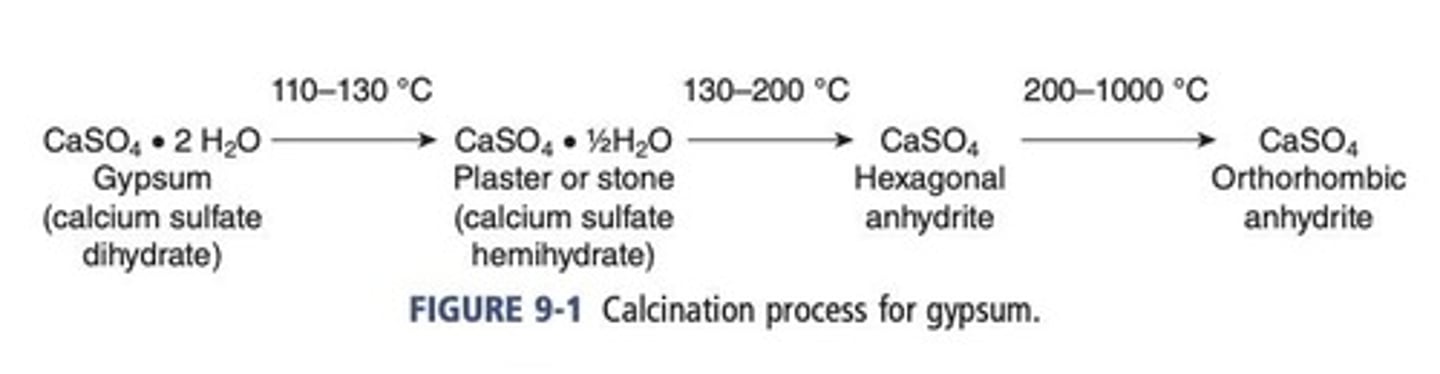
Production of gypsum products
Commercially, the gypsum is ground and subjected to temperatures of 110 °C to 130 °C (230 °F to 266 °F) in open containers to drive off part of the water of crystallization.
Calcination of gypsum
α-hemihydrate, β-hemihydrate.
Mineral mined on earth
Gypsum is also produced as a by-product of flue gas desulfurization.
Appearance of gypsum
It usually appears white to milky yellowish and is found as compact mass in nature.
Supplied form of gypsum
Supplied as fine hemihydrate powders that are produced by heating ground gypsum particles.
Use of gypsum in ancient Egypt
A mixture of plaster (a gypsum product), lime, and water was used in joining the stone blocks of ancient Egypt's pyramids.
Uses in dentistry
Occasionally used intraorally for completely edentulous patients, preparation of study models, mounting of study models in articulators, study cast in form of die, embed used on flask then for dental prostheses, and as an additive to silica in dental casting investments.
Grounding of calcium sulfate dihydrate
Grounding it to powder and heated in an oven to dry.
Method of calcination
Based upon the method of calcination, different forms of hemihydrate can be obtained.
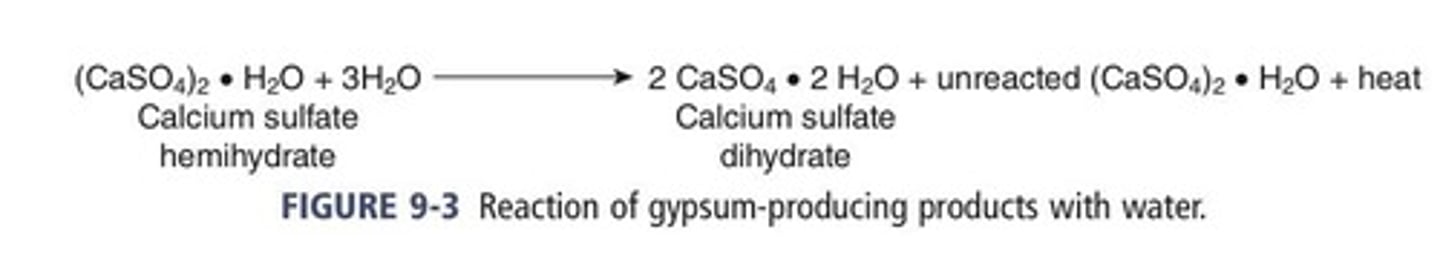
Calcium sulfate hemihydrate
is the principal constituent of gypsum-based products (CaSO4• ½ H2O)
Colloidal Theory
When mixed with water, hemihydrate enters into the colloidal state through a sol-gel mechanism
Hydration Theory
Rehydrated plaster particles unite through hydrogen bonding with sulfate groups to form the set material
Dissolution-Precipitation Theory
Dissolution of hemihydrate particles in water followed by instant recrystallization to the dihydrate
Wet Calcination
Heating gypsum between 120-130 °C under pressure of 17 lbs/in² for 5-7 hours in a kettle, vat, or rotary kiln
Stronger and harder dihydrate structure
Prismatic and more regular in shape
Mixing Time
Mechanical mixer: 20-30 seconds; Hand spatulation: 1 minute; Time from addition of powder until mixing is completed
Working Time
Time required for the mixture to remain workable after mixing
Dry Calcination
Ground and heated to 110-130 °C in an open kettle/containers
Crystals are spongy
Irregular in shape, more porous in character; Require more water to wet powder particles for stirring and pouring
Anhydrite
As temperature is raised, hemihydrate becomes anhydrite.
Setting Time
Duration until the consistency is no longer acceptable for intended purposes.
Loss of gloss
9 minutes.
Initial Set
Duration: 13 minutes; time elapsed when the mix can resist penetration by a Gillmore needle (2.12mm in diameter, 113.4g).
Final Setting Time
Duration: 30 minutes; elapsed time at which a Gillmore needle (1.06mm tip, 453.6g) leaves only barely perceptible mark on surface.
MODIFIED α- HEMIHYDRATE/ DIE STONE
Also called improved stone; formed by boiling gypsum in 30% aqueous solution of calcium chloride or in the presence of more than 1% of sodium succinate at 100°C.
Crystals
Will be shorter and thicker than those produced in a closed container.
Cube shaped
Reduces surface area that improves flow of mixture.
Primatic
More regular and densely packed.
Wet/ Green Strength
Increases rapidly as the material hardens after the initial set; shortens setting time.
Fineness
The finer the material, the faster the setting time.
Water-Powder Ratio
Weight of the water divided by the weight of hemihydrate powder; ↑ W:P causes prolonged setting time, decrease in strength and setting expansion.
Dry Strength
Strength obtained after excess water is removed by drying.
Mixing
The longer and more rapid the mixing, the shorter the setting time.
Temperature
The higher the temperature, the longer the setting time.
Retarders and Accelerators
Chemical modifiers that increase or decrease the setting time.
Compressive strength
Strength of set gypsum products is generally expressed in terms of this.
Microwave irradiation
Can speed up gypsum cast drying, achieving similar strength in 1 minute as 24 hours of air drying.
Excess water
Presence weakens the set product.
Increased mixing time
Increases the strength; overmixing decreases it.
Incorporation of accelerators and retarders
Decreases both the wet and dry strength.
Chloric Acid/Borax
At 0.2mM or 0.08g/L.
Calcium salts
Includes Potassium Sulfate, NaCl (2%), Sodium Sulfate.
Too much salt
Will cause it not to set.
Type I: Impression Plaster
Used to take impression; rarely used; replaced by hydrocolloids and elastomers; modifiers are added to regulate setting time (4-5 min.) and setting expansion.
Type II: Model Plaster/Plaster of Paris
Used to make study cast/model for record purposes stronger than Type I Plaster; lower water: powder ratio; therefore less porosity, higher expansion; used to make study casts or to mount stone casts in the articulator and to fill a flask in denture construction; natural white.
Type III: Dental Stone (Class 1 Dental Stone)
Stronger than Plaster of Paris/model plaster; used as working cast that can withstand forces; used for production of dentures, for indirect wax patterns of indirect restorations; setting time is 45-60 min. depending on the stone and type of impression material; 82 Rockwell Hardness.
Type IV: Dental Stone, High Strength, or Die Stone (Class II Dental Stone)
Stronger than Type III; used as working die for crown restoration; for accurate measurements; die material refers to single tooth/several teeth; inlay and onlay restoration; harder with minimum setting expansion; 92 Rockwell Hardness.
Type V: Dental Stone, High Strength, High Expansion
Used for the construction of removable partial dentures (RPD) and fixed partial dentures (FPD); higher compressive strength than Type IV with increased setting expansion to compensate for the greater shrinkage of newer, high-melting alloy used for dental castings.
Water: Powder ratio for Type I
0.060 ml.
Water: Powder ratio for Type II
0.050 ml.
Water: Powder ratio for Type III
0.030 ml.
Water: Powder ratio for Type IV
0.24 ml or less.
Water: Powder ratio for Type V
0.18 to 0.22 ml.
Care cast
Store in water bath saturated with calcium sulfate solution.
Care for gypsum
Sealed container in dry atmosphere.
Disinfection of casts
Spray the casts with disinfecting solutions such as iodophor, glutaraldehyde, or phenol and store in sealed plastic bag for 10 minutes; there are stone materials that already contain disinfecting agents.
Mixing of gypsum products
Involves proper proportioning of water and powder to achieve desired consistency.
Manipulation of gypsum products
Includes mixing, proportioning, and ensuring proper setting conditions.
Setting time for Type III
45-60 min. depending on the stone and type of impression material.
Rockwell Hardness for Type III
82
Rockwell Hardness for Type IV
92
Rockwell Hardness for Type V
Higher than Type IV.
Boxing in model plaster
Strips of wax are wrapped around the impressions extending ½ inch beyond the tissue side of the impression.
Mixture preparation for model plaster
Mixture piled on glass slab; the filled impression is inverted over the pile, shaped with spatula before it sets.
Longer spatulation effect
Reduces working time.
Effect of repeated addition of water or powder
Will result in uneven set, low strength, and distortion which will produce an inaccurate gypsum product.