AP Biology Units 1-4
1/203
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
204 Terms
organic
Contains carbon

Types of Organic Compounds
carbohydrates, lipids, proteins, nucleic acids

carbohydrates
the starches and sugars present in foods, broken down to provide energy

polysaccharide
Carbohydrates that are made up of more than two monosaccharides
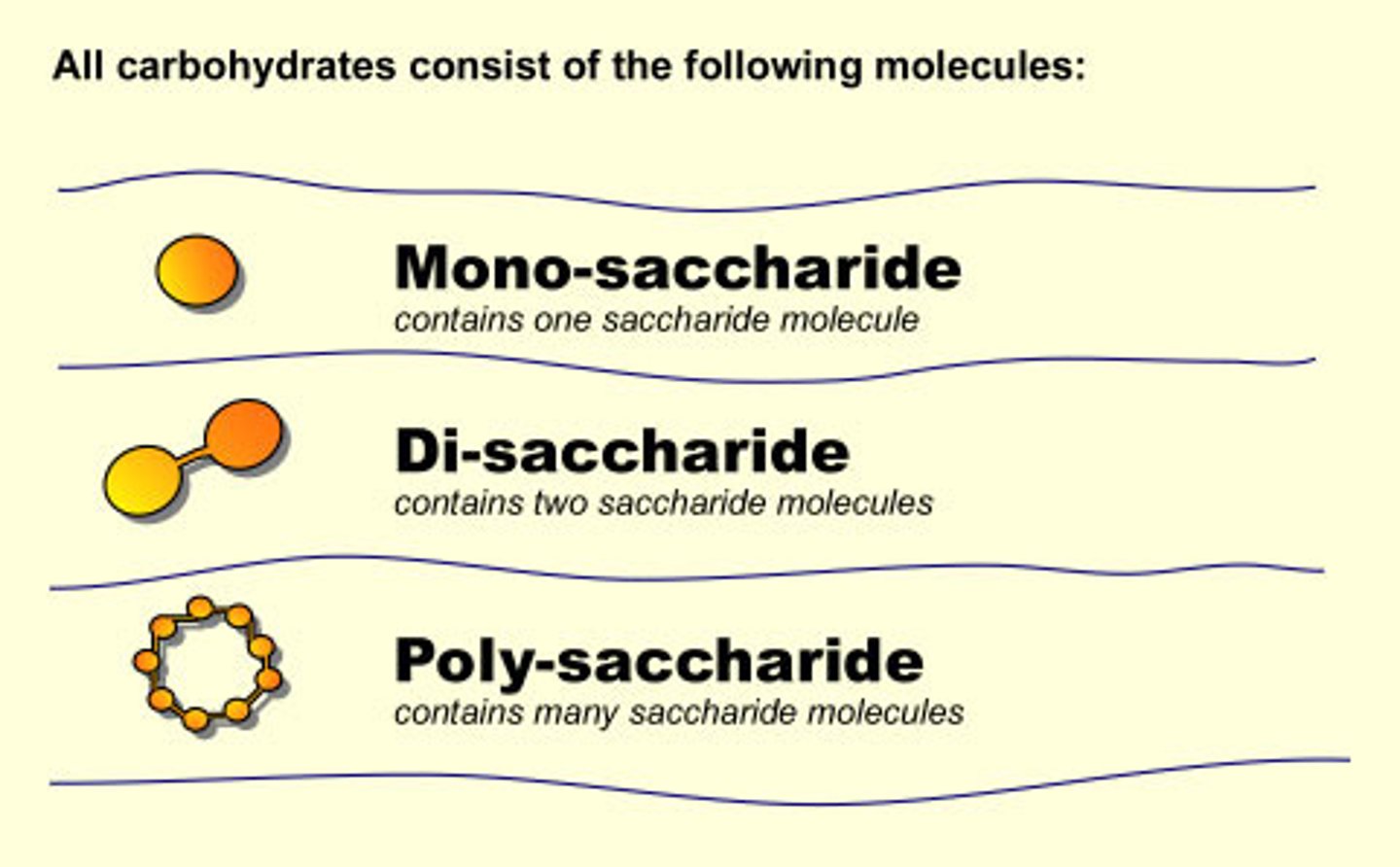
monosaccharide
A single sugar molecule such as glucose or fructose, the simplest type of sugar.

disaccharide
A double sugar, consisting of two monosaccharides joined by dehydration synthesis.
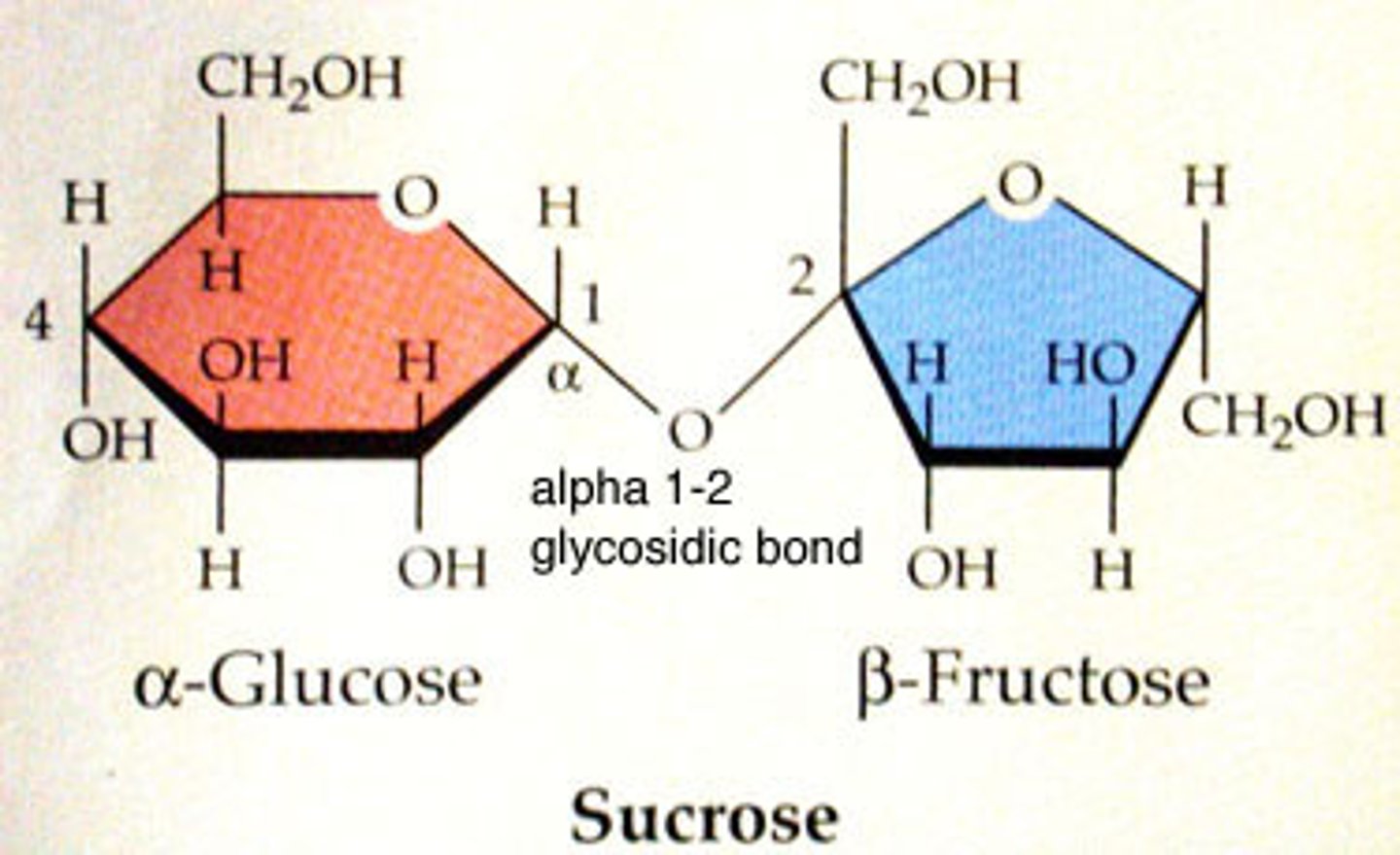
dehydration synthesis
A chemical reaction in which two molecules covalently bond to each other with the removal of a water molecule.

Polysaccharide examples
starch, glycogen, cellulose

Most Carbohydrates end in
-ose
Fats/ Lipids
Long molecules made up primarily of carbon and hydrogen; used to store energy in the body

Structure of fats/lipids
glycerol with 1, 2, or 3 fatty acid chains attached
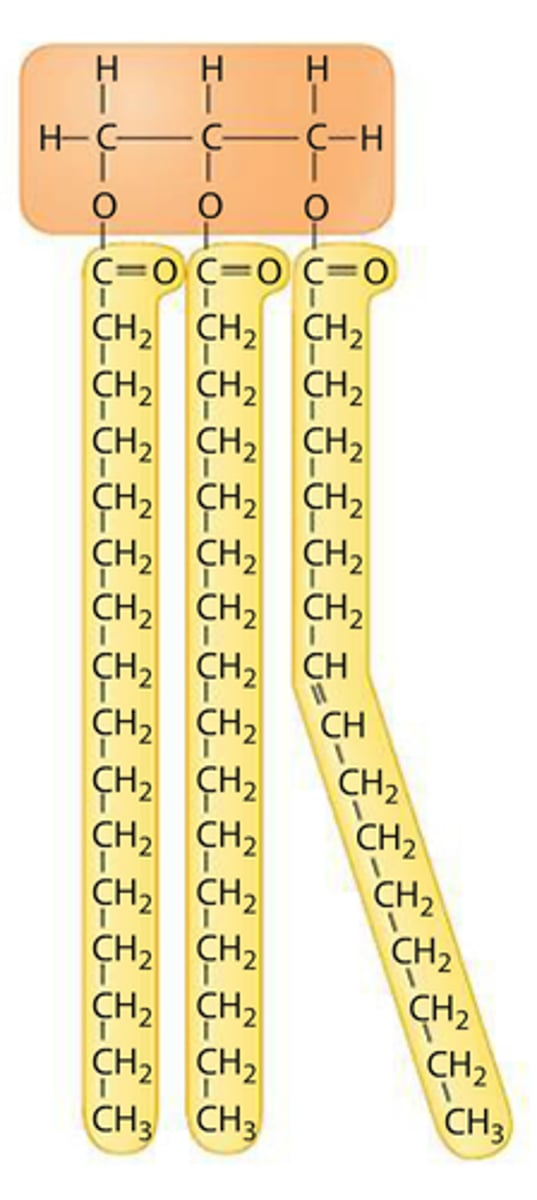
Fatty Acid
hydrocarbon chain often bonded to glycerol in a lipid

Triglycerides
an energy-rich compound made up of a single molecule of glycerol and three molecules of fatty acid.

Phospholipid
a lipid that contains phosphorus and that is a structural component in cell membranes
Saturated Lipid
each carbon atom in a lipid's fatty acid chain is joined to another carbon atom by a single bond, solid at room temperature

Unsaturated Lipid
at least one carbon-carbon double bond in a fatty acid, liquid at room temperature

saturated vs unsaturated fats
Both are lipids - saturated have NO double bonds, are saturated with hydrogen, are animal based, solid at room temperature
unsaturated - have double bonds, are plant based, liquid at room temperature

Proteins
Large biomolecules and macromolecules that comprise one or more long chains of amino acid residues.
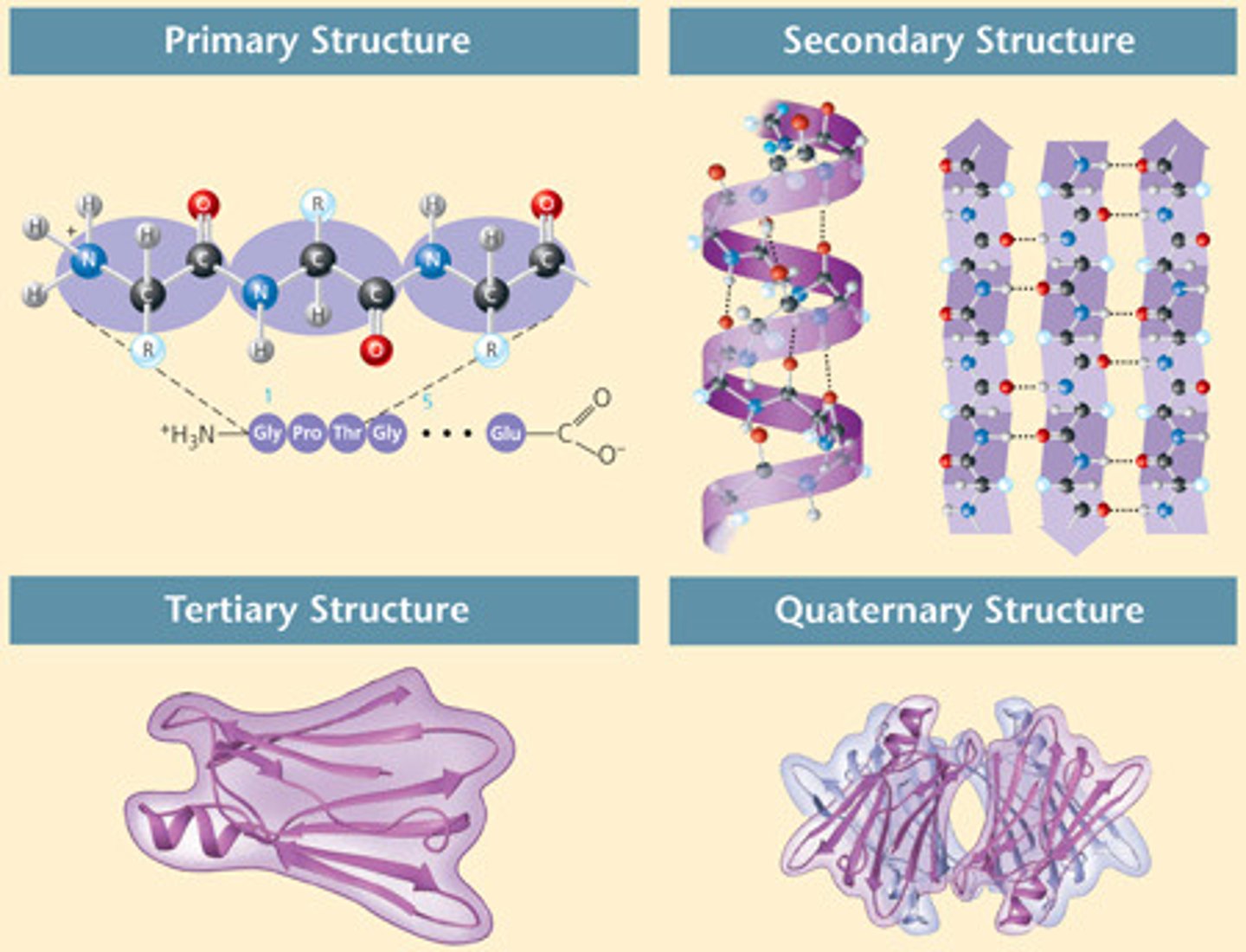
Primary structure of Proteins
linear sequence of amino acids

secondary structure of proteins
Regularly repeating local structures such as α-Helices and ß-pleated sheets often formed by hydrogen bonds between residues nearby on the chain.

tertiary structure of proteins
The overall three-dimensional arrangement of its polypeptide chain in space. It is generally stabilized by outside polar hydrophilic hydrogen and ionic bond interactions, and internal hydrophobic interactions between nonpolar amino acid side chains

quaternary structure of proteins
Refers to proteins that consist of more than 1 polypeptide chain. EX: hemoglobin

Enzyme
protein that acts as a biological catalyst (speeds up chemical reactions); lowers the activation energy

Functions of Proteins
structural support, protection, transport, regulation, defense, movement
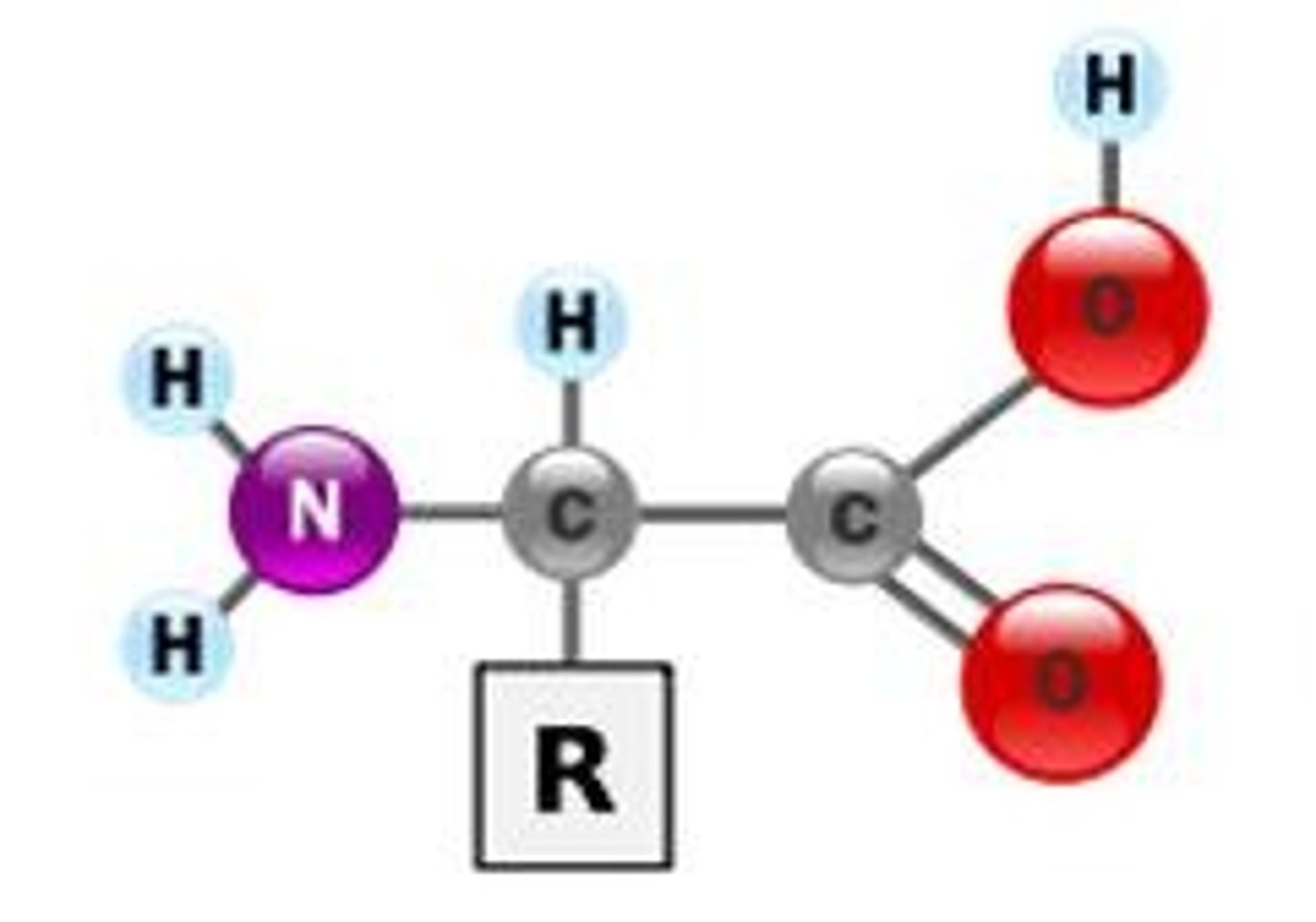
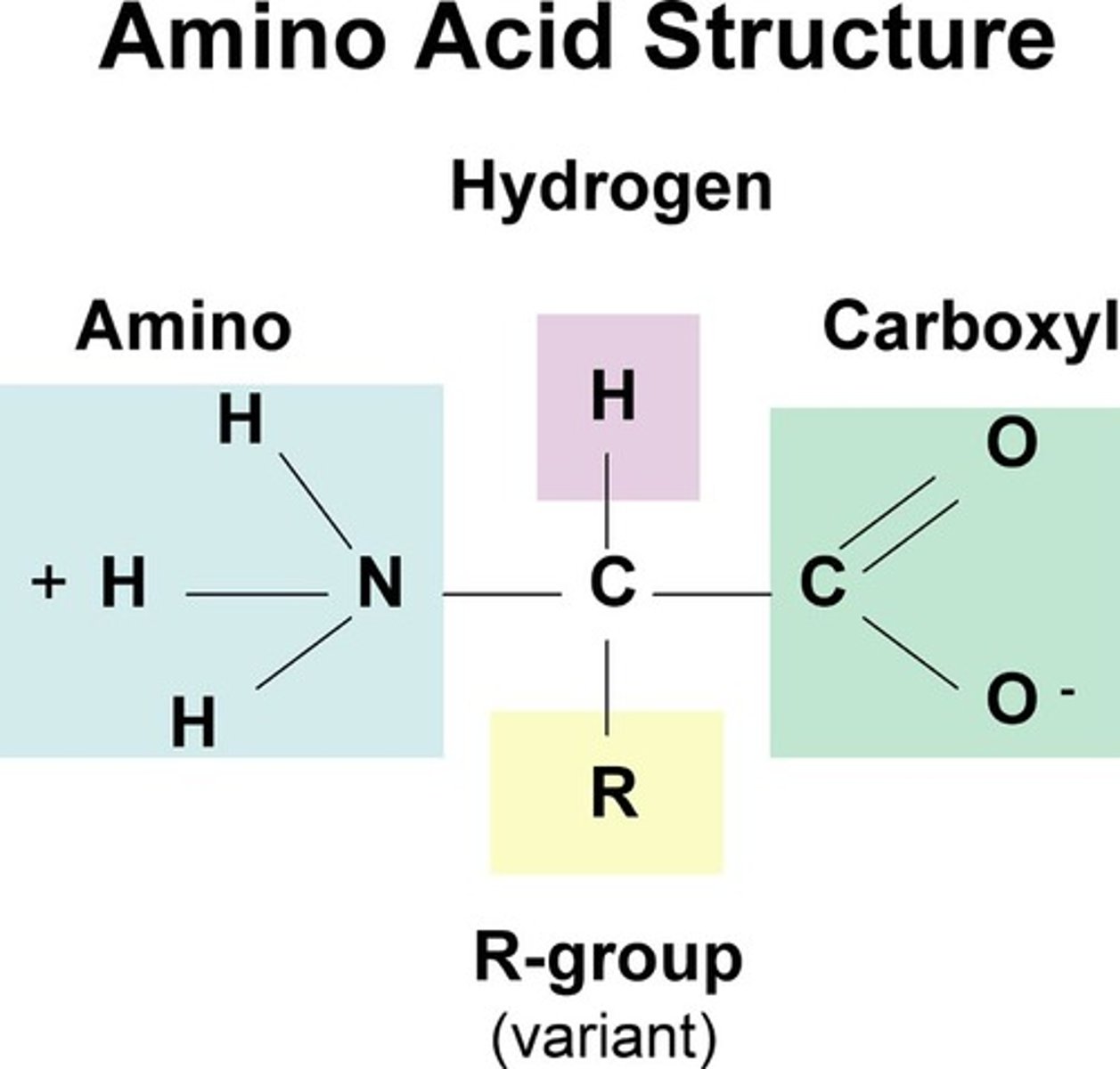
amino acids
building blocks of proteins, 20 types
polypeptide chain
long chain of amino acids linked by peptide bonds
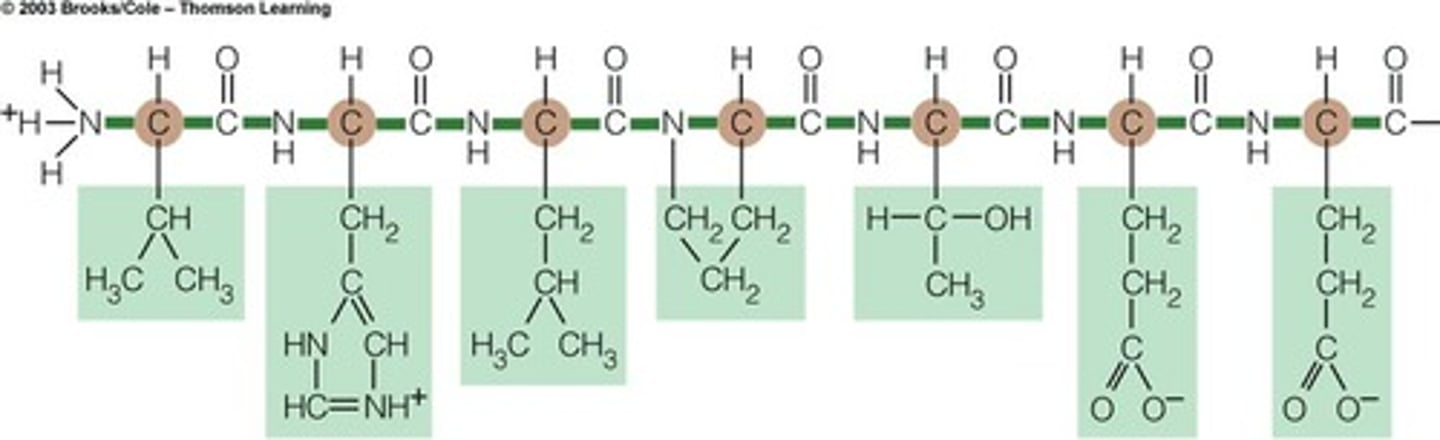
peptide bond
The chemical bond that forms between the carboxyl group of one amino acid and the amino group of another amino acid

carboxyl group
A functional group present in organic acids and consisting of a single carbon atom double-bonded to an oxygen atom and also bonded to a hydroxyl group, COOH
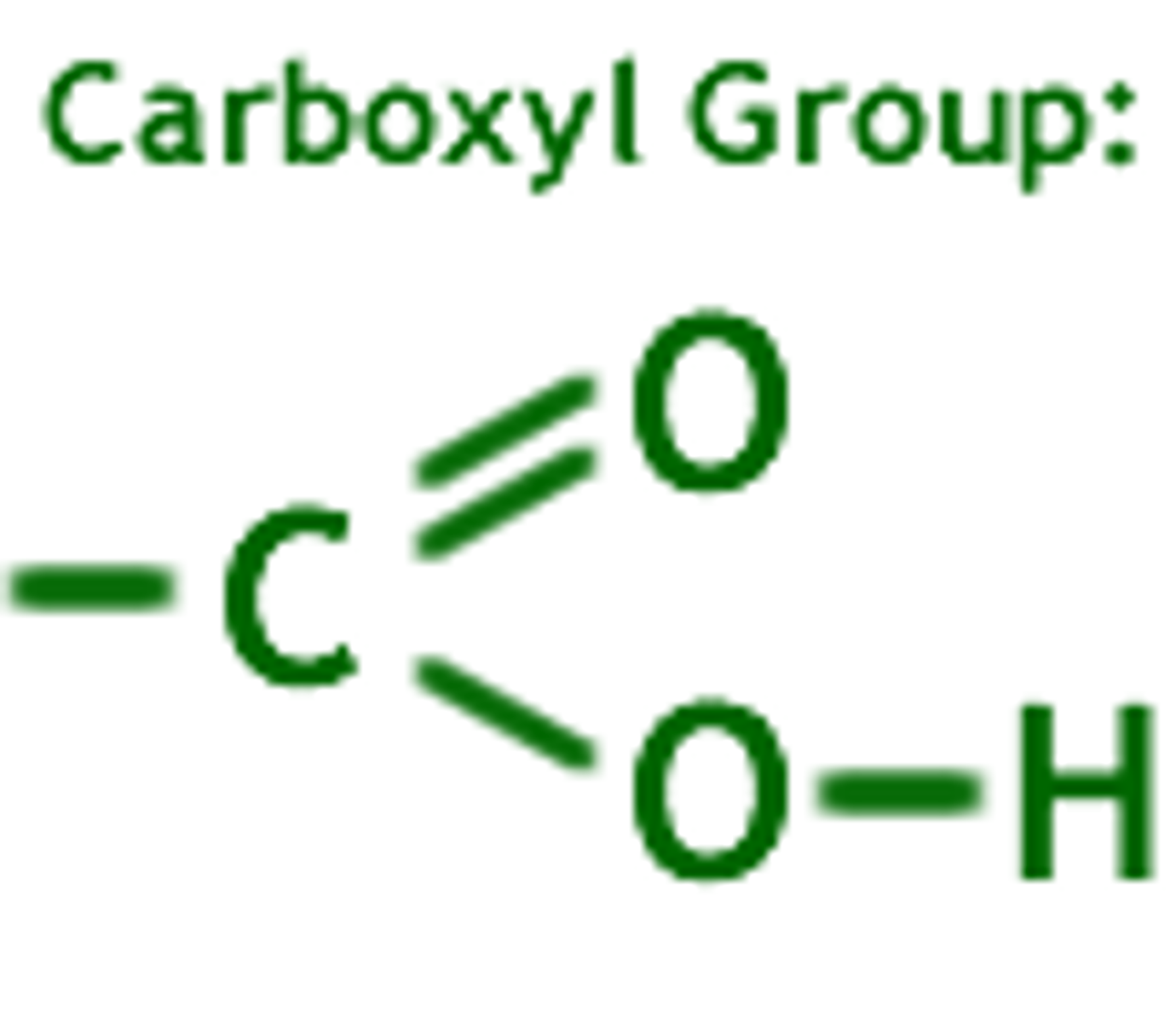
amino group
A functional group that consists of a nitrogen atom bonded to two hydrogen atoms, NH2
Amino acid sequence
determines protein structure and function

alpha helix
A spiral shape constituting one form of the secondary structure of proteins, arising from a specific hydrogen-bonding structure.

beta pleated sheet
One form of the secondary structure of proteins in which the polypeptide chain folds back and forth, or where two regions of the chain lie parallel to each other and are held together by hydrogen bonds.

disulfide bonds
Strong chemical side bonds formed when the sulfur atoms in two adjacent protein chains are joined together.

R group
a functional group that defines a particular amino acid and gives it special properties.

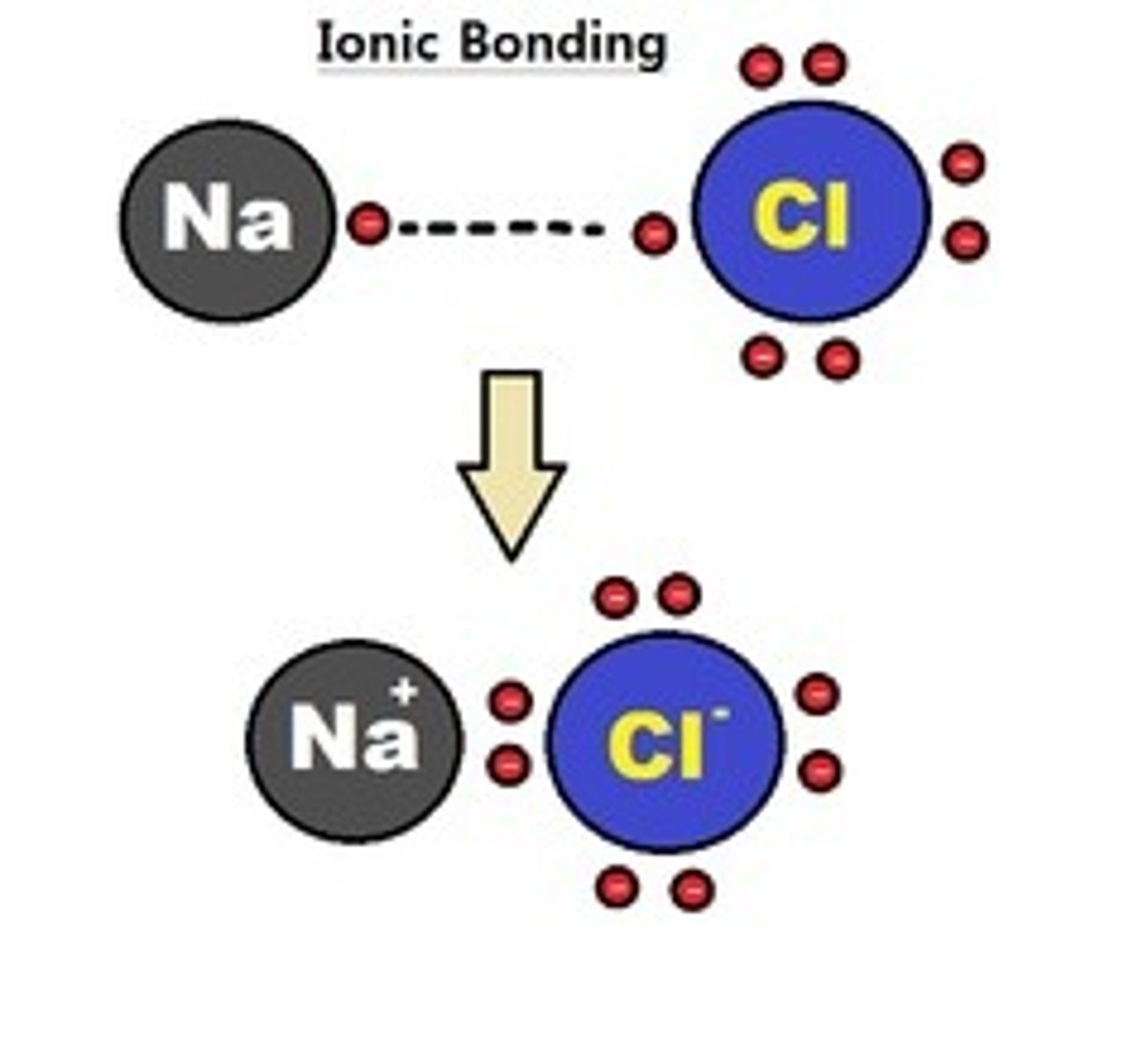
Ionic bonds
Formed when one or more electrons are transferred from one atom to another, between + and - chain deep in a protein
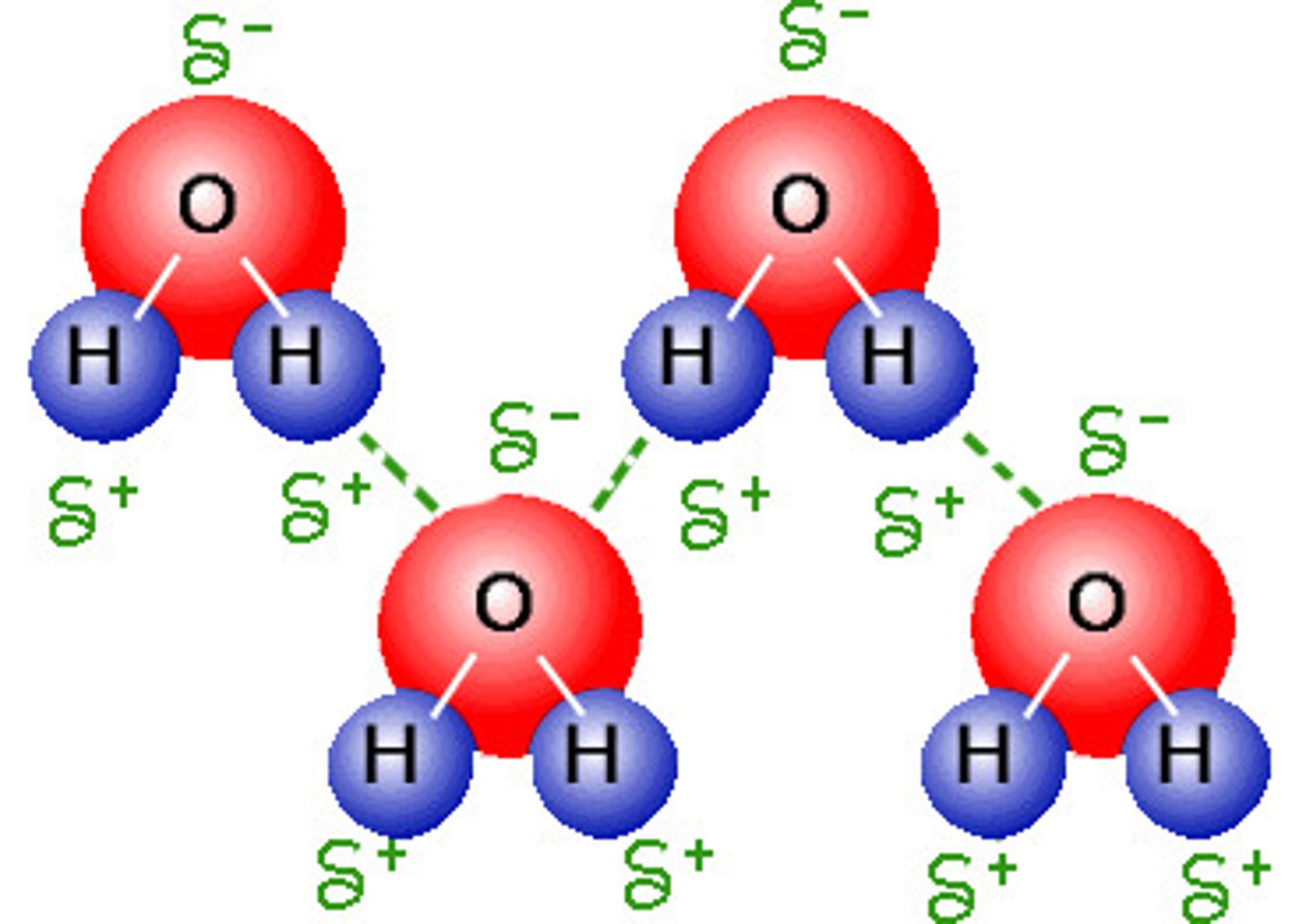
Hydrogen bond
weak attraction between a hydrogen atom and another atom, also between side chains
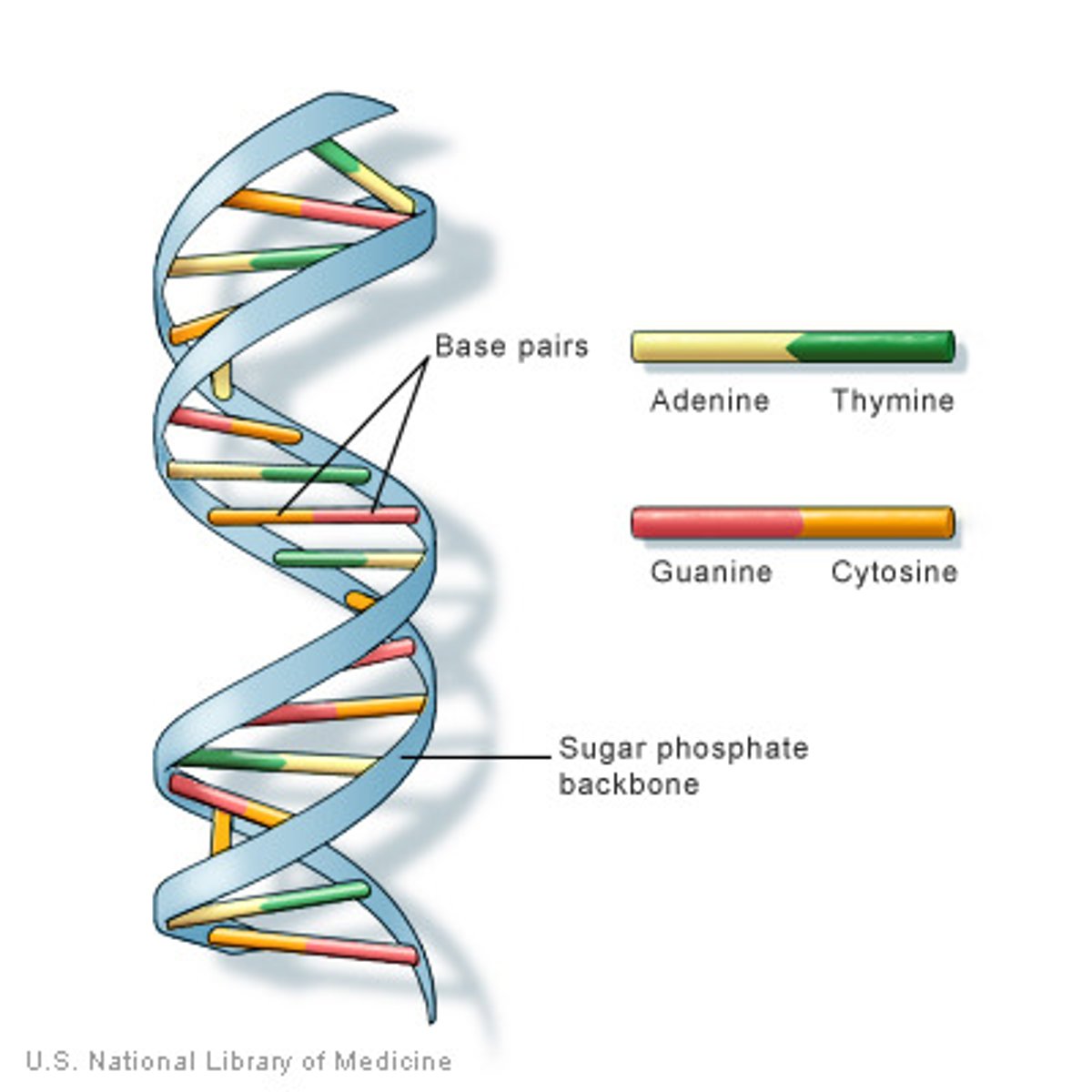
Nucleic Acids
macromolecules containing hydrogen, oxygen, nitrogen, carbon, and phosphorus, DNA and RNA

RNA
single-stranded nucleic acid that contains the sugar ribose

DNA
double stranded double helix nucleic acid that contains teh sugar deoxyribose
Role of RNA
Decoding mechanism for DNA. Decodes and carries message of DNA from nucleous to ribosomes in cytoplasm for manufacture. Uses Messenger RNA (mRNA), Ribosomal RNA (rRNA), Transfer RNA (tRNA). rRNA and tRNA act together to "translate" the message carried by mRNA

Role of DNA
storing, copying, and transmitting information, genetic information

Purine
a nitrogenous base that has a double-ring structure; either adenine or guanine

Pyrimidine
a nitrogenous base that has a single-ring structure; thymine, cytosine, or uracil

RNA bases
Adenine, Uracil, Cytosine, Guanine

DNA bases
Adenine, Thymine, Guanine, Cytosine

Double helix bonds
- attraction due to hydrogen bonds
- sugar phosphate chains hold these together

Hydrolysis
Breaking down complex molecules by the chemical addition of water
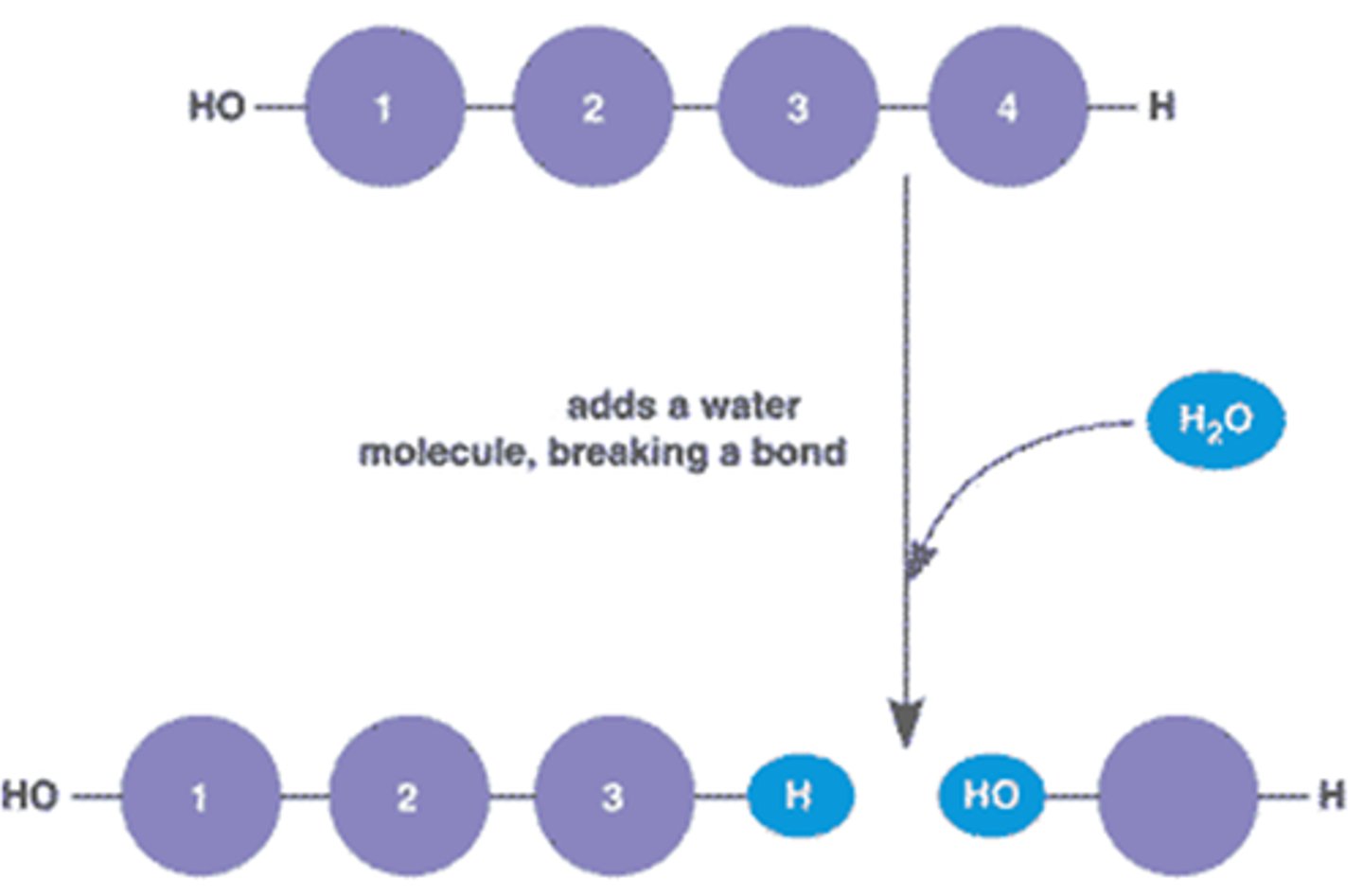
Enzymes end in
-ase
substrate
A specific reactant acted upon by an enzyme

transpiration
Transpiration is the process of water movement through a plant and its evaporation from aerial parts, such as leaves, stems and flowers. This is caused by cohesion, adhesion and osmotic potential.

endothermic thermoregulation
Heated from within the body
Relies on hydrogen bonds between water molecules to decrease speed of temperature change
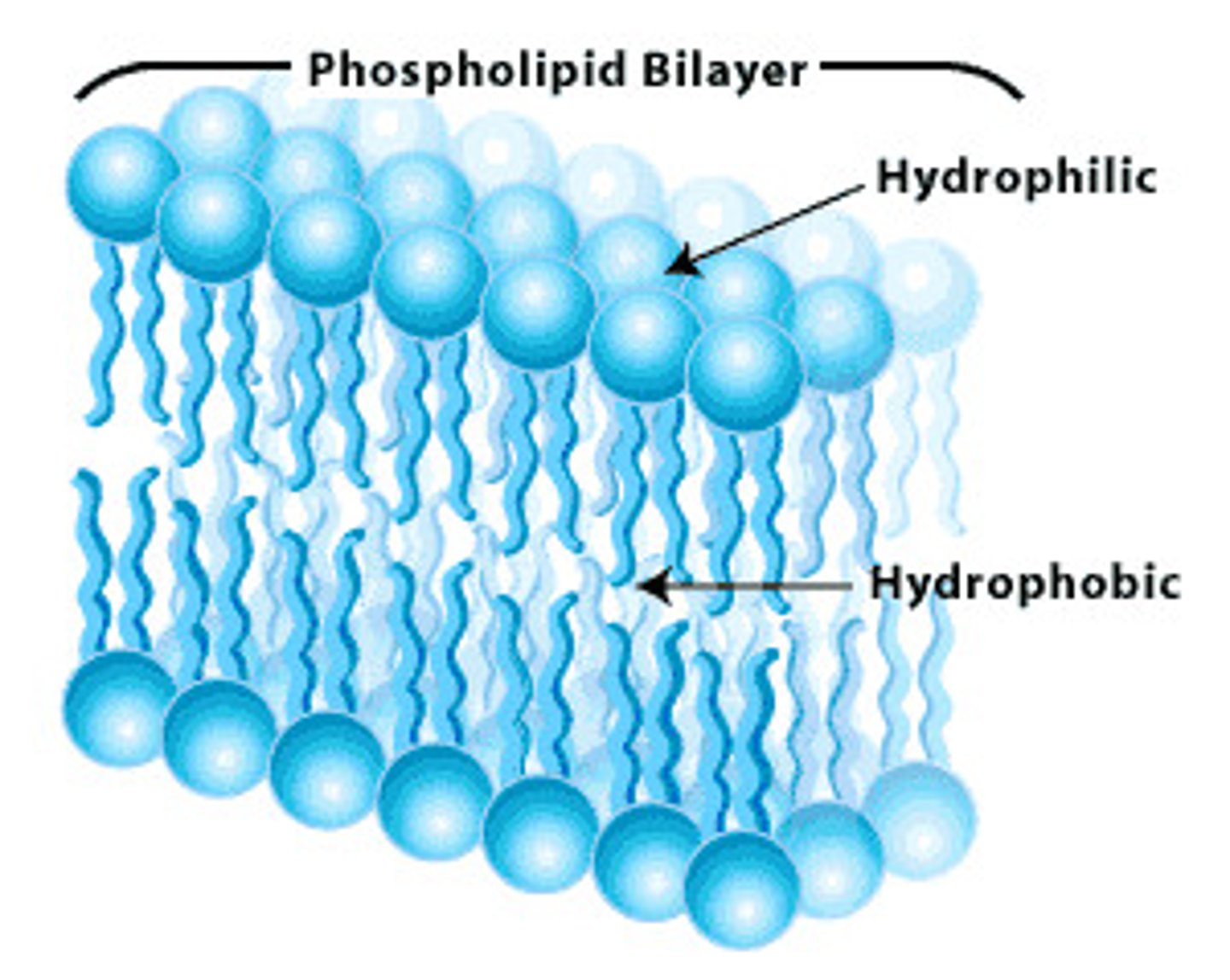
Plasma membrane structure
phospholipid bilayer with embedded proteins, hydrophobic tails, hydrophyllic heads

eukaryotic genetic material
linear, introns, in nucleus, histone to wrap around, usually many chromosomes, chromatin

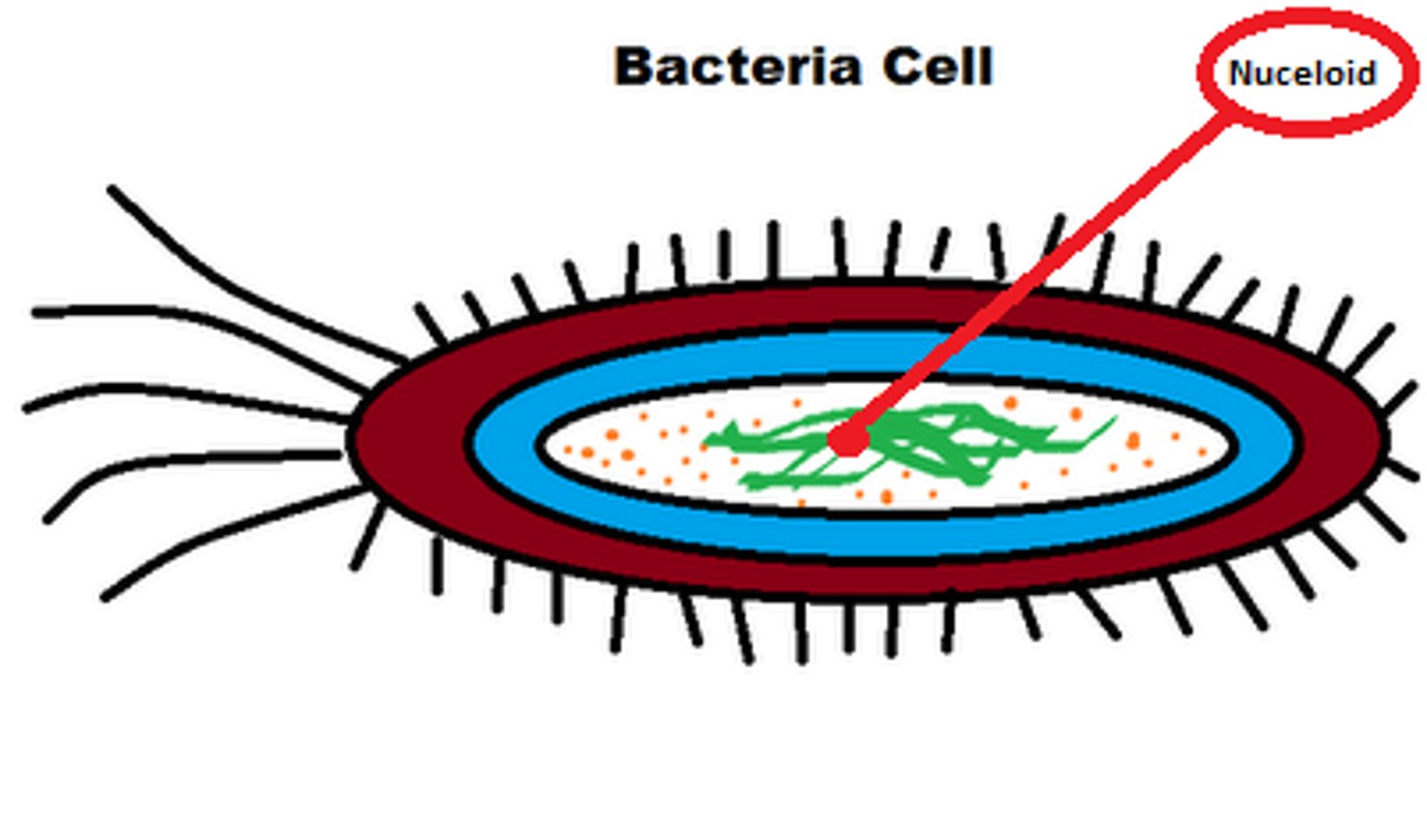
prokaryotic genetic material
circular, plasmids, no introns, not in nucleus, supercoiled, usually 1 chromosome
Water polarity
uneven sharing of electrons that produces a negative charge near the oxygen atom and positive charges near each hydrogen atom; due to high electronegativity of oxygen

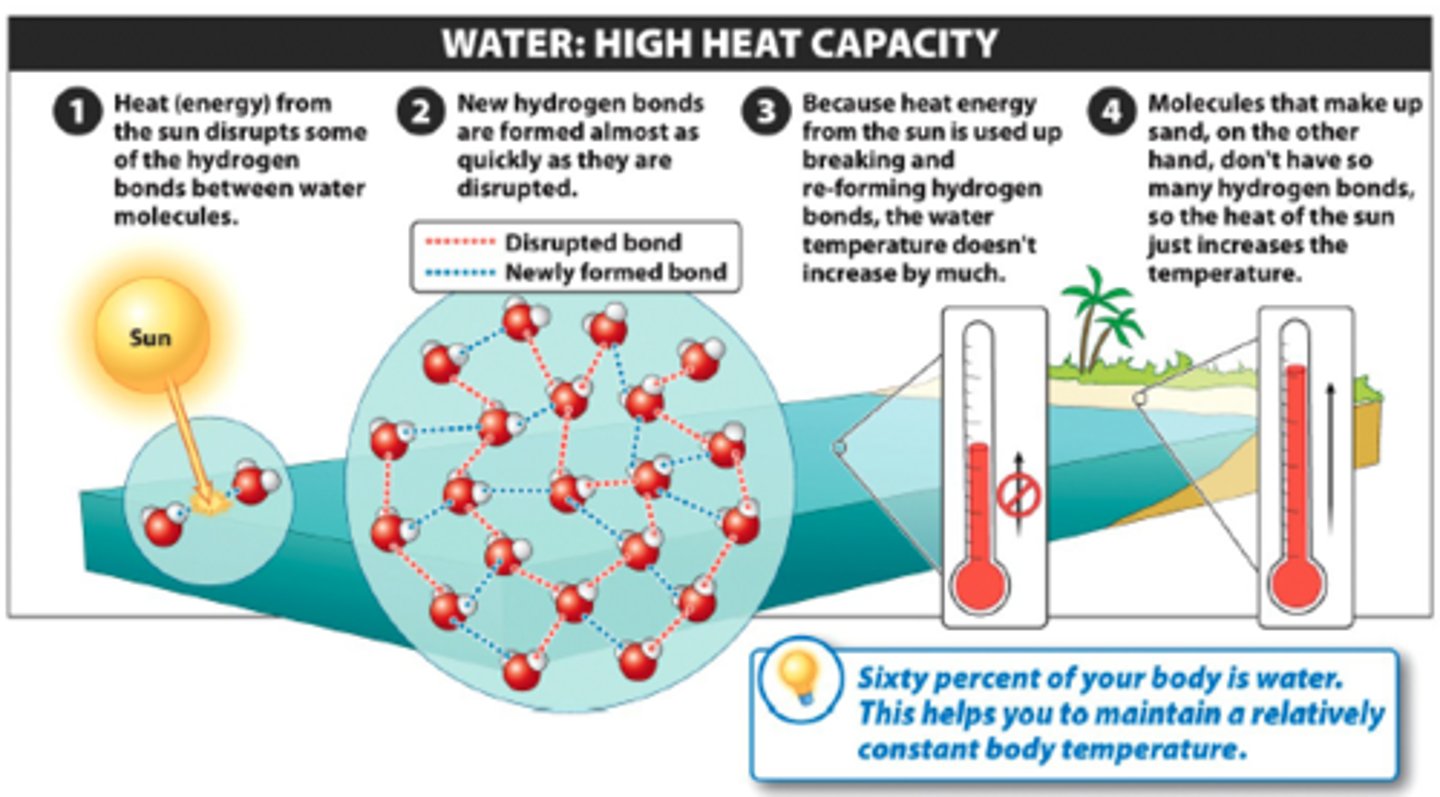
Specific Heat/ High Heat capacity of H2O
Water is able to absorb a large amount of energy before changing temperature by one degree celsius. It takes water a long time to heat and a long time to cool.
High heat of vaporization of H2O
As a result of the network of hydrogen bonding present between water molecules, a high input of energy is required to transform one gram of liquid water into water vapor,

Adhesion of water
Water sticks to any surface that is polar or has exposed nitrogens or oxygens.

Cohesion of water
Attraction between molecules of the same substance. Due to hydrogen bonding that contributes to the transport of water against gravity in plants. Hydrogen bonds between the oxygen of one water molecule and a hydrogen of another water molecule.

Surface tension (of water)
the intermolecular hydrogen bonds between molecules of water at the surface due to cohesion

Frozen water density
Less than liquid water as molecules move apart below 4 degrees C due to polarity

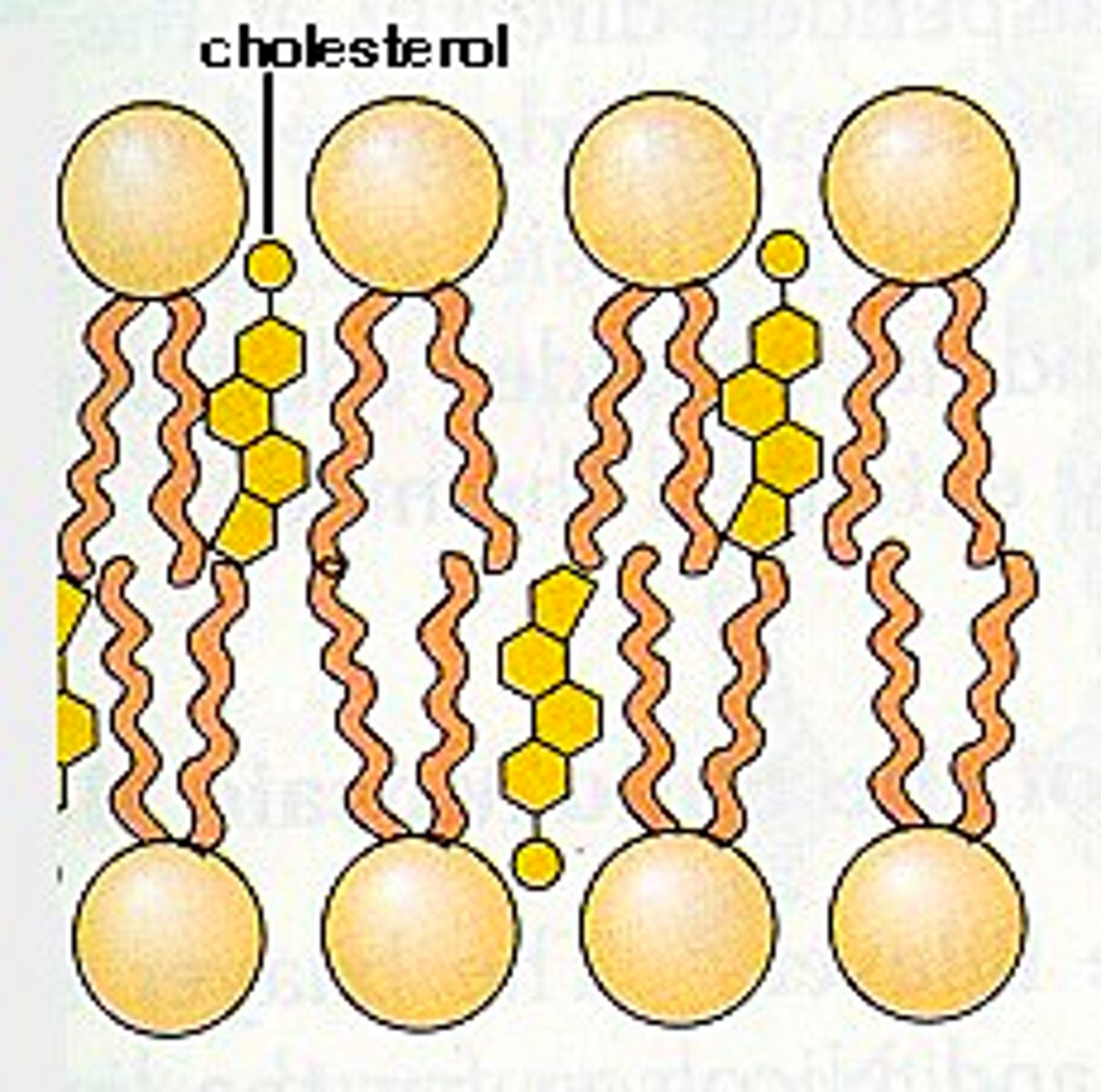
Cholesterol in Plasma Membrane
Ring Structure, embedded
stabilizes the structure of the plasma membrane when the temperature changes
Proteins in Plasma Membrane
-receptors: proteins found on the outer surface of the plasma membrane that transmit signals from the outside of the cell to the inside
- structural proteins: proteins on the inner side of the plasma membrane
- transport proteins: span the entire membrane and create tunnels through which certain substances can enter and leave the cell

Glycolipids
Membrane carbohydrates that are covalently bonded to lipids.
Cell Recognition

Phospholipids in Cell Membrane
polar head, non polar tails
Selectively permeable
fluidity

Null hypothesis
a statement or idea that can be falsified, or proved wrong
cell
The basic unit of structure and function in living things
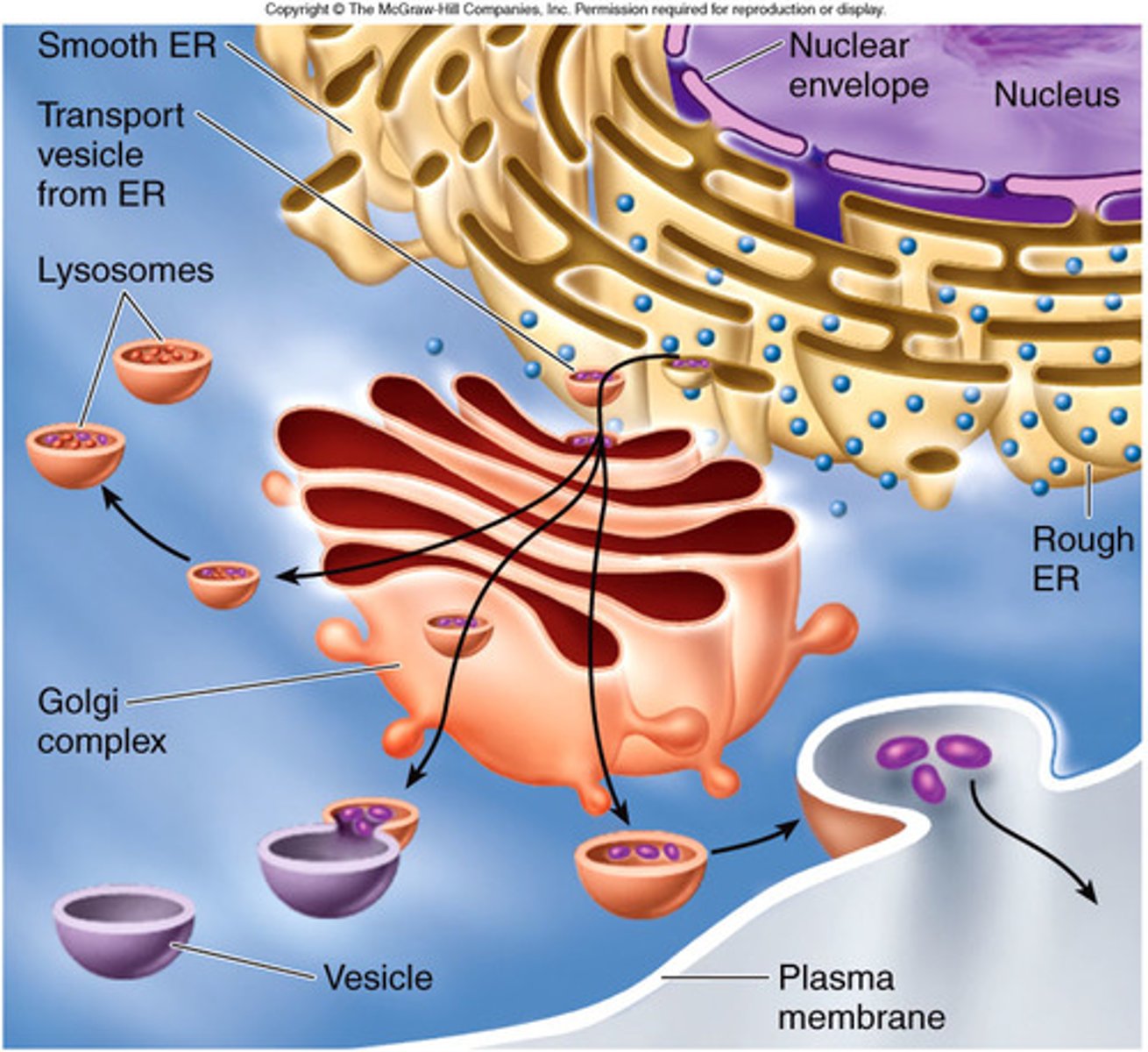
endomembrane system
The collection of membranes inside and around a eukaryotic cell, related either through direct physical contact or by the transfer of membranous vesicles.
ribosomes
site of protein synthesis, rRNA and proteins, common in ALL cells (common ancestry)
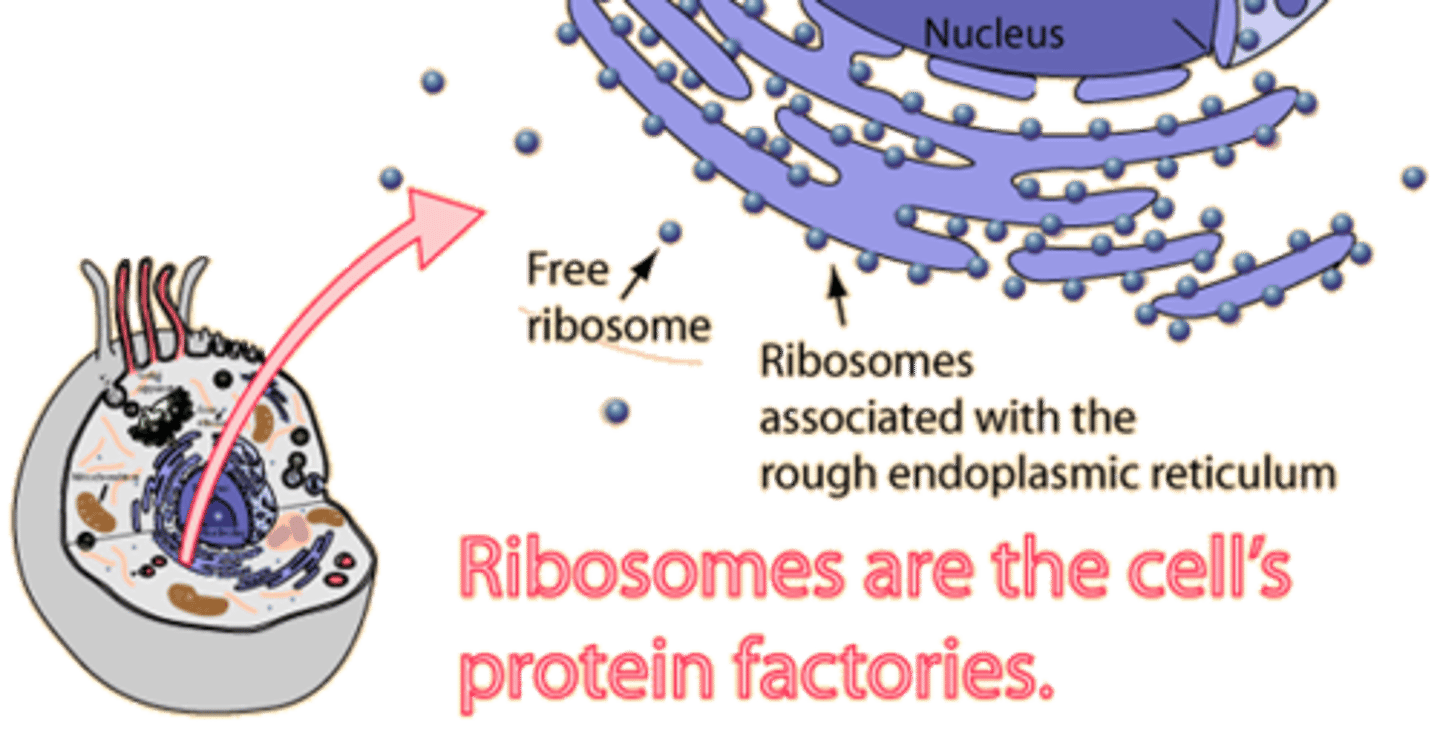
Endoplasmic Reticulum
A system of membranes that is found in a cell's cytoplasm and that assists in the production, processing, and transport of proteins and in the production of lipids.
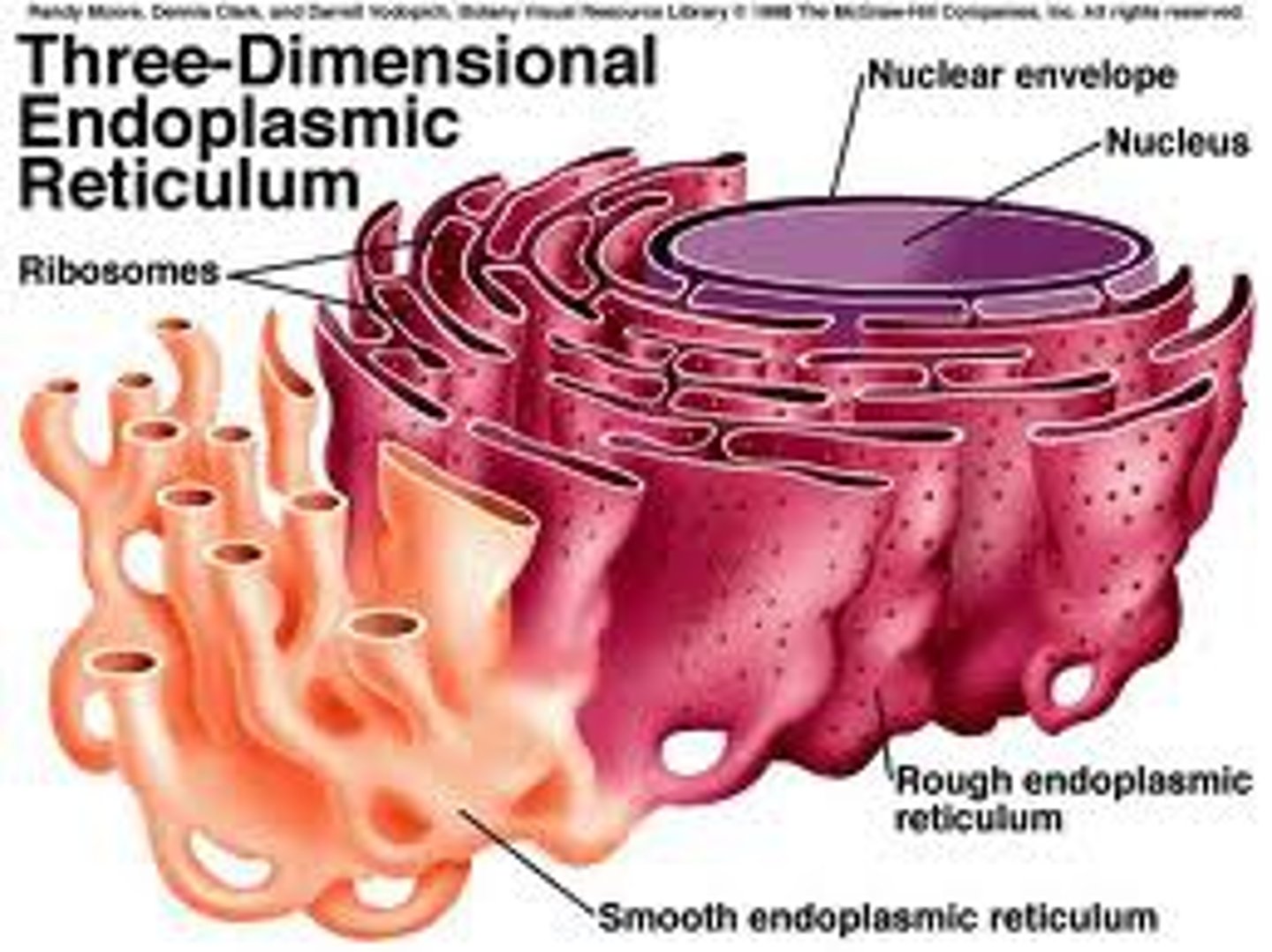
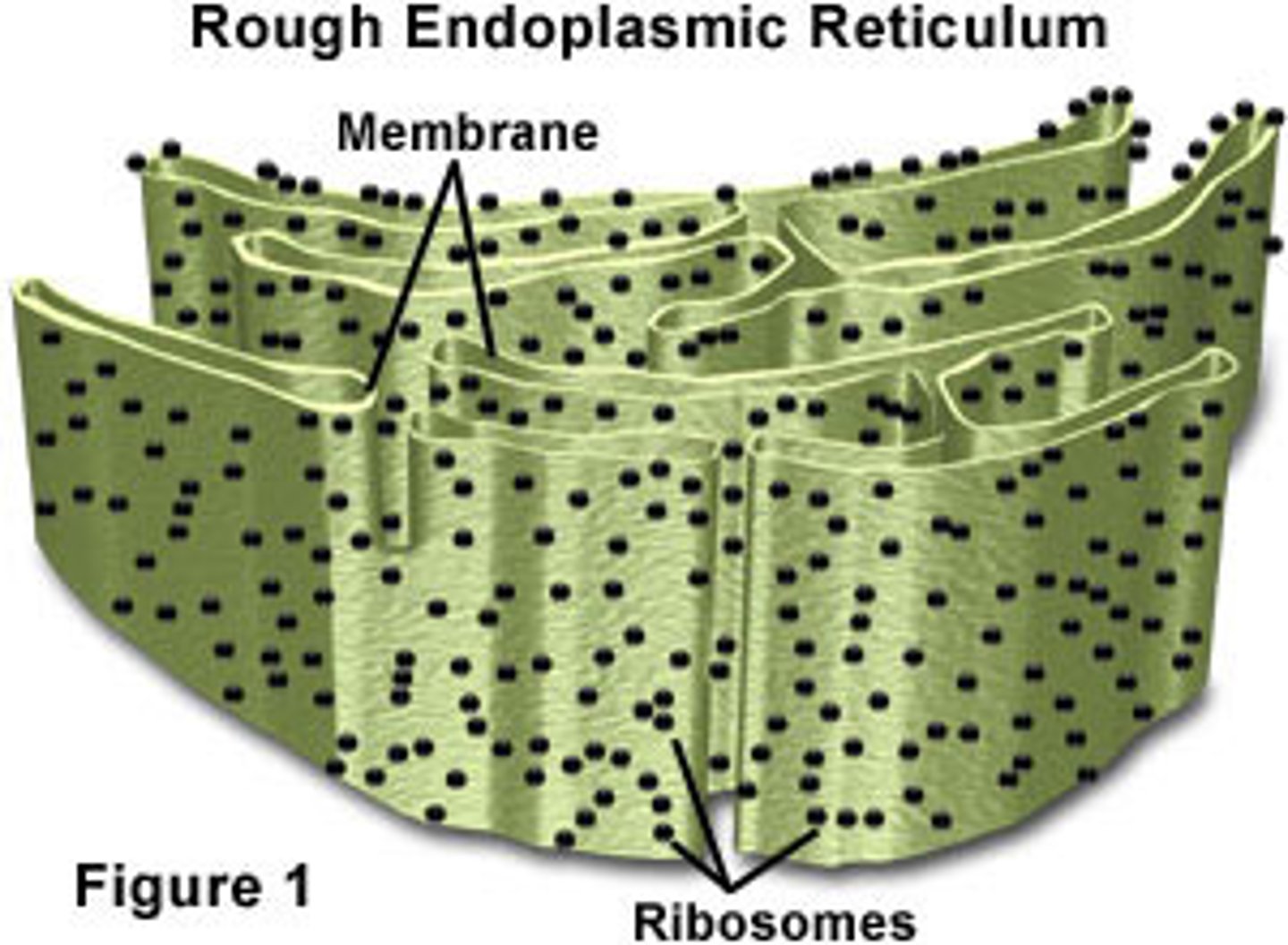
Rough Endoplasmic Reticulum
ER with ribosomes to make proteins, detoxifies, lipid synthesis, fold polypeptides into tertiary from
Smooth Endoplasmic Reticulum
ER without ribosomes, lipids synthesized, calcium levels regulated, and toxic substances broken down.

Golgi Body
receives proteins and lipids from the endoplasmic reticulum, packages them, and distributes them to other parts of the cell or out of the cell via vessicles

Exocytosis
a process by which the contents of a cell vacuole are released to the exterior through fusion of the vacuole membrane with the cell membrane.

Mitochondria
Sites for cellular respiration to make ATP, has its own DNA and ribosomes, Only in eukaryotes.

Structure of Mitochondria
2 membranes, highly folded (convoluted) inner membrane increases surface area

Aerobic Respiration
Requires oxygen to release energy from glucose, occurs in mitochondria

Phases of Aerobic Respiration
glycolysis, citric acid cycle, electron transport chain
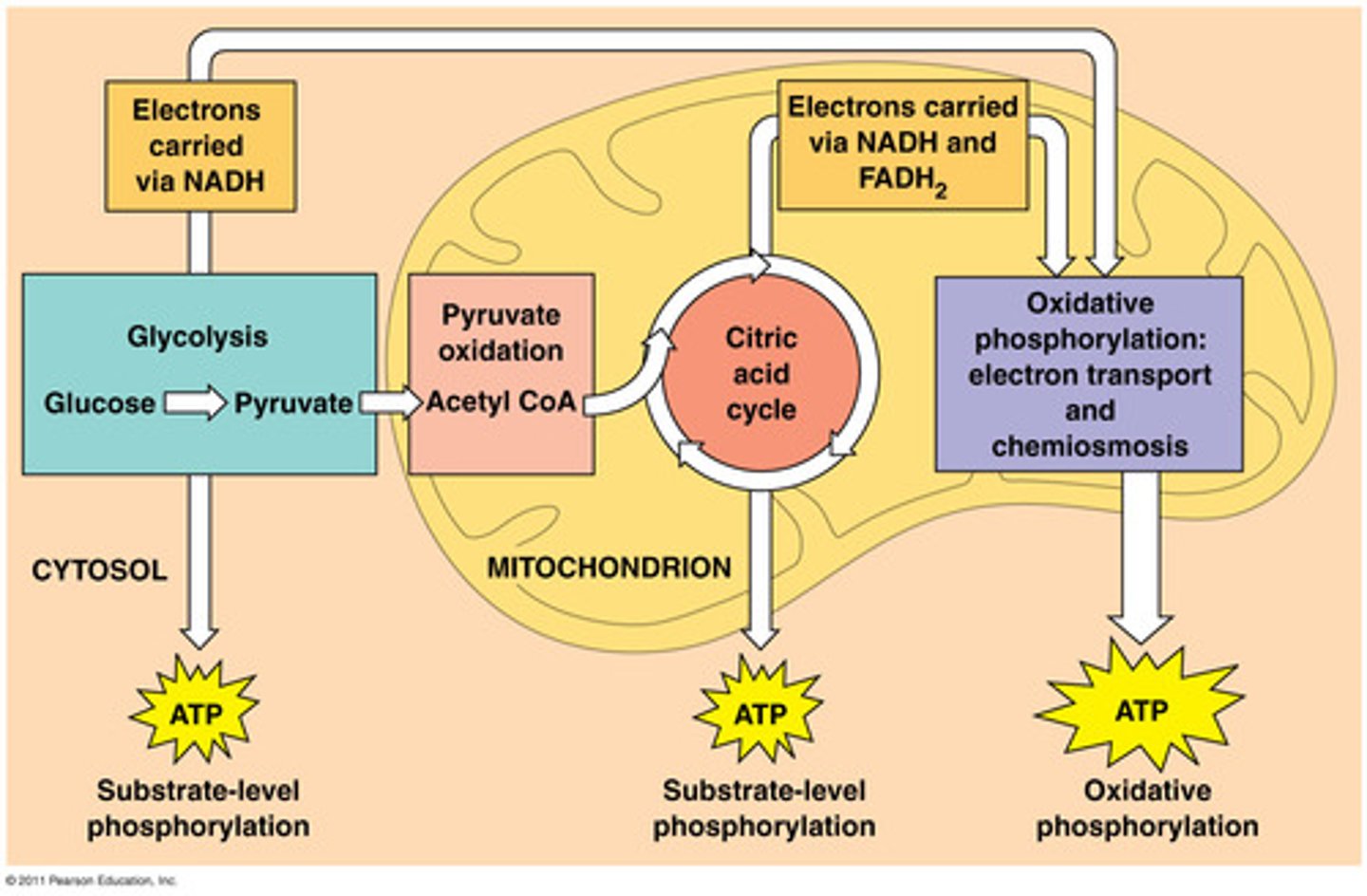
Glycolysis
first step in releasing the energy of glucose, in which a molecule of glucose is broken into two molecules of pyruvic acid, occurs in cytoplasm

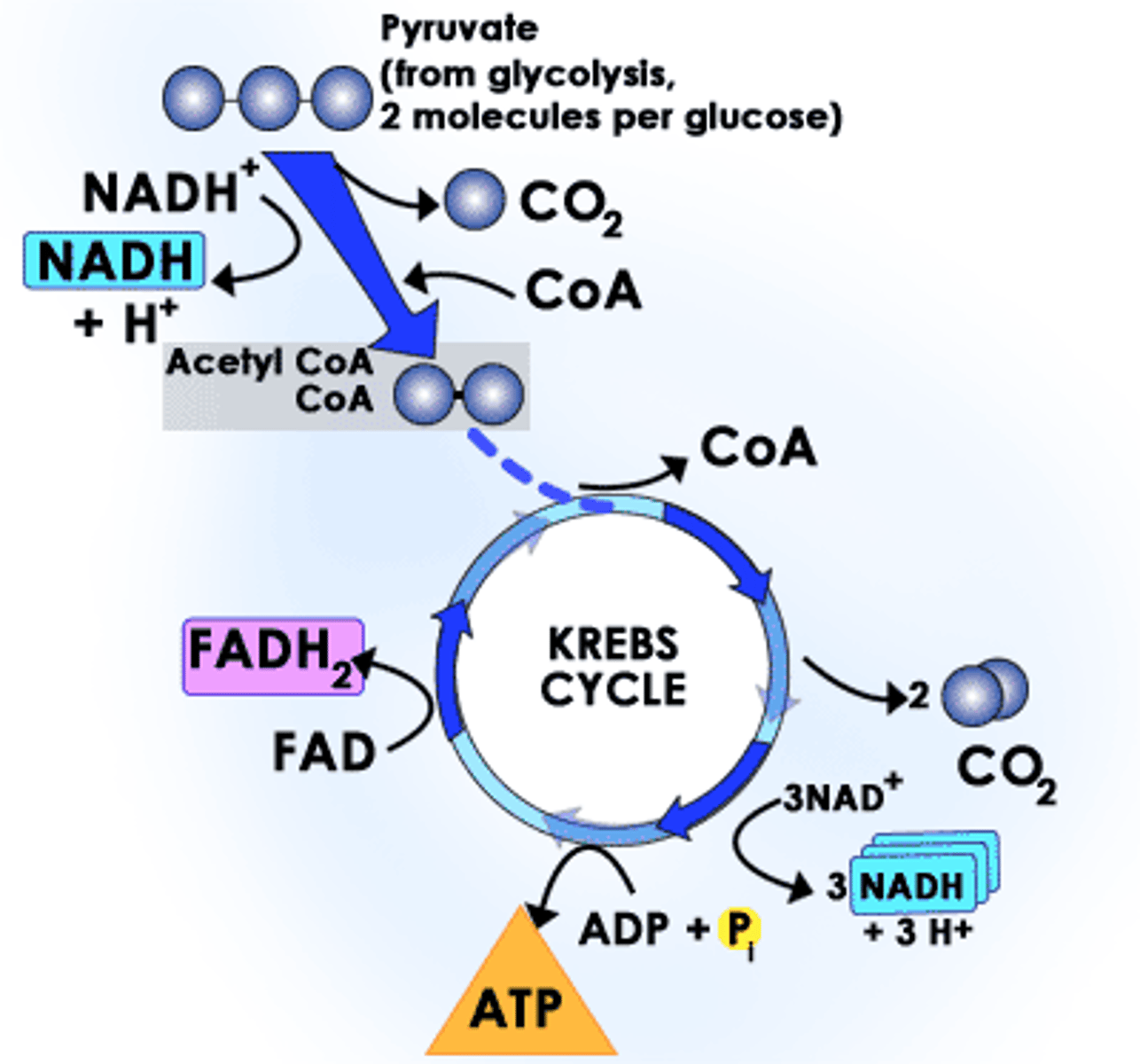
Krebs Cycle (Citric Acid Cycle)
second stage of cellular respiration, in which pyruvic acid is broken down into carbon dioxide in a series of energy-extracting reactions, in mitochondrial matrix
Electron Transport Chain/ Chemiosmosis (Specific)
-in mitochondrial cristae membrane
Part 1: A Proton Gradient
-ETC produces proton gradient
NADH and FADH2 carry high energy electrons from Krebs cycle and Glycolysis to ETC where they power the pumping protons H+ across cristae membrane creating proton gradient. Potential energy like water behind dam
Part 2: Chemiosmosis or Oxidative Phosphorylation
-uses energy stored in protein gradient to power synthesis of ATP
-ATP synthetase- a proton channel structure that spins like a turbine. As protons pour through, it spins and attached P to ADP
each NADH produce 3 ATP
each FADH produce 2 ATP
Electron Transport Chain (General)
series of electron carrier proteins that shuttle high-energy electrons during ATP-generating reactions, across inner mitochondrial membrane in respiration

redox reaction
electron transfer reactions

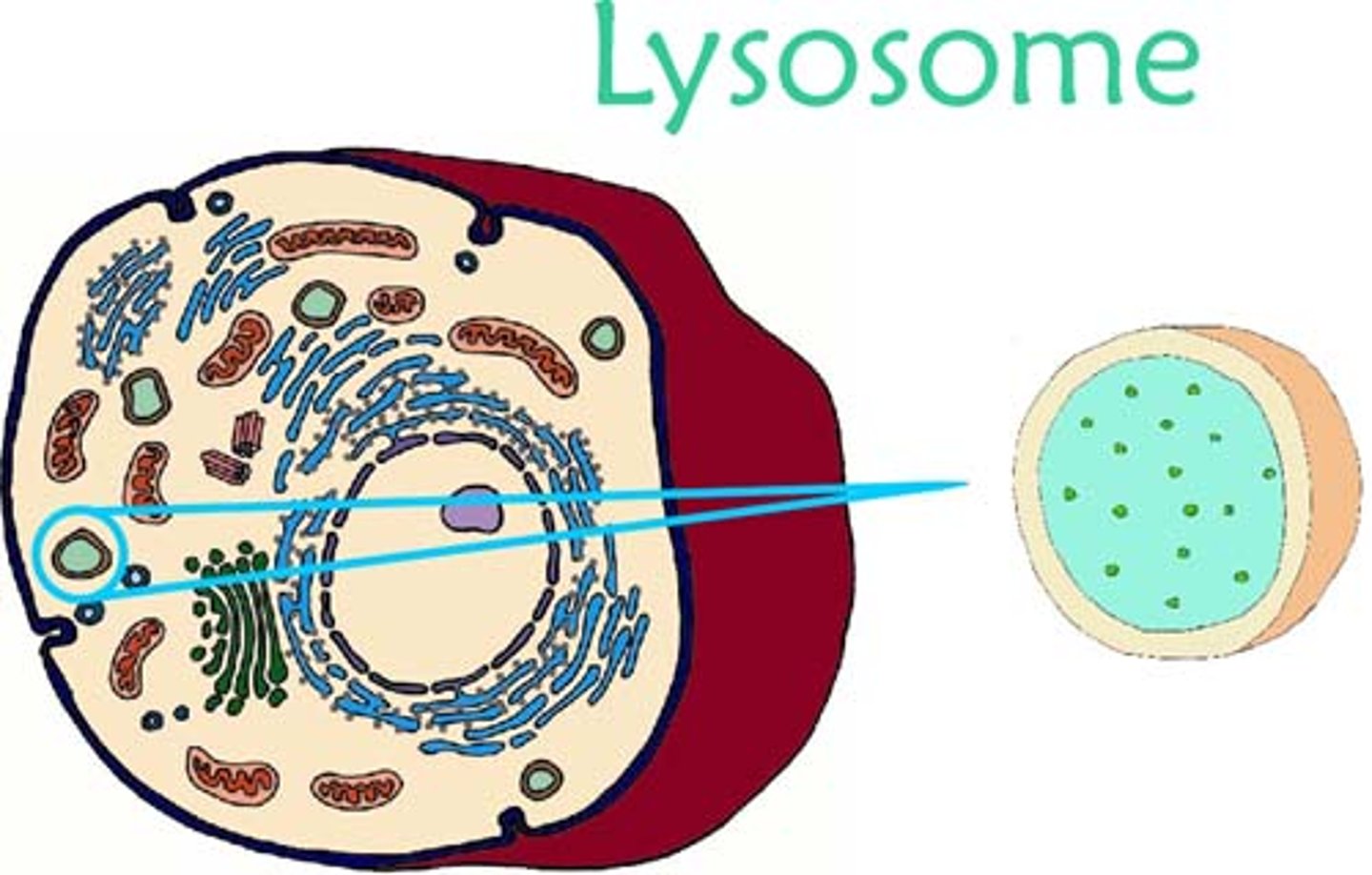
Lysosomes
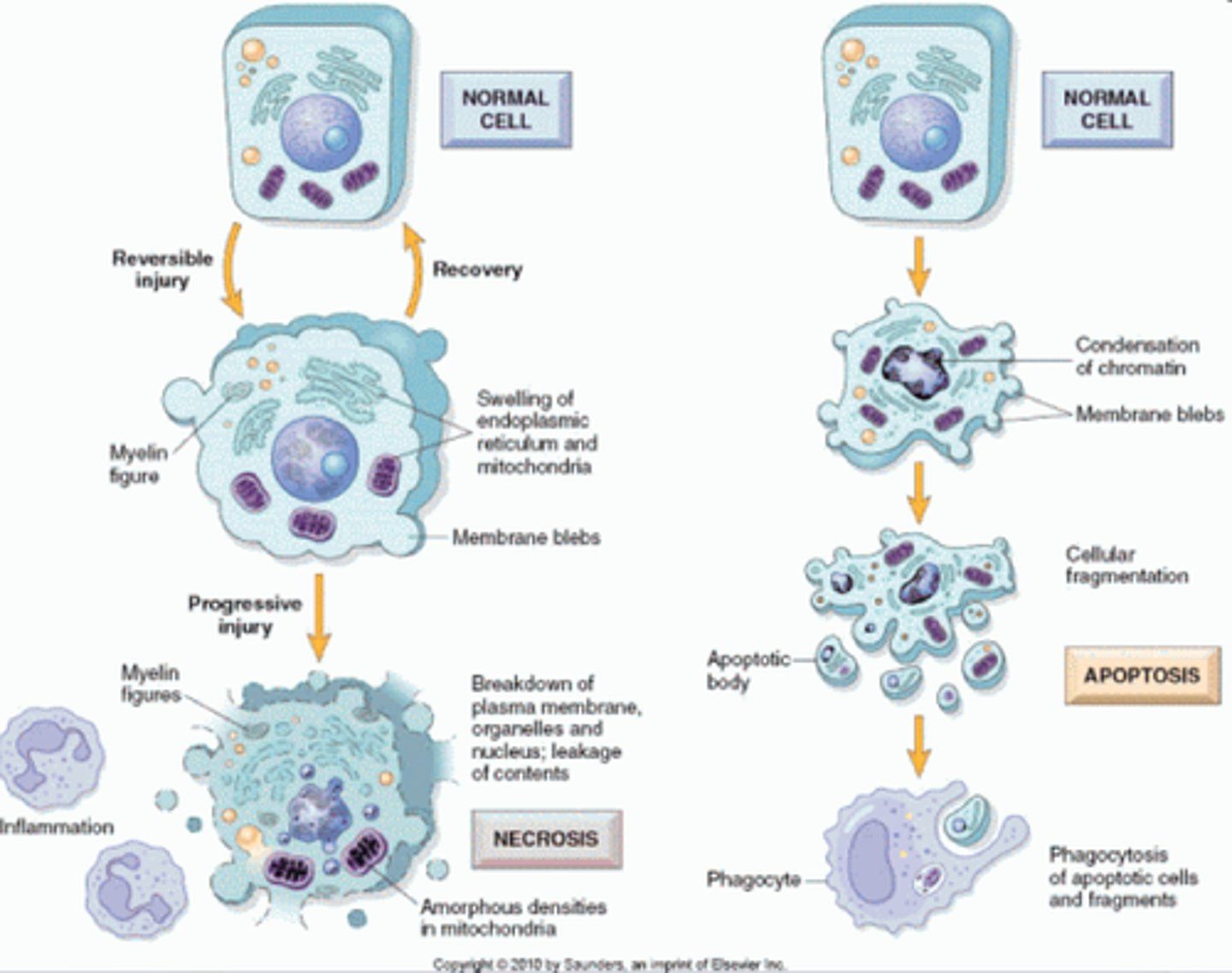
cell organelle filled with enzymes needed to break down certain materials in the cell, also plays role in cell death
Apoptosis
programmed cell death
Vacuoles
Cell organelle that stores materials such as water, salts, proteins, and carbohydrates, store and dispose wastes

Central Vacuole
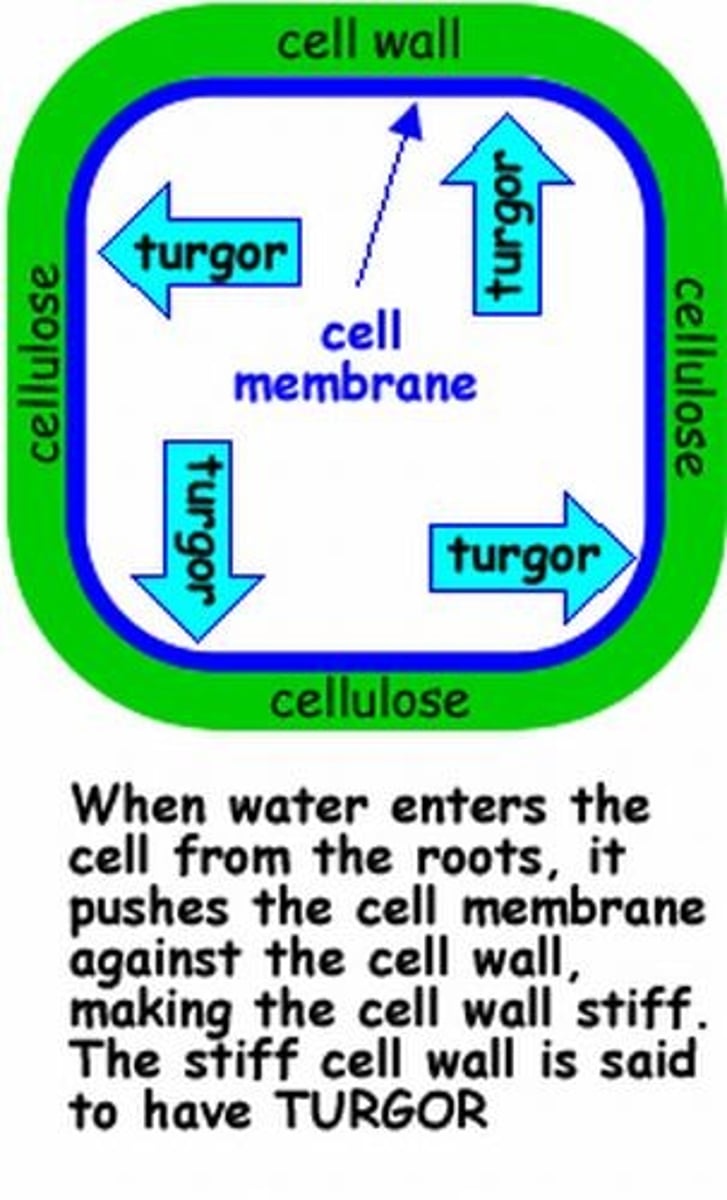
A large vacuole that rests at the center of most plant cells and is filled with a solution that contains a high concentration of solutes; turgor pressure

Turgor Pressure
the pressure that is exerted on the inside of cell walls and that is caused by the movement of water into the cell
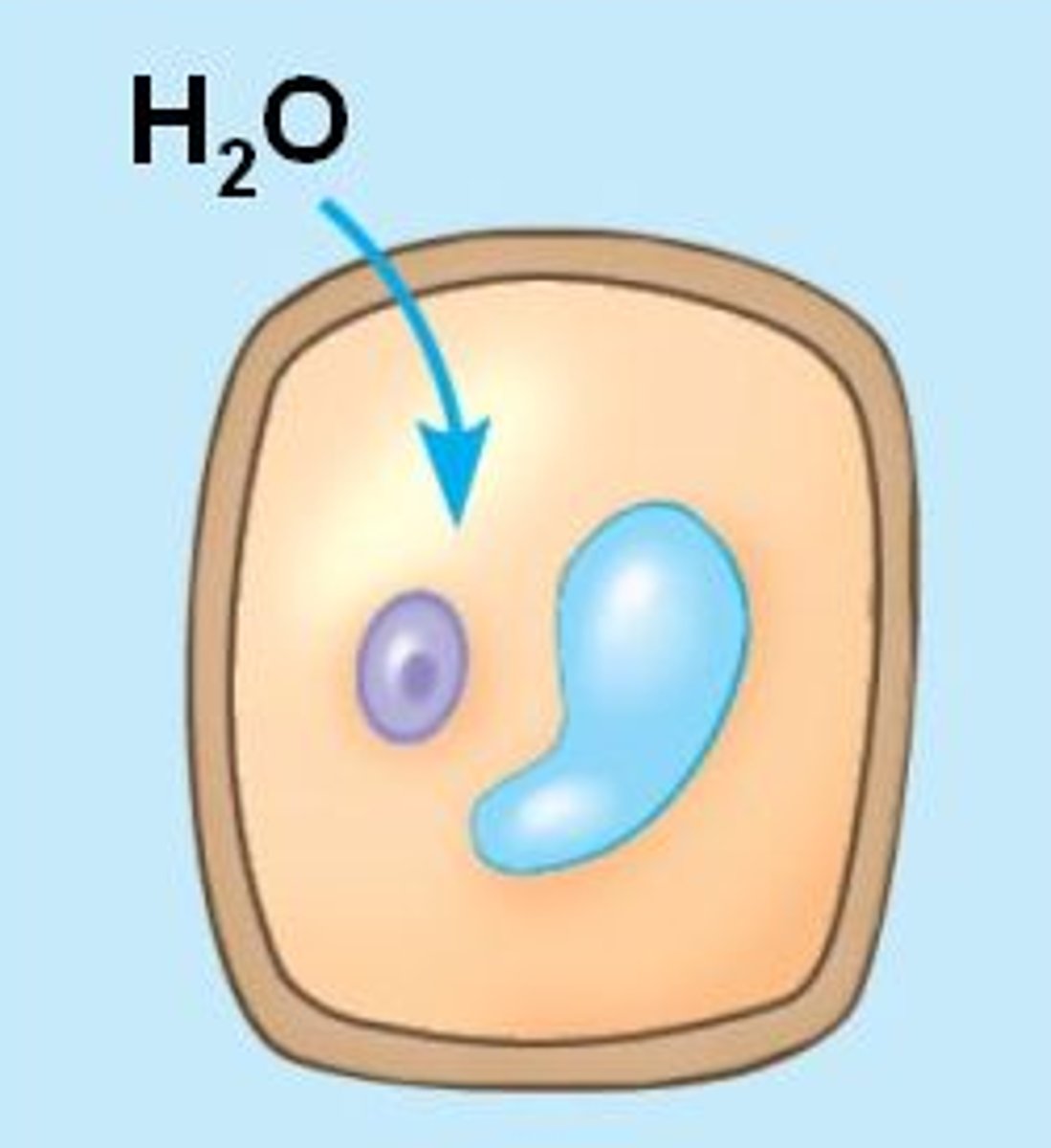
Osmosis
Diffusion of water through a selectively permeable membrane

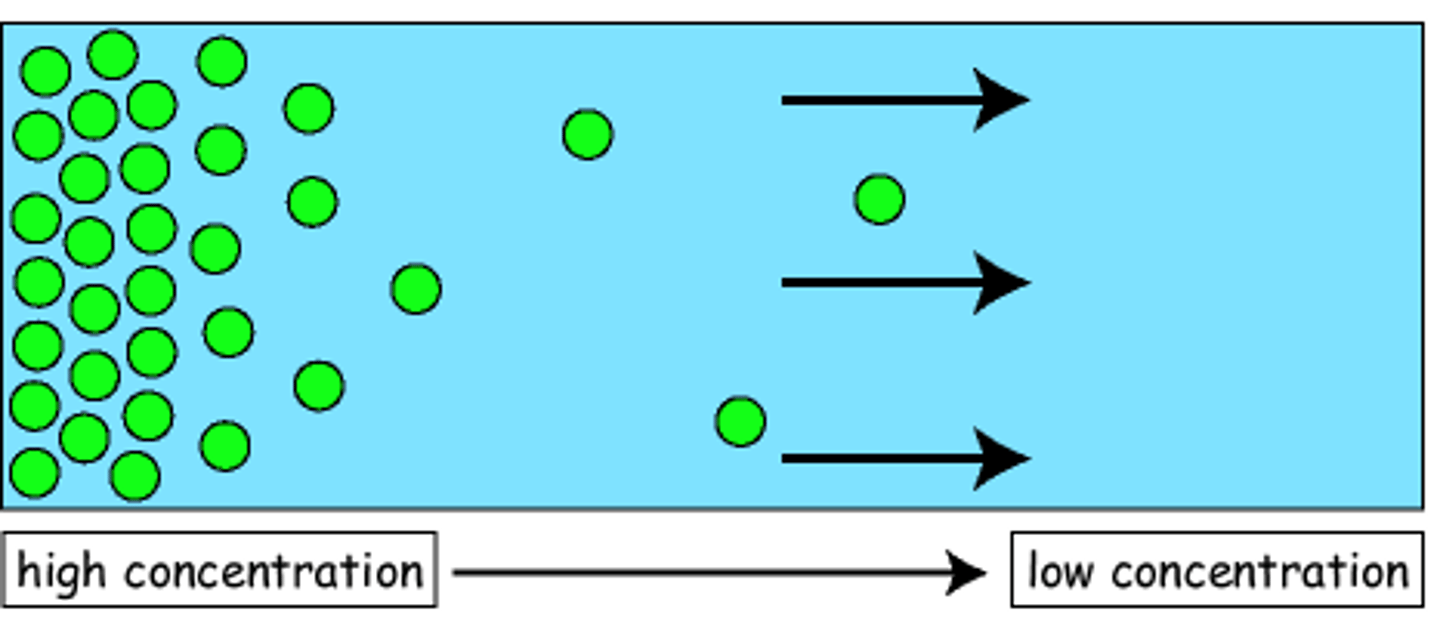
Diffusion
Movement of molecules from an area of higher concentration to an area of lower concentration.
Osmotic Potential
The tendency of water to move across a permeable membrane into a solution

Water Potential Equation
Ψ = Ψs + Ψp
water potential = solute potential + pressure potential
OR water potential = -iCRT
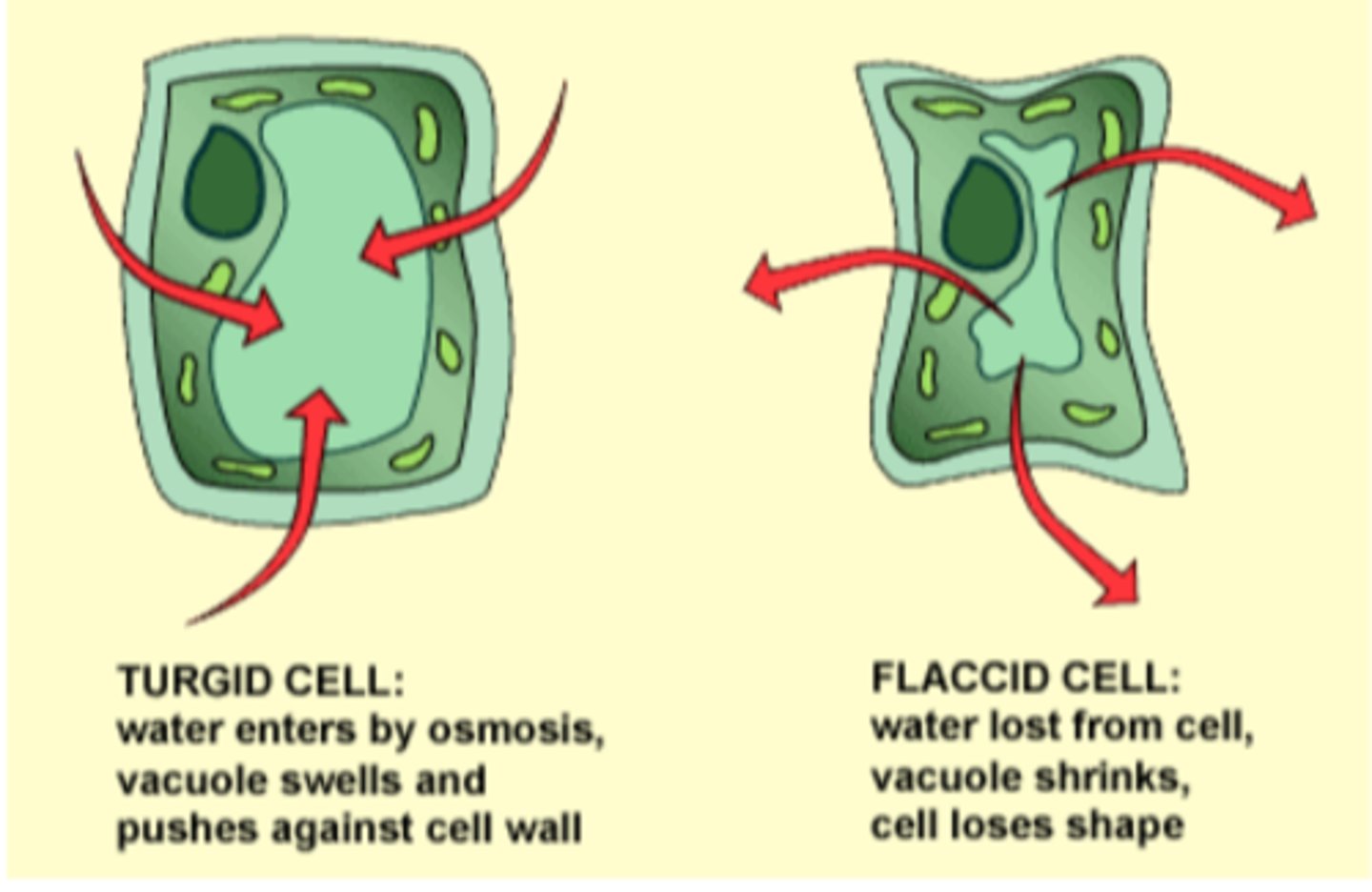
Turgid Cell
Firm cell due to water gained by osmosis
Flaccid Cell
water lost from cell, vacuole shrinks, cell loses shape
Plasmolysis
Collapse of a walled cell's cytoplasm due to a lack of water
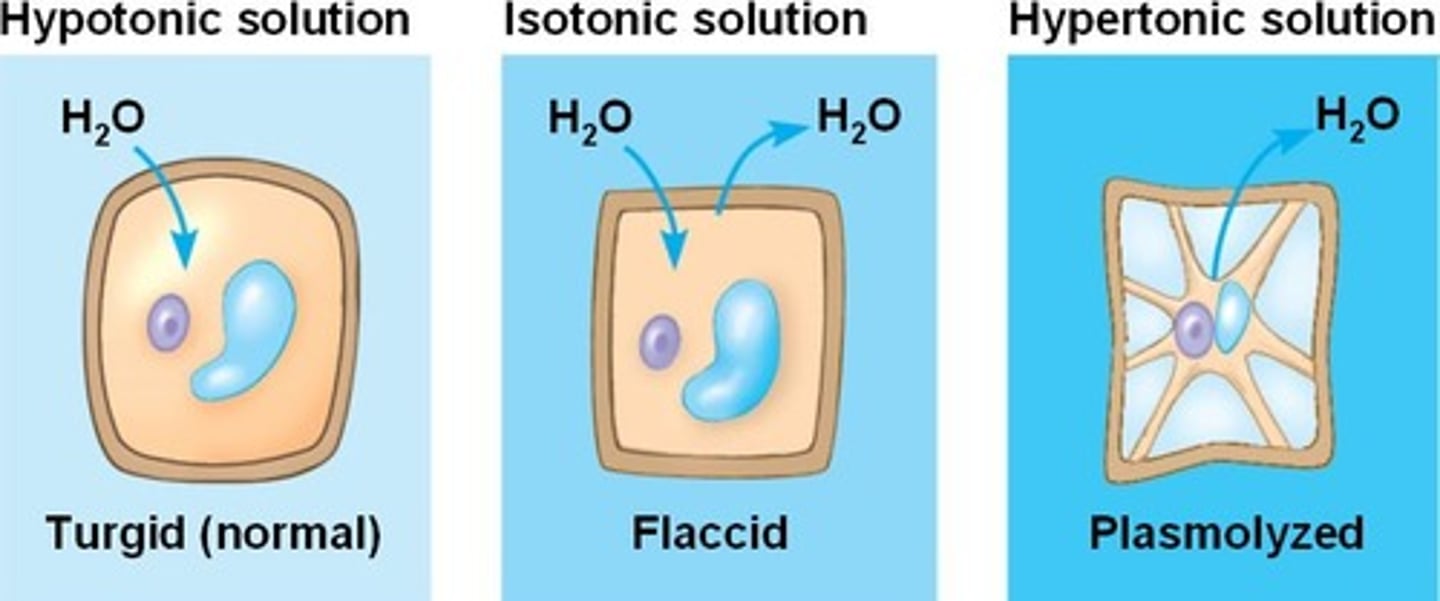
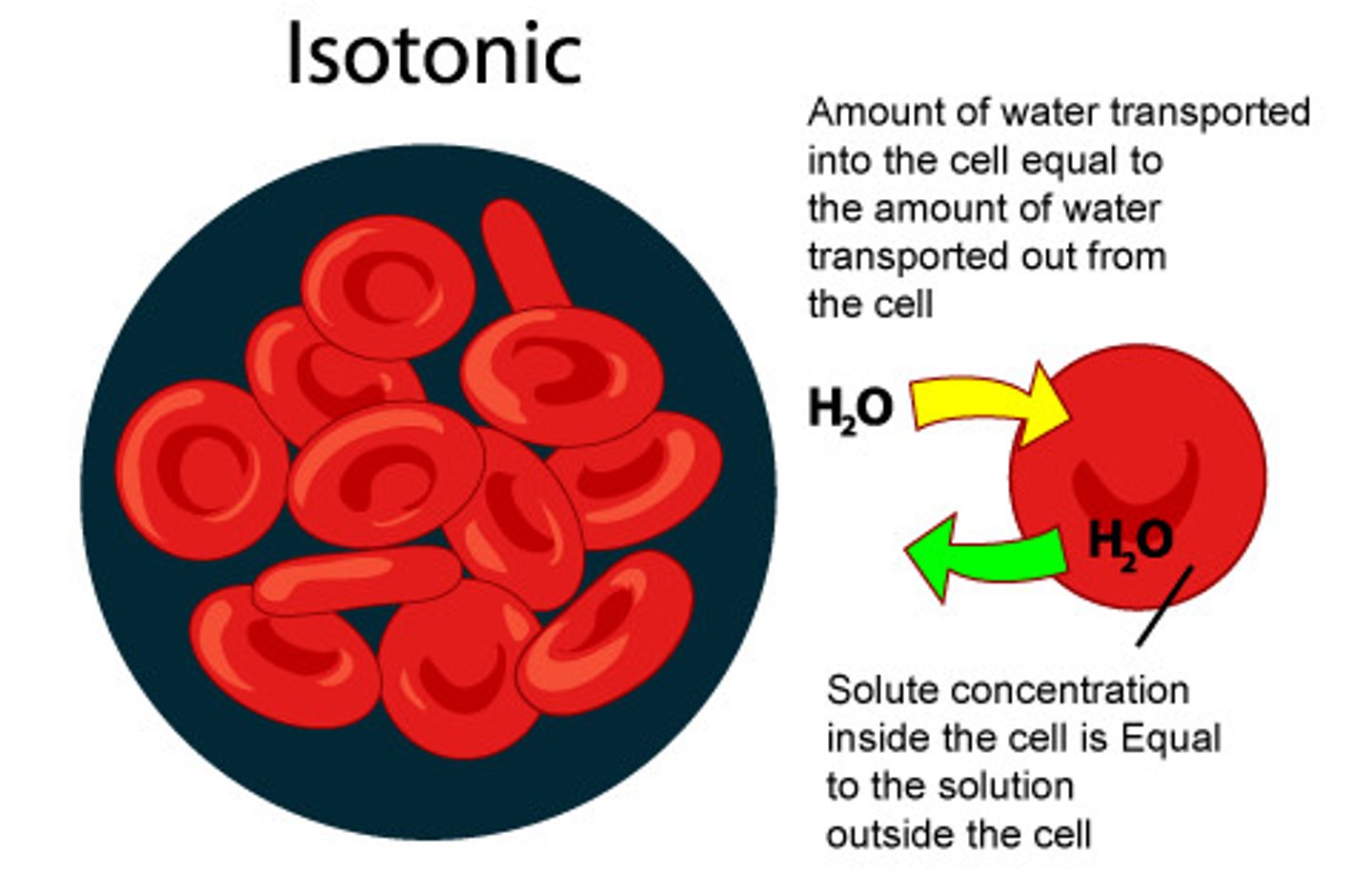
Isotonic solution
a solution whose solute concentration is equal to the solute concentration inside a cell; water enters and leaves the cell at equal rates
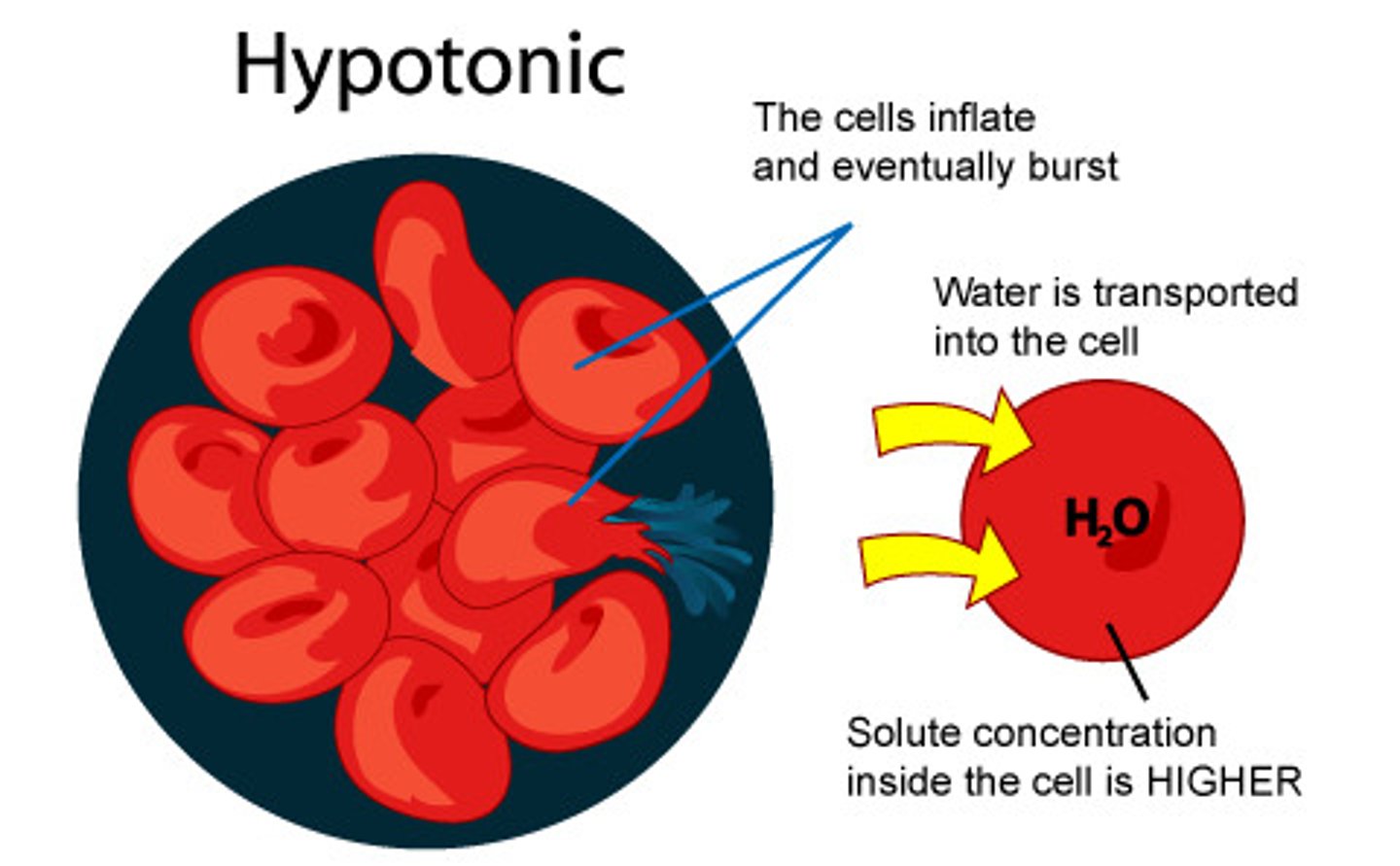
Hypotonic solution
A solution in which the concentration of solutes is less than that of the cell that resides in the solution; water leaves the cell
Hypertonic solution
A solution in which the concentration of solutes is greater than that of the cell that resides in the solution; water leaves the cell

Chloroplast
An organelle found in plant and algae cells where photosynthesis occurs

Structure of Chloroplast
organelle for photosynthesis
has double membrane
contains discs called thylakoids
thylakoids contain chlorophyll
stack of thylakoids called granum
thylakoids surrounded by a fluid called stroma
