Structure of an atom grade 9
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
11 Terms
What is an atom
An element is a substance made of only one type of atom
FACTS
An atom is mostly empty space
It has a nucleus in the middle consisting of protons
Where is everything in an atom? Protons? Electrons? Neutrons?
Neutrons- inside nucleus
Protons- inside nucleus
Electrons- OUTSIDE nucleus
how does SAN go?
Atomic mass —-SYMBOL
atomic #
What is the Bohr Rutherford rule? What goes in shells?
Rule: 2-8-8
Shells- # of protons
MOLECULES
Two of THE SAME atoms bonded together
All compounds are molecules, but not all molecules are compounds.
COMPOUNDS
A compound is a molecule made of atoms from DIFFERENT elements
All compounds are molecules, but not all molecules are compounds.
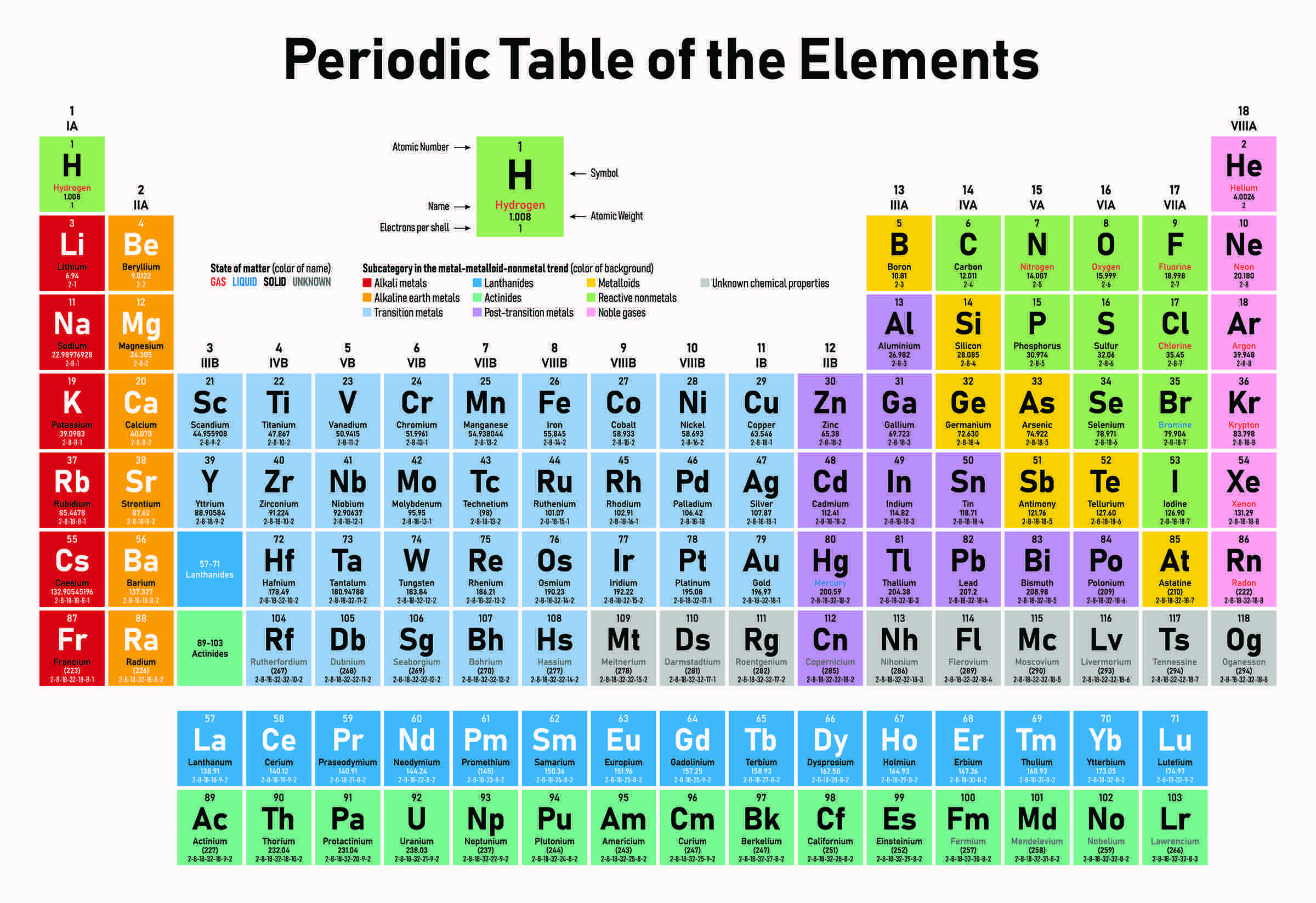
What family is where?
alkali metals
AlkaLINE earth metals
Transition metals
Metalloids
Non-metals
Halogens
Noble gases
PERIODS VS. FAMILES
Elements in a period are NOT ALIKE.
Elements in a group are similar.