MCB 102- Midterm 2
1/114
Earn XP
Description and Tags
Thomas
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
115 Terms
1st Law of Thermodynamics
energy can not be created or destroyed
2nd Law of Thermodynamics
entropy of the universe increases with every spontaneous reaction
___ pathways converge and anabolic pathways _____
catabolic, diverge
ΔG° > 0
endergonic, Keq < 1, prefers reactants
ΔG° < 0
exergonic, Keq > 1, prefers products
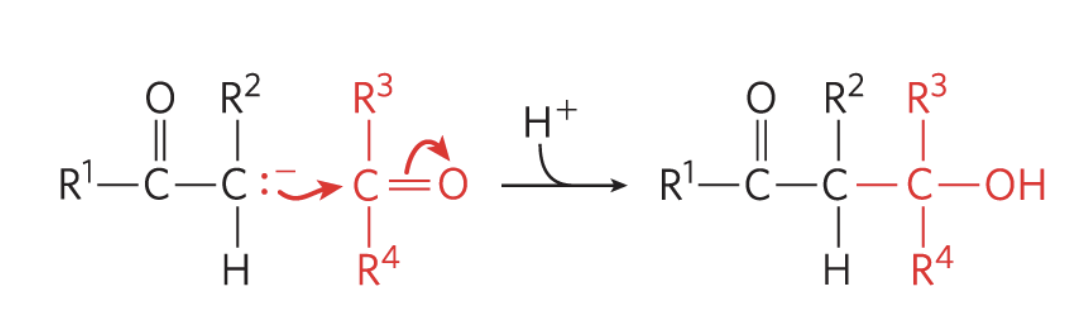
what reaction is this?
aldol condensation
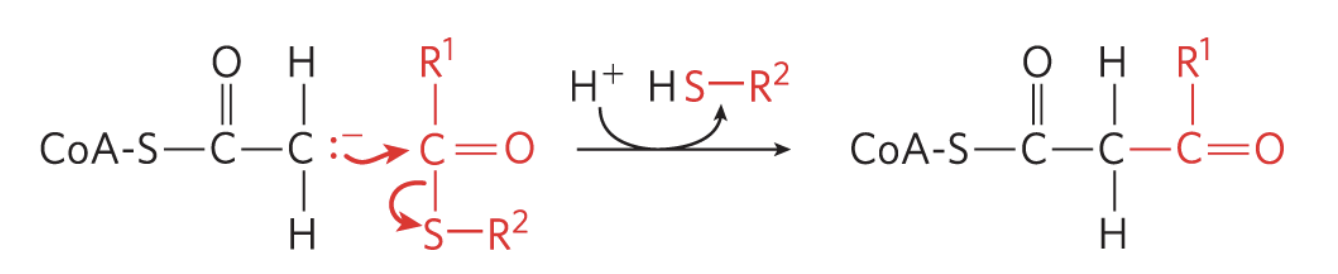
what reaction is this?
Claisen Condensation
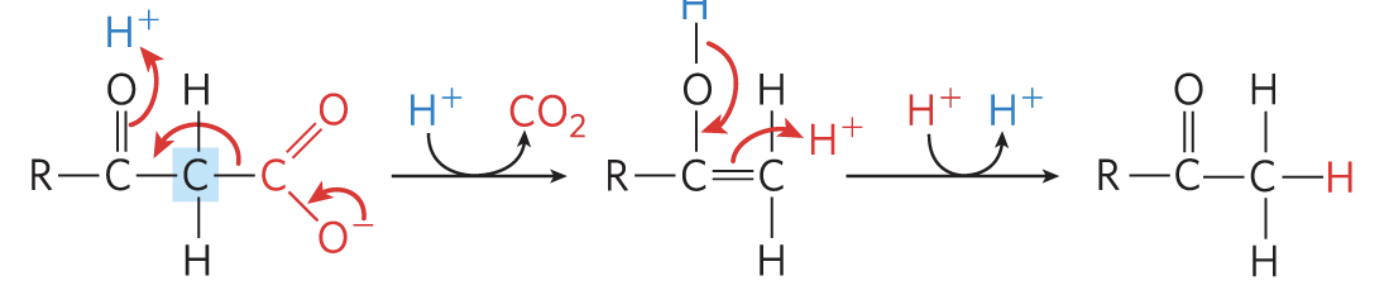
what reaction is this?
decarboxylation
ATP hydrolysis
Dependent on Mg2+
free energy change is large and negative
products of ATP are more stable: they repel each other and Pi is stabilized by resonance delocalization
How does ATP provide energy?
via group transfers
what group in ATP is the leaving group
gamma-phosphate
oxidation is ____ of electrons
reduction is ____ of electrons
loss; gain
if something is a reducing agent it is being ____
oxidized
first source of electrons in glycolysis
reduced carbon
final acceptor in glycolysis
O2
how are all half-reactions listed
as reductions
what E value is reduced v. oxidized
higher E is reduced, lower E is oxidized
E (more positive) - E (more negative)
what does high ΔE mean
redox reaction is favorable
how are e- transferred between biomolecules
directly as electrons
as hydrogen atoms
as hydride ion
through combination with oxygen
HEEP
high energy electron pairs
NAD+, NADP, FMN, FAD
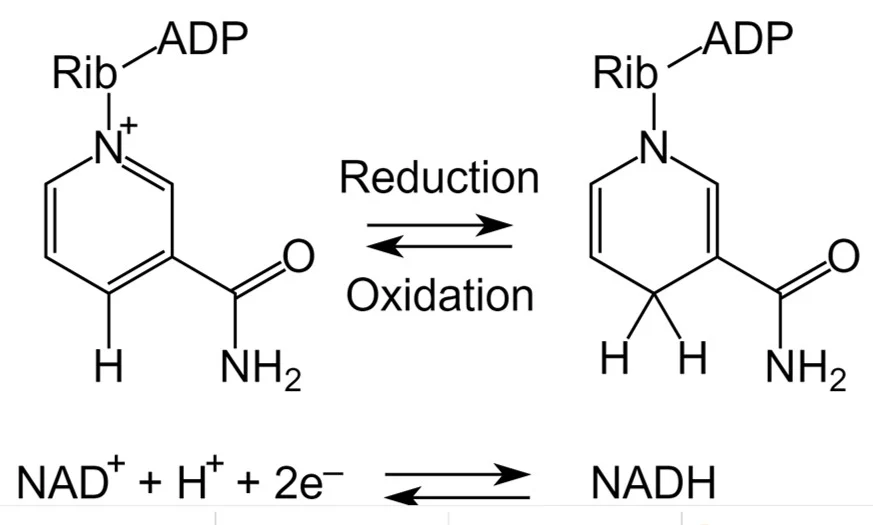
what cofactor is this?
NAD+/NADH, NADP+/NADH
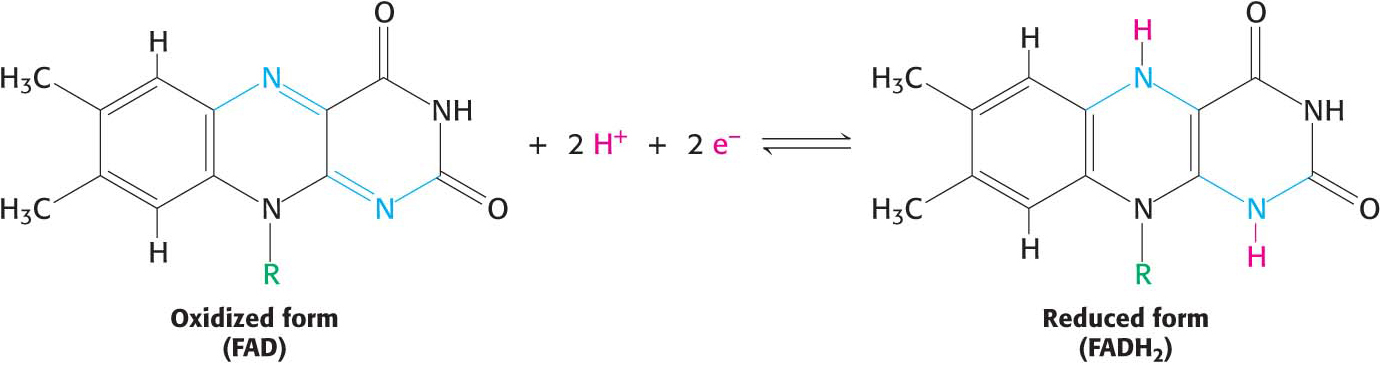
what cofactor is this?
FAD → FADH (vitamin B2)
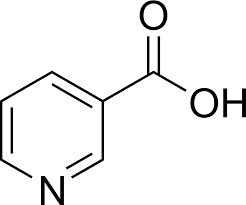
what cofactor is this?
Niacin (vitamin B3)
steps of catabolism
glycolysis
citric acid cycle
oxidative phosphorylation
where does glycolysis occur?
cytoplasm/cytosol
net glycolysis equation
glycolysis + 2 NAD+ + 2 ADP + 2 Pi → 2 pyruvate + 2 NADH + 2 ATP + 2 H+ + 2 H20
glycolysis: step 1 (first priming reaction)
glucose → glucose 6-phosphate
hexokinase
ATP required
adding phosphate to oxygen of C6
irreversible step
glycolysis: step 2
glucose 6-phosphate → fructose 6-phosphate
phosphohexose isomerase
5 carbon ring to 4 carbon ring (carbon 1 becomes substituent of carbon 2)
glycolysis: step 3 (second priming reaction)
fructose 6-phosphate → fructose 1,6-biphosphate
ATP required
phosphofructokinase-1 (PFK-1)
add Pi to oxygen of C1
irreversible step
allosterically inhibited by ATP; activated by AMP/ADP
glycolysis: step 4
fructose 1,6-biphosphate → glyceraldehyde 3-phosphate (G3P)
adolase
cleavage of 6-carbon sugar to 2 3-carbon sugar phosphates (reverse aldol reaction)
glycolysis: step 5
dihydroxyacetone phosphate → glyceraldehyde 3-phosphate
triose phosphate isomerase
right sugar phosphate with ketone needs to be moved around to become aldehyde (2 of the products now)
glycolysis: step 6 (start of payoff phase)
2 glyceraldehyde 3-phosphate + 2 Pi + 2 NAD+→ 1,3-bisphosphoglycerate + 2 NADH + 2 H+
glyceraldehyde 3-phosphatase dehydrogenase: active site of cysteine forms high-energy thioester intermediate
oxidation and phosphorylation
replaces hydrogen (goes to NADH) with oxygen + phosphate at carbonyl carbon
glycolysis: step 7
2 1,3-bisphosphoglycerate + 2 ADP → 3-phosphoglycerate + 2 ATP
phosphoglycerate kinase
substrate-level phosphorylation (removing phosphate from carbonyl oxygen)
glycolysis: step 8
2 3-phosphoglycerate → 2 2-phosphoglycerate
phosphoglycerate mutase
moves phosphate from carbon 3 to carbon 2
glycolysis: step 9
2 2-phosphoglycerate → 2 phosphoenolpyruvate + 2 H2O
enolase
removes OH group from end carbon (dehydration)
glycolysis: step 10
2 phosphoenolpyruvate + 2 ADP → 2 pyruvate + 2 ATP
pyruvate kinase
substrate-level phosphorylation (remove phosphate from oxygen and turn it into double-bonded oxygen)
irreversible step
how is glycolysis favorable?
some of the intermediate steps of glycolysis are unfavorable, but some are very favorable making the net reaction favorable
two fates of pyruvate
fermentation: ethanol or lactic acid
oxidation: acetyl-CoA
what happens to all the HEEPs from glycolysis in fermentation
Le Chat’s Principle: the concentration of NADH is too high which results in it being used to create NAD+
lactic acid fermentation
2 pyruvate + 2 NADH → 2 lactate + 2 NAD+ + 2 H+
lactate dehydrogenase
turns carbonyl into alcohol
seen in active muscle and lactic acid bacteria
why is pyruvate being reduced and not oxidized?
purpose of reaction and no final O2 acceptor in fermentation
ethanol fermentation: step 1
2 pyruvate → 2 acetaldehyde + 2 CO2
pyruvate decarboxylase
removes CO2 and creates aldehyde
TPP as a cofactor
ethanol fermentation: step 2
2 acetaldehyde + 2 NADH + 2 H+→ 2 ethanol + 2 NAD+
alcohol dehydrogenase
reduction reaction
get rid of double bond and give hydrogen to O and carbonyl C
cofactor: Zn2+ (polarized aldehyde oyxgen)
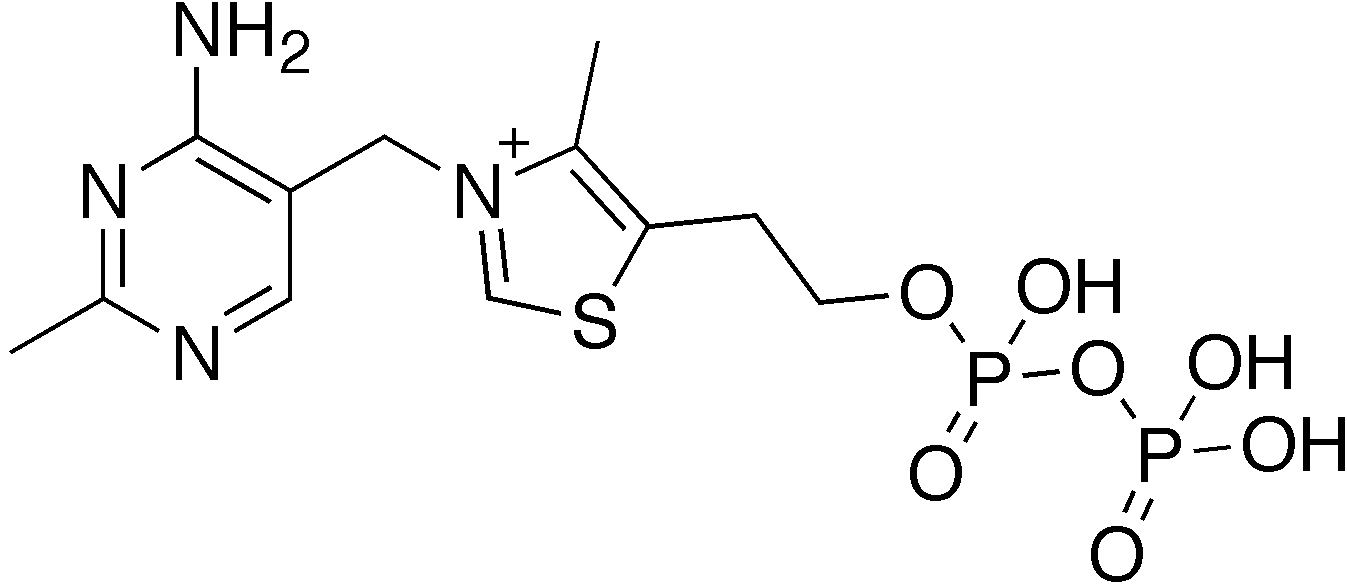
what coenzyme is this
TPP (vitamin B1)
TPP mechanism
stabilizes carbanion intermediates during decarboxylation of pyruvate
3 stages of cellular respiration and where do they occur
Acetyl-CoA production (pyruvate oxidation), matrix
Citric Acid Cycle (acetyl-coA oxidation), matrix
Oxidative Phosphorylation (HEEP carrier oxidation), inner membrane
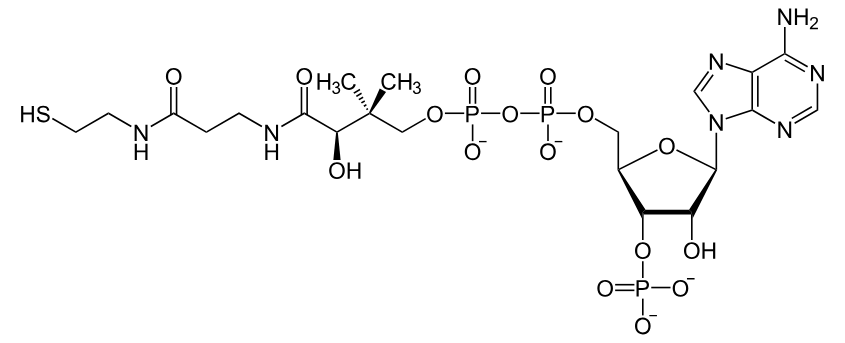
what coenzyme is this?
coenzyme A (vitamin B5)
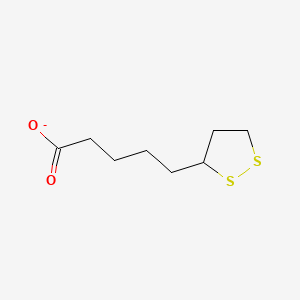
what coenzyme is this?
lipoyllysine (lipoate)
net reaction of acetyl-CoA production
pyruvate + CoA-SH + NAD+ + TPP + lipoate + FAD → acetyl-CoA + NADH + CO2
pyruvate dehydrogenase complex (E1 + E2 + E3)
inhibited by products and ATP, activated by reactants and AMP
pyruvate dehydrogenase complex enzymes
pyruvate dehydrogenase, dihydrolipoyl transacetylase, dihydrolipoyl dehydrogenase
acetyl-CoA production: step 1
pyruvate + TPP → CO2 + hydroxyethyl-TPP (pyruvate decarboxylation)
pyruvate dehydrogenase
TPP attacks carbonyl group and kicks off CO2
acetyl-CoA production: step 2
transfering acetyl group to enzyme 2
TPP is regenerated
hydroxyethyl-TPP + lipoic acid → acetyl lipoyllysine + TPP
acetyl-CoA production: step 3
actually producing Acetyl-CoA
dihydrolipoyl transacetylase (E2)
acetyl lipoyllysine + CoA-SH → acetyl-CoA + reduced lipoyllysine
removing lipoic acid and adding CoA-SH to acetyl group
acetyl-CoA production: steps 4-5
dihydrolipoyl dehydrogenase (E3)
transfer 2 hydrogens from reduced lypoyl groups to FAD
FADH2 gets oxidized by NAD+ turning it into NADH + H+
net reaction of citric acid cycle
acetyl-CoA + 3 NAD+ + FAD + GDP + Pi + 2 H2 O → CoA-SH + 3 NADH + FADH2 + 3 H+ + GTP + 2 CO2
what is the goal and logic of CAC?
oxidize acety-coA as much as possible
combine acetyl-coA with 4 carbon molecule making it easier to oxidize
CAC: step 1
acetyl-CoA + H2O + oxaloacetate→ citrate + CoA-SH
citrate synthase
aldol condensation (methyl group attacks carbonyl on oxaloacetate)
inhibited by NADH, succinyl-CoA, citrate, and ATP; activated by ADP
what product of citric acid cycle inhibits glycolysis and why
citrate inhibits PFK-1 to keep glycolysis and CAC in sync
what is weird about citrate synthase
it needs precise positioning of the active site
CAC: step 2
citrate → H2O + cis-Aconitate → isocitrate
aconitase
dehydration and then rehydration (OH group is respositioned because secondary alcohol is good substrate for oxidation)
CAC: step 3
isocitrate + NAD+ → a-ketoglutarate + NADH + H+
isocitrate dehydrogenase
oxidative decarboxylation (COO- leaves and double on adjacent carbon is formed by oxygen)
NAD (1/3)
irreversible step
inhibited by ATP; activated by Ca2+ and ADP
CAC: step 4
a-ketoglutarate + CoA-SH → succinyl-CoA + CO2
a-ketoglutarate dehydrogenase complex
oxidative decarboxylation (TPP leads to decarboxylation and CoA-SH performs acyl sub)
similar to pyruvate-dehydrogenase
NAD (2/3)
inhibited by succinyl-CoA and NADH; activated by Ca2+
CAC: step 5
succinyl-CoA + GDP + Pi → succinate + GTP + CoA-SH
succinyl-CoA synthetase
substrate-level phosphorylation (thioester is kicked off and that energy goes to GTP)
CAC: step 6
succinate + FAD+ → fumarate + FADH2
succinate dehydrogenase
dehydrogenation (double bound between two CH2s is created kicking off 2 Hs)
CAC: step 7
fumarate + H2O → malate
fumarase
hydration (add water and removed CH-CH double bound)
CAC: step 8
malate → oxaloacetate
malate dehydrogenase
dehydrogenation (remove hydrogen from oxygen and make double bound)
NAD (3/3)
low [oxaloacetate] can help make this reaction more favorable
which CAC intermediates can be used in anabolism?
citrate → fatty acids
a-ketoglutarate, oxaloacetate → amino acids
oxaloacetate → gluconeogenesis
net equation for gluconeogenesis
2 pyruvate + 4 ATP + 2 GTP + 2 NADH + 2 H+ + 6 H2O → glucose + 4 ADP + 2 GDP + 6 Pi + 2 NAD+
anabolic pathway (build molecules, requires ATP)
kidneys and livers perform gluconeogenesis
what enzymes are used in glucogenesis to bypass irreversible steps
glucose 6-phosphatase instead of hexokinase
fructose 1,6-bisphosphatase-1 instead of PFK1
pyruvate carboxylase and PEP carboxylase instead of pyruvate kinase
gluconeogenesis: 1st bypass step 1
bicarbonate + pyruvate + ATP → oxaloacetate + ADP + Pi
cofactor: biotin (activates CO2 to leave and add to pyruvate)
pyruvate carboxylase (attach CO2 to methyl end of pyruvate)
malate-aspartate shuttle (used in step 1 of gluconeogenesis)
oxaloacetate cannot cross membrane to get to cytoplasm
oxaloacetate + NADH → malate + NAD+
malate + NAD+ → oxaloacetate + NADH
oxaloacetate + glutamate → aspartate + a-ketoglutarate
aspartate + a-ketoglutarate → oxaloacetate + glutamate
gluconeogenesis: 1st bypass step 2
oxaloacetate + GTP → phosphoenolpyruvate + GDP + CO2
PEP carboxykinase (remove CO2, make enolate, add phosphate to carbonyl)
gluconeogenesis: bypass 2
fructose 1,6-bisphosphate + H2O → fructose 6-phosphate + Pi
fructose 1,6-bisphosphatase-1 (adds water, removes phosphate)
inhibited by AMP
gluconeogenesis: bypass 3
glucose 6-phosphate + H2O → glucose + Pi
glucose 6-phosphatase (adds water, removes phosphate)
how much more ATP and GTP is used in gluconeogenesis and why
2 of each; because we are reversing glycolysis and trying to overcome lots of irreversible steps
what hormone starts glycolysis
insulin
what hormone starts gluconeogenesis
glucagon
isoenzymatic regulation: difference between hexokinase 1 and hexokinase 4
1 is expressed in most tissues (low Km, inhibited by GCP product)
4 is expressed in liver (high Km, ensures that it only stores glucose when its plentiful)
CAC is an _____ pathway
amphibolic (can be catabolic or anabolic)
how are CAC intermediates constantly replenished
via anaplerotic reactions: pyruvate carboxylase replenishes oxaloacetate
what are the regulated steps of CAC
step 0, 1, 3, 4
what are the two parts of oxidative phosphorylation?
ETC and ATP Synthase
3 important coenzymes in ETC
NADH
FADH2
Ubiquinone (can take 1 or 2 e-)
2 metalloproteins in ETC
cytochromes: carry heme prosthetic groups
Fe-S: coordinated by cysteine residues
both are 1e- acceptor/donorsn
net ETC equation
2NADH + 22H+ + O2 → 2NAD+ + 20H+ + 2H2O
ETC: step 1
NADH donates 2 electrons to complex 1 (NADH dehydrogenase)
electrons go to Q → QH2 (reduced) (go to Fe-S cluster before)
4 H+ pumped out
ETC: step 2
complex 2 (succinate dehydrogenase), also part of CAC
FADH2 donates 2 electrons to Q → QH2 (electrons go to Fe-S cluster before)
FAD gets its electrons from succinate → fumarate in CAC
no protons pumped
ETC: step 3
complex 3 (medley of iron centers)
QH2 transfers its electrons to complex 3
e- move to cyt-c (1 at a time)
4 H+ are pumped out
ETC: step 4
e- are transferred to complex 4
O2 is the final electron acceptor
2 H+ are pumped out
what is the chemiosmotic theory
ETC creates proton gradient (more H+ outside)
accumulation of H+ outside creates an electrochemical gradient which stores energy as proton motive force
protons flow back into matrix via ATP synthase
ATP synthase rotates leading to creation of ATP
ATP synthase structure
F1: in the matrix, catalyzes ATP synthesis in beta subunit
F0: transports protons from intermembrane space to matrix, c ring spins, energy transferred to F1 to catalyze ADP phosphorylation
c ring spins (bc of protons) and wacks beta unit up → changes shape of beta → this force stimulates ATP synthesis
3 conformations of beta subunit of F1
B-ADP = binds ADP + Pi loosely
B-ATP = binds ATP tightly
B-empty = loose-binding
how many ATP from 1 spin of ATP synthase
3 ATP
how much ATP per NADH and FADH2
2.5 ATP
1.5 ATP
total ATP yield
7 ATP (glycolysis) + 5 ATP (pyruvate oxidation) + 20 ATP (citric acid) = 32 ATP
why are fats great for long-term energy storage
lots of reduced carbons in CH2 (oxidative potential)
pack molecules tightly
very stable
long term storage
slow delivery
fatty acid breakdown steps
make fatty acid-CoA
transport from cytosol to mitochondria
beta oxidation (break down 2C’s at a time)
fatty acid breakdown: step 1
activate FA by attaching CoA
consumes 2 ATP for activation
dehydrogenation → hydration → dehydrogenation → thiolysis
fatty acid breakdown: step 3
beta-oxidation
similar to citric acid cycle step
dehydrogenation with FAD (lose 2 H+)
hydration
dehydrogenation with NAD+
1 round of b-oxidation makes how many acetyl-CoA, NADH, and FADH2
1 acetyl-CoA, 1 NADH, 1 FADH2
for N carbon fatty acid → N/2 acetyl CoA, (N/2) - 1 NADH, FADH2