2003: Glycolysis, Gluconeogenesis, Glycogen
1/70
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
71 Terms
Glucose structure
Glucose can exist in a linear form, but at physiological pH (around pH 7), it typically forms a cyclic structure.
Stage 1 vs Stage 2 of glycolysis
Stage 1: Trapping and Preparation Phase: This initial stage involves investing energy (steps 1 and 3) to prepare the glucose molecule for cleavage (ends in step 4).
Stage 2: Payoff Phase: This stage focuses on extracting energy from the prepared 3-carbon molecules (ATP from steps 7 and 10). It occurs twice because 2 molecules of GAP are formed at the end of stage 1, generating 4 ATP
Step 1 of glycolysis: reactant and product, enzyme, requirements, reversibility
Phosphorylation of glucose: Glucose is converted into glucose 6-phosphate.
Enzyme: This reaction is catalyzed by the enzyme hexokinase.
Cation Requirement: All kinases require a divalent cation for activity; specifically, kinases involved in glycolysis, like hexokinase, require Mg2+.
ATP to ADP: An ATP molecule donates its terminal phosphate group, becoming ADP, to form glucose 6-phosphate.
Irreversible Reaction: The phosphorylation of glucose traps the molecule in the cell. Glucose can leave a cell via glucose transporters, while glucose-6-phosphate cannot
Step 2 of glycolysis: reactant and product, enzyme, reversibility
Isomerization of Glucose 6-phosphate: Glucose 6-phosphate, which is an aldose, is converted to fructose 6-phosphate, which is a ketose.
Enzyme: This transformation is carried out by phosphoglucose isomerase.
Reason for Isomerization: A fructose-based sugar (a ketose) can be cleaved into two 3-carbon molecules more easily than a glucose-based sugar (an aldose).
Step 3 of glycolysis: reactant and product, enzyme, reversibility
Phosphorylation of Fructose 6-phosphate: Fructose 6-phosphate is converted into Fructose 1,6-bisphosphate.
Enzyme: This reaction is catalyzed by phosphofructokinase-1 (PFK-1). PFK-1 is a key regulatory enzyme in glycolysis, controlling the overall rate of the pathway.
Cation Requirement: This enzyme also requires Mg2+ for its activity.
ATP to ADP: Similar to step 1, another ATP molecule donates a phosphate, becoming ADP.
Irreversible Reaction: This is the second of three irreversible reactions in glycolysis, making it another committed step in the pathway.
Step 4 of glycolysis: reactant and product, enzyme, reversibility
Cleavage of Fructose 1,6-bisphosphate: Fructose 1,6-bisphosphate is cleaved into two distinct 3-carbon molecules: glyceraldehyde-3-phosphate (GAP) and dihydroxyacetone phosphate (DHAP).
Enzyme: This cleavage is performed by the enzyme aldolase, which carries out an aldol cleavage reaction.
Reversible Reaction: This reaction is reversible.
Step 5 of glycolysis: reactant and product, enzyme, reversibility
Isomerization of DHAP into GAP: Dihydroxyacetone phosphate (DHAP) is converted into glyceraldehyde-3-phosphate (GAP).
Enzyme: This isomerization is carried out by triose phosphate isomerase.
Driven Forward: Although this isomerization is reversible, it is driven forward because GAP is rapidly consumed in Stage 2 of glycolysis. As GAP is used up, more DHAP is converted to GAP to maintain equilibrium, ensuring that both 3-carbon molecules eventually proceed through the payoff phase.
Step 6 of glycolysis: reactant and product, enzyme, reversibility
Oxidation of GAP and Reduction of NAD+: Glyceraldehyde-3-phosphate (GAP) is converted to 1,3-bisphosphoglycerate (1,3-BPG).
Enzyme: This reaction is catalyzed by GAP dehydrogenase, which is a redox enzyme.
NAD+ Reduction: In this oxidation-reduction reaction, GAP is oxidized by an inorganic phosphate while NAD+ is reduced to NADH. This NADH carries electrons that will later be used to generate more ATP in oxidative phosphorylation.
High Phosphoryl Donor Potential: The 1,3-BPG molecule formed has a higher phosphoryl donor potential than ATP. This means it can readily donate a phosphate group to ADP, a key feature for the next step.
Step 7 of glycolysis: reactant and product, enzyme, reversibility
ATP Synthesis using 1,3-BPG as a Phosphoryl Donor:1,3-bisphosphoglycerate is converted to 3-phosphoglycerate.
Enzyme: This reaction is carried out by phosphoglycerate kinase.
Cation Requirement: This enzyme also requires Mg2+ for its activity.
ATP Payoff: This is the first of two "payoff" reactions in glycolysis that directly yield ATP through substrate-level phosphorylation.
Phosphate Transfer: A phosphate group is transferred from 1,3-bisphosphoglycerate to ADP, forming ATP.
Step 8 of glycolysis: reactant and product, enzyme, requirements, reversibility
Rearrangement of 3-phosphoglycerate into 2-phosphoglycerate: 3-phosphoglycerate is converted to 2-phosphoglycerate (phosphate group is simply moved to a different position).
Enzyme: This reaction is catalyzed by phosphoglycerate mutase.
Cation Requirement: This enzyme requires Mg2+
Step 9 of glycolysis: reactant and product, enzyme, requirements, reversibility
Dehydration of 2-phosphoglycerate to Yield PEP: 2-phosphoglycerate is converted to phosphoenolpyruvate (PEP). A molecule of water is removed, creating a high-energy phosphate bond.
Enzyme: This reaction is carried out by enolase.
High-Energy Donor: PEP is a high-energy phosphoryl donor, even more so than ATP, setting up the final ATP-generating step.
Step 10 of glycolysis: reactant and product, enzyme, reversibility
TP Synthesis using PEP as a Phosphoryl Donor: Phosphoenolpyruvate (PEP) is converted to pyruvate.
Enzyme: This reaction is carried out by pyruvate kinase.
Cation Requirement: This enzyme requires both K+ and Mg2+
Irreversible Reaction: This is the third of three irreversible reactions of glycolysis, making it another key regulatory point.
ATP Payoff: This is the second of two "payoff" reactions that yield ATP.
Step 10 of glycolysis: Non-enzymatic Tautomerization of Pyruvate
After the enzymatic reaction, pyruvate undergoes a spontaneous non-enzymatic tautomerization.
Enol to Keto Form: The enol form of pyruvate, initially formed, is converted to its keto form.
Stability: At physiological pH 7, the keto form of pyruvate is the more stable and predominant form.
Kinases
Kinases are a class of enzymes that add a phosphoryl group (usually from ATP) to an acceptor molecule.
Aldoses vs Ketoses
Aldoses have an aldehyde group (-CHO) at the end of the carbon chain (specifically at C1), like glucose.
Ketoses have a ketone group (C=O) within the carbon chain (typically at C2), like fructose.
Fates of pyruvate: Production of ethanol (anaerobic)
Pyruvate is converted to acetaldehyde. - This reaction is catalyzed by the enzyme pyruvate decarboxylase. - CO2 is formed in this step. This CO2 provides the carbonation in beer and leavened bread, while the subsequent product leads to alcohol in beverages.
Acetaldehyde is converted to ethanol. - This reaction is catalyzed by the enzyme alcohol dehydrogenase. - Crucially, NAD+ is restored from NADH, allowing glycolysis to continue.
Fates of pyruvate: Production of lactate (anaerobic)
Pyruvate is directly converted to lactate. - This reaction is catalyzed by the enzyme lactate dehydrogenase (LDH). - Similar to alcoholic fermentation, the regeneration of NAD+ allows glycolysis to continue.
Fates of pyruvate: Production of Acetyl CoA
Under aerobic conditions (when oxygen is present), pyruvate is further oxidized to acetyl CoA, which then enters the citric acid cycle for much greater ATP production.
What are the practical implications of anaerobic glycolysis?
Ethanol Synthesis
Used in the production of beer and wine through fermentation.
Essential for leavened bread, where the CO2 produced causes the dough to rise.
Lactate Synthesis
Associated with muscle fatigue during strenuous exercise when oxygen supply cannot meet demand.
A key indicator in medical conditions like myocardial infarction (heart attack).
Central to the Warburg Effect observed in cancer cells.
What is Lactate Dehydrogenase (LDH) and its forms?
LDH is an enzyme with five isoforms, each composed of different combinations of two subunits: H (heart) and M (muscle).
Isoforms: LDH1(HHHH), LDH2(HHHM), LDH3(HHMM), LDH4(HMMM), and LDH5(MMMM).
The H and M subunits are produced by different genes.
What is the LDH Tissue Specificity (dominant LDH isoform) in the liver and muscle compared to the heart and kidney
Liver and muscle: Primarily LDH4/LDH5, which favors the conversion of pyruvate to lactate. This is common in tissues that frequently perform anaerobic glycolysis.
Heart and kidney: Primarily LDH1, which favors the conversion of lactate to pyruvate. These tissues are highly aerobic and can convert lactate back to pyruvate for energy production via oxidative phosphorylation.
How does LDH relate to a Myocardial Infarction (Heart Attack), and how is this detected?
Cardiac muscle is rich in LDH₁ (H₄)
When a myocardial infarction (heart attack) occurs:
Heart cells are damaged
LDH₁ leaks into the bloodstream
This causes a rise in LDH₁ relative to LDH₂ in serum
This change can be detected by separating serum LDH isoforms using electrophoresis, providing a diagnostic marker for heart damage.
What is the Warburg Effect?
Cancer cells carry out anaerobic glycolysis to yield lactate, even if O2 levels are in abundance
Glycolysis occurs at a faster rate in cancer cells versus non-cancer cells
How doctors take advantage of the Warburg Effect?
By using 2-18F-fluoro-D-glucose (FDG) a chemically modified version of glucose that gets trapped in cancer cells
Cancer cells uptake FDG more rapidly than non-cancer cells due to their increased glucose metabolism.
FDG can be phosphorylated by hexokinase (the first enzyme in glycolysis).
However, 6-phospho-FDG cannot be metabolized further down the glycolysis pathway. It gets "trapped" in the cancer cells.
This helps show doctors where glucose is being overused right now, indicating a tumor
This accumulation of radioactive FDG results in a high signal in cancer cells, making them visible on the scan. A high signal is also detected in the bladder because excess FDG is secreted, which can sometimes be mistaken for a tumor.
Where are the primary sites of regulation in glycolysis?
The three irreversible reactions (Steps 1, 3, and 10)
How does Phosphofructokinase (PFK-1) regulate glycolysis?
PFK-1 regulates glycolysis by sensing cellular energy status.
High ATP allosterically inhibits PFK-1, reducing fructose-1,6-bisphosphate formation,
high ADP or AMP relieves ATP inhibition and activates glycolysis.
In the liver, PFK-1 is also inhibited by citrate and strongly activated by fructose-2,6-bisphosphate produced by PFK-2.
How does hexokinase regulate glycolysis?
Hexokinase is regulated by feedback inhibition from glucose-6-phosphate.
When PFK-1 is inhibited, fructose-6-phosphate accumulates and is converted back to glucose-6-phosphate, which indirectly inhibits hexokinase and prevents further glucose phosphorylation.
How does Pyruvate Kinase regulate glycolysis?
Pyruvate kinase is allosterically inhibited by ATP, signaling high energy levels. It is activated by fructose-1,6-bisphosphate through feed-forward regulation, ensuring glycolysis proceeds to completion once committed.
Where is Glucose transporter 2 (GLUT2) located, and what is its role in these areas?
GLUT2 is found primarily in the liver, but also in pancreatic islet cells, the intestine, and kidneys
In liver and kidney: removes excess glucose from blood (bidirectional glucose transport - in and out of cells depending on blood glucose levels).
In pancreas: regulates insulin release
In intestine: allows glucose to enter blood
What is the Kt value of GLUT2 and what does this mean?
GLUT2 has a very high Kt value of 17 mM, thus it is usually never saturated (as blood glucose is usually ~5 mM fasted and ~11 mM following a meal)
Where is Glucose transporter 4 (GLUT4) located, and what is its role in these areas?
GLUT4 is found in muscle, adipose, and the heart
Its job is to move glucose from the blood into cells
What is GLUT4 sensitive to and how does this determine how much glucose GLUT4 takes up?
They are insulin-sensitive, meaning insulin controls how much glucose they take up.
Insulin acts as the signal for glucose uptake. - The presence of GLUT4 on cell surfaces is increased in the fed state (after eating, when insulin levels are high) and decreases in the fasted state (when insulin levels are low).
How does Insulin provide the "switch" dictating whether carbohydrates or fats are used as fuel.
In the fed state (high plasma insulin):
Insulin stimulates glucose uptake in tissues by activating glucose transporters (like GLUT4).
Insulin inhibits intracellular lipolysis (the breakdown of stored fats into fatty acids), promoting glucose use for energy.
In the fasted state (low plasma insulin):
There is no GLUT stimulation (meaning less glucose uptake by insulin-sensitive tissues).
There is increased lipolysis, releasing fatty acids from storage for energy.
GLUT4 Glucose Transporter Cycle
Storage (low insulin):
GLUT4GLUT4 transporters are stored inside the cell within intracellular vesicles.
Very few GLUT4GLUT4 proteins are present on the plasma membrane.
Insulin Binding:
Insulin binds to the insulin receptor on the cell surface.
Vesicle Translocation:
Insulin signaling triggers a cascade that causes the GLUT4GLUT4-containing vesicles to move toward the plasma membrane.
Membrane Fusion:
These vesicles fuse with the plasma membrane.
GLUT4GLUT4 transporters are then inserted into the membrane.
Glucose Uptake:
The increased number of GLUT4GLUT4 transporters on the cell surface allows for rapid glucose uptake into the cell.
Insulin Levels Drop:
When insulin levels decrease (e.g., as blood glucose returns to normal), GLUT4GLUT4 is removed from the membrane by endocytosis.
Recycling:
The GLUT4GLUT4-containing vesicles fuse with endosomes.
Ready for Reuse:
GLUT4GLUT4 is then repackaged into storage vesicles, ready to return to the membrane when insulin levels rise again.
Where and What is Gluconeogenesis?
Gluconeogenesis is the metabolic process that converts non-carbohydrate precursors into glucose.
In mammals, there are two primary sites for gluconeogenesis:
The liver (the predominant site).
The kidney cortex.
2 functions of gluconeogenesis
1. A source of glucose independent of diet - Starvation
2. Cori Cycle – conversion of lactate to glucose during or after heavy exercise.
Why is it important for the brain and red blood cells to maintain glucose levels?
The end products of many biochemical pathways are salvaged and used to synthesize glucose in gluconeogenesis.
Brain depends on glucose as its primary fuel and red blood cells use glucose as their only fuel.
Are glycolysis and gluconeogenesis just reverse processes?
Glycolysis and gluconeogenesis share some enzymes, but they are not simply the reverse of each other.
The highly exergonic, irreversible steps of glycolysis are bypassed in gluconeogenesis by different reactions. These bypass reactions ensure that gluconeogenesis is also exergonic (favorable) under cellular conditions.
What are the 3 major non-carbohydrate precursors of glucose that are converted into pyruvate or later intermediates for glycolysis
Lactate: Skeletal muscle produces lactate through lactic acid fermentation during intense exercise. The liver can convert this lactate back into pyruvate using lactate dehydrogenase.
Amino acids: Derived from dietary proteins or, during starvation, from the breakdown of proteins in skeletal muscle. Certain amino acids can be converted to pyruvate or other gluconeogenic intermediates.
Glycerol: Produced from the hydrolysis of triacylglycerols (fats). Glycerol can enter the gluconeogenic or glycolytic pathway at the level of dihydroxyacetone phosphate (DHAP).
What’s the first bypass of gluconeogenesis? (location, requirements, enzyme)
Carboxylation of pyruvate to form oxaloacetate:
a. This step requires energy, costing one molecule of ATP.
b. Pyruvate takes a CO2 molecule from bicarbonate
c. It requires biotin, a coenzyme used in CO2 transfer and carboxylation reactions. It is required pyruvate carboxylase (PC), which catalyzes this reaction.
d. The reaction is catalyzed by pyruvate carboxylase.
e. This reaction occurs exclusively in the mitochondria.
f. Oxaloacetate is converted into phosphoenolpyruvate
How is Oxaloacetate converted into phosphoenolpyruvate in the first bypass of gluconeogenesis?
a. Oxaloacetate must be transported from the mitochondria to the cytoplasm to complete PEP synthesis.
b. Oxaloacetate is first reduced to malate by malate dehydrogenase inside the mitochondria.
c. Malate is then transported across the mitochondrial membrane and reoxidized back to oxaloacetate in the cytoplasm by a cytoplasmic NAD+-linked malate dehydrogenase.
d. This formation of oxaloacetate from malate in the cytoplasm also provides NADH for use in subsequent steps of gluconeogenesis.
e. Finally, oxaloacetate is simultaneously decarboxylated and phosphorylated by phosphoenolpyruvate carboxykinase (PEPCK) to generate phosphoenolpyruvate (PEP).
f. The phosphoryl donor for this step is GTP.
g. The CO2 that was added to pyruvate by pyruvate carboxylase is released in this step.
What’s the second bypass of gluconeogenesis?
Conversion of Fructose 1,6-Bisphosphate into Fructose 6-Phosphate and Orthophosphate (Pi)
The enzyme responsible for this step is fructose 1,6-bisphosphatase (FBPase).
a. FBPase is an allosteric enzyme and serves as the primary regulatory point of gluconeogenesis.
b. It is an example of a phosphatase, an enzyme that catalyzes the hydrolysis of a phosphate ester to form inorganic phosphate.
What’s the third bypass of gluconeogenesis?
The Generation of Free Glucose Is an Important Control Point
The fructose 6-phosphate generated by fructose 1,6-bisphosphatase is readily converted into glucose 6-phosphate.
Glucose 6-phosphate is transported into the lumen of the endoplasmic reticulum of liver cells (only liver) by a transporter called T1. Here, it is hydrolyzed to free glucose by glucose 6-phosphatase. This enzyme is found only in the liver.
A pair of transporters, T2 and T3, then shuttle glucose and Pi back to the cytoplasm, from where glucose can be released into the bloodstream.
What happens to gluconeogenesis in most other tissues?
In most other tissues capable of gluconeogenesis (like the kidney cortex), the process ends with the formation of glucose 6-phosphate.
The fate of glucose 6-phosphate in these tissues is commonly conversion into glycogen for storage or use in the biosynthesis of other molecules, such as nucleotides.
What is the energy cost of gluconeogenesis? Does this make the reaction favorable or unfavorable?
The formation of glucose from pyruvate is energetically unfavorable unless it is coupled to favorable reactions.
Six nucleoside triphosphate molecules are hydrolyzed to synthesize glucose from pyruvate in gluconeogenesis, whereas only two molecules of ATP are generated in glycolysis.
How are glycolysis and gluconeogenesis regulated together?
These two crucial pathways are reciprocally regulated to prevent a futile cycle where both pathways operate simultaneously at high rates, wasting energy.
When glucose is abundant, glycolysis predominates.
When glucose is scarce, gluconeogenesis predominates.
The cell ensures that one pathway is relatively inactive while the other is highly active.
How are glycolysis versus gluconeogenesis regulated by metabolite concentrations?
Glycolysis
The rate of glycolysis is regulated by glucose concentration.
High glucose levels lead to increased glycolytic flux (rate).
Low glucose levels lead to decreased glycolysis.
Gluconeogenesis
The rate of gluconeogenesis is regulated by the availability of precursors.
High levels of precursors (e.g., lactate, alanine, glycerol) lead to increased gluconeogenesis.
How are glycolysis versus gluconeogenesis regulated by energy (ATP/ADP/AMP) changes?
Low ATP / high AMP signals an energy deficit, thus glycolysis is favored to produce ATP.
High ATP signals energy abundance, thus gluconeogenesis is favored to synthesize and store glucose.
What are the key sites of reciprocal regulation between glycolysis and gluconeogenesis?
The interconversion of fructose 1,6-bisphosphate and fructose 6-phosphate is a key regulatory site.
Additionally, glycolysis and gluconeogenesis are reciprocally regulated at the interconversion of phosphoenolpyruvate and pyruvate, in the liver.
Describe the liver’s sensitivity to blood glucose levels in terms of hormones released, and what’s an exception to this?
High blood glucose: Insulin is secreted, which stimulates glycolysis and normally inhibits gluconeogenesis.
Low blood glucose: Glucagon is secreted, which stimulates gluconeogenesis and inhibits glycolysis.
In type 2 diabetes, insulin fails to act effectively, a condition known as insulin resistance, leading to imbalances in these pathways.
What is the Cori cycle?
Muscle and liver display inter-organ cooperation in a series of reactions called the Cori cycle.
During intense muscle contraction, muscle produces lactate through anaerobic glycolysis and releases it into the blood.
The liver then removes this lactate from the blood and converts it back into glucose through gluconeogenesis.
This newly synthesized glucose can then be released back into the blood to be used by other tissues, including the muscle itself.
Structure of Glycogen
Glycogen is the major animal storage polysaccharide.
It is a polymer of glucose residues linked by α(1→4) glycosidic bonds.
It has branches formed by α(1→6) glycosidic bonds, occurring approximately every 8-15 glucose residues.
Glycogen is also linked to a protein called glycogenin, which is found right in the center of the glycogen molecule and serves as a primer for its synthesis.
How does the regulation of glycogen synthesis and degradation differs between the liver and muscles?
Liver: Regulates glycogen metabolism to maintain the blood-glucose concentration required by the entire organism, slowly and steadily releasing glucose during a nocturnal fast to maintain brain function. The liver is "altruistic."
Muscles: Regulate these processes to meet the energy needs of the muscle itself. Glycogen breakdown in muscles fuels the ATP needs for muscle contraction. Muscle is "selfish."
What Glycogen phosphorylase and its functions?
Glycogen phosphorylase: Degrades glycogen from the non-reducing ends of the glycogen molecule.
Glycogen phosphorylase catalyzes a phosphorolysis reaction (cleavage by adding a phosphate group) that yields glucose 1-phosphate.
Why are additional debranching enzymes needed for glycogen breakdown? What are their names and functions?
Glycogen phosphorylase cannot cleave near branch points (it only cleaves α(1→4) glycosidic bonds).
A transferase enzyme shifts a small oligosaccharide (a few glucose units) near the branch point to a nearby chain, making those glucose units accessible to glycogen phosphorylase.
A debranching enzyme (also called α-1,6-glucosidase) then cleaves the α(1→6) bond at the branch point, releasing a free glucose molecule.
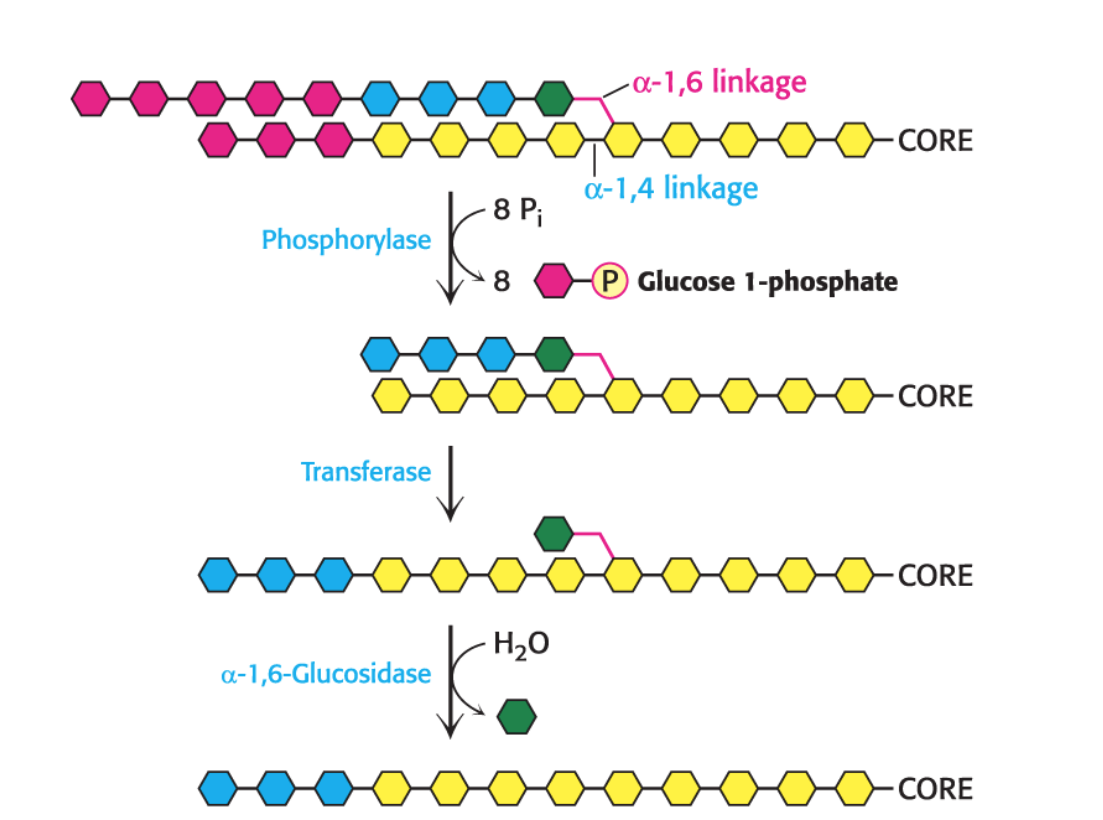
How is glucose-1-phosphate converted to glucose-6-phosphate to be used in glycolysis and other tissues to finally make glucose?
Glucose 1-phosphate (from glycogen breakdown) is converted into glucose 6-phosphate by phosphoglucomutase.
Liver: Contains glucose 6-phosphatase, a hydrolytic enzyme that generates free glucose from glucose 6-phosphate. This free glucose is then released into the blood for use by other tissues (like the brain and red blood cells).
Most other tissues (including muscle): Lack glucose 6-phosphatase. Therefore, they cannot release free glucose into the blood. The glucose 6-phosphate produced from glycogen breakdown in muscle is used directly within the muscle cell for its own energy needs (e.g., in glycolysis).
What are the two hormones that control glycogen breakdown in the liver versus the muscles?
Glucagon and epinephrine: These hormones signal the need for glycogen breakdown.
Muscular activity → The muscle: Leads to the release of epinephrine. This stimulates glycogen breakdown in muscle (for immediate energy) and, to a lesser extent, in the liver.
Low blood glucose → The liver: more responsive to glucagon, which is a signal of low blood glucose (the starved state). Glucagon primarily acts on the liver to release glucose into the bloodstream.
What is the biological insight relating glycogen depletion with the onset of fatigue?
The onset of fatigue coincides with the depletion of glycogen reserves.
• However, it is not clear whether glycogen depletion is an actual cause of fatigue.
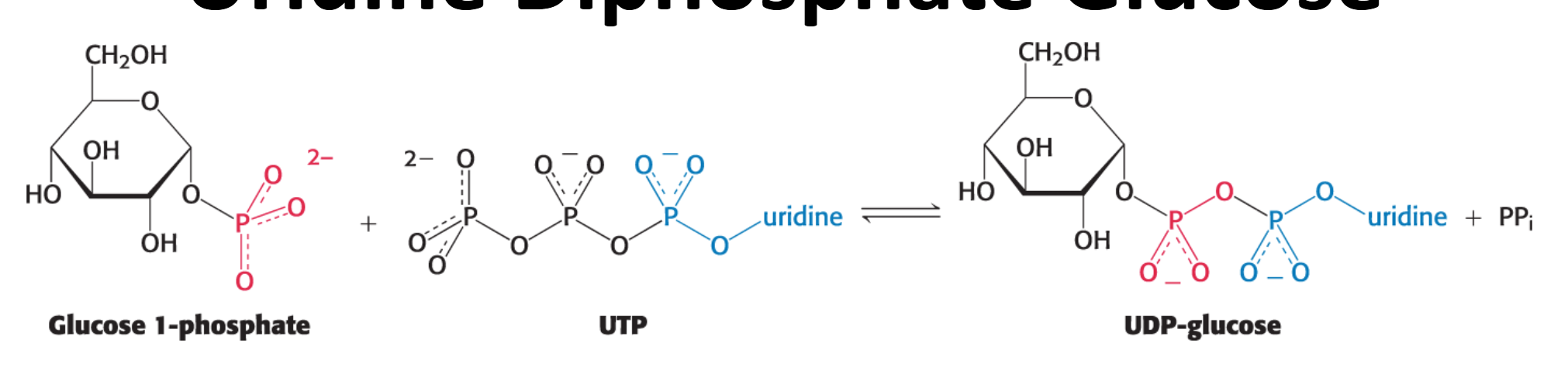
What is the activated monomer used to extend the glycogen chain during glycogen synthesis?
UDP-glucose: synthesized from glucose 1-phosphate and uridine triphosphate (UTP)
How is UDP-glucose synthesized?
It is catalyzed by UDP-glucose pyrophosphorylase
This reaction liberates the outer two phosphoryl residues of UTP as pyrophosphate (PPi) and attaches the remaining phosphate and uridine to glucose 1-phosphate.
This reaction is readily reversible, but the subsequent hydrolysis of PPi to two Pi molecules makes the overall process irreversible and drives it forward.
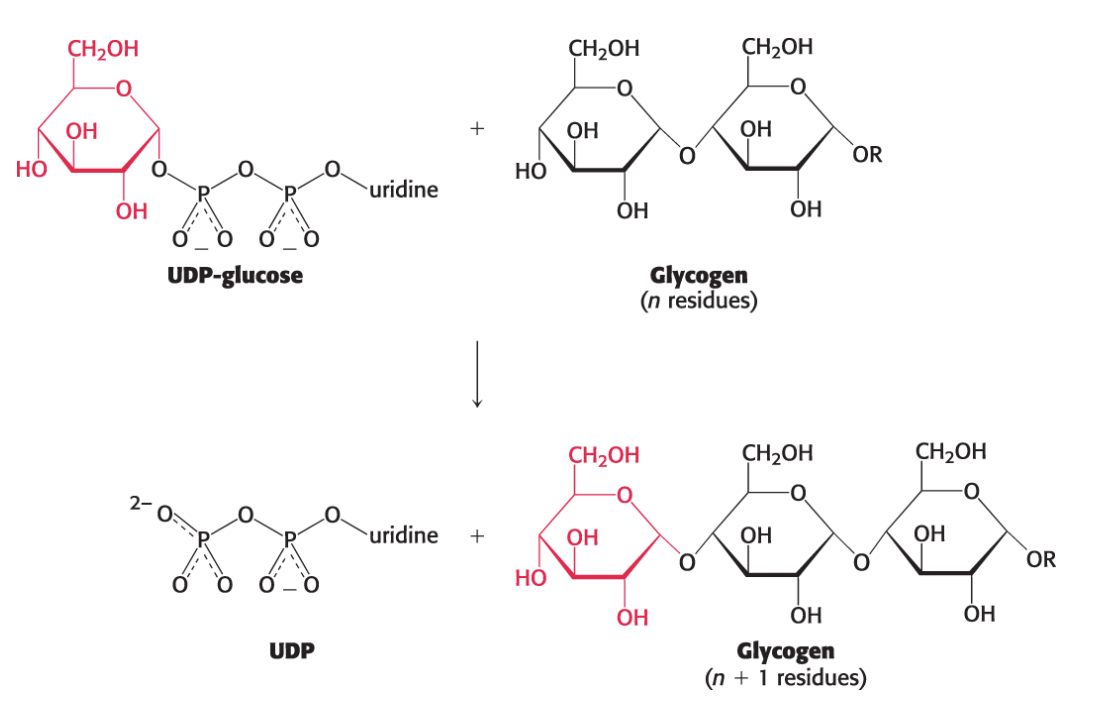
How is glycogen synthesized from UDP-glucose? What catalyzes this reaction?
New glucosyl units (from the UDP-glucose) are added to the nonreducing terminal residues of glycogen.
The activated glucosyl unit from UDP-glucose is transferred to the hydroxyl group at C-4 of a terminal residue in an existing glycogen chain, forming an α−1,4-glycosidic linkage.
UDP is displaced by the terminal hydroxyl group of the growing glycogen molecule.
This reaction is catalyzed by glycogen synthase
How many isozymic forms of glycogen synthase do humans have? Where are they expressed?
Humans have two isozymic forms of glycogen synthase:
one expressed in the liver
another in muscle and other tissues.
Glycogen synthase can add glucose units to an existing polysaccharide chain containing how many units? What does this mean glycogen requires and how does it work?
Glycogen synthase can add glucosyl residues only to a polysaccharide chain already containing more than four residues.
Therefore, glycogen synthesis requires a primer, which is provided by glycogenin.
Each subunit of glycogenin can catalyze the formation of short 1,4-glucose polymers (10 to 20 glucosyl units long) on its partner subunit, providing the initial primer.
If glycogen synthase only synthesizes α−1,4 linkages, how can α−1,6 linkages be created that branch the glycogen polymer. Give 3 reasons why branching is important
Another enzyme is required to form the α-1,6 linkages that make glycogen a branched polymer.
Branching is important because it increases the solubility of glycogen.
Branching creates a large number of terminal residues which are the sites of action of glycogen phosphorylase (for degradation) and synthase (for synthesis).
Branching increases the rate of glycogen synthesis and degradation.
What does the glycogen branching enzyme (α-1,6 branching enzyme) do?
It creates α-1,6 branch points in glycogen by breaking an α-1,4 bond
It transfers a block of ~7 glucose residues from the nonreducing end of a chain (which was at least 11 residues long) to an interior site.
It reattaches them with an α-1,6 linkage at least 4 residues away from an existing branch and allowing further extension at multiple nonreducing ends.
What is a powerful activator of the enzyme glycogen synthase. What are the two forms of glycogen synthase?
Glucose 6-phosphate is a powerful allosteric activator of glycogen synthase, especially the 'b' form, making it active even when phosphorylated.
Glycogen synthase exists in two forms:
An active, nonphosphorylated 'a' form.
A usually inactive, phosphorylated 'b' form
How is Glycogen Is an Efficient Storage Form of Glucose?
Only two molecules of ATP are required to incorporate dietary glucose into glycogen.
The complete oxidation of glucose derived from glycogen (through glycolysis and oxidative phosphorylation) yields 31 molecules of ATP. This highlights the efficiency of glycogen as an energy storage form.
How are Glycogen Breakdown and Synthesis reciprocally regulated?
Glycogen synthesis is inhibited by the same glucagon and epinephrine signaling pathways that stimulate glycogen breakdown.
How does Insulin Stimulates Glycogen Synthesis?
Insulin stimulates glycogen synthesis by activating a signal transduction pathway.
Insulin also facilitates glycogen synthesis by increasing the number of glucose transporters (GLUT4).
How does Glycogen Metabolism in the Liver Regulates Blood-Glucose Concentration? Which enzymes are involved?
High blood glucose leads to inhibition of glycogen degradation (glycogen phosphorylase) in the liver and increases glycogen synthesis (glycogen synthase). This helps bring elevated blood glucose levels down.
Conversely, low blood glucose would trigger the opposite response, breaking down liver glycogen to release glucose.
Clinical insight: Explain Diabetes Mellitus (the two types of diabetes)
Diabetes is characterized by the presence of excess glucose and underutilization of the fuel.
• Excess glucose is excreted in the urine.
• In type 1 diabetes, insulin in not produced.
• In type 2 diabetes, insulin is produced but the insulin signaling pathway is not responsive, a condition referred to as insulin resistance