Cellular Respiration
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
parts of a nucleotide
nitrogenous base
pentose/five-carbon sugar
one or more phosphate groups
properties of ATP which make it suitable for energy currency of the cell
soluble in water, can move freely through the cytoplasm and other aqueous solutions in the cell
stable at pH levels close to neutral as in cytoplasm
cannot pass freely through phospholipid bilayer of membranes, allowing movement between membrane-bound organelles to be controlled
third phosphate group of ATP can be easily removed and reattached by hydrolysis and condensation reactions
hydrolyzing ATP to ADP and phosphate releases a relatively small amount of energy
ATP hydrolysis and condensation reactions
ATP + H2O <—> ADP + phosphate + energy
hydrolyzing ATP to ADP and phosphate releases a relatively small amount of energy
enough energy for many processes within the cell
not an excess which would be wasted by conversion to heat
life processes within cells that ATP supplies with eenergy
synthesizing macromolecules
active transport
movement
role of ATP in synthesizing macromolecules
anabolic reactions that link monomers together into large polymers would be endothermic and therefore unlikely to happen without coupling them to conversion of ATP to ADP (e.g. synthesis of DNA during replication)
role of ATP in active transport
ATP energy used to cause reversible changes in conformation of pump protein, allowing particle to enter pump protein from one side of the membrane
when the pump is in the other conformation, the particle can exit on the other side of the membrane
one of the two shapes is more stable than the other
ATP used to cause change from more stable to less stable conformation, change back to stable conformation happens without need for energy
role of ATP in movement
movement of cell components
change shape of cell, locomotion
source of energy required to convert ADP and phosphate back to ATP
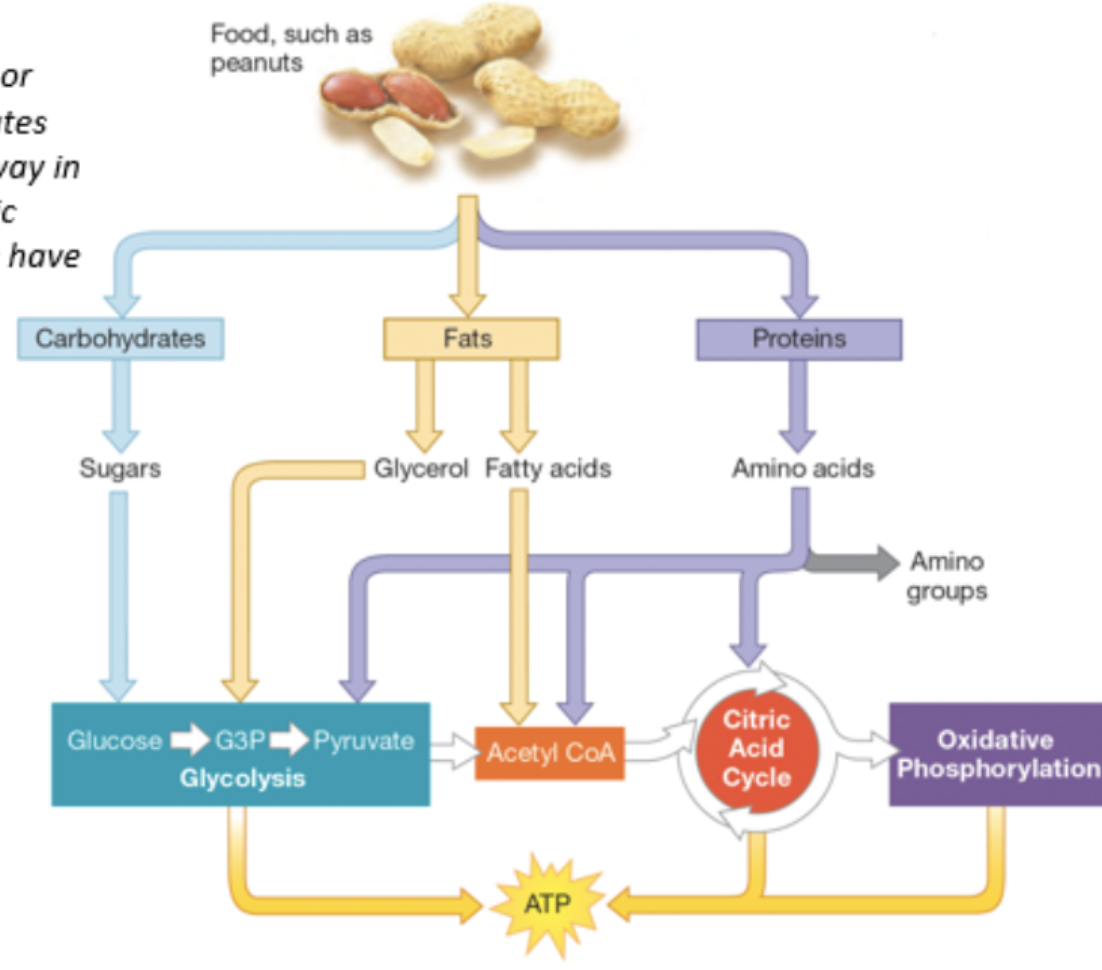
cell respiration - oxidation of carbohydrates, fats, or proteins
photosynthesis - light energy converted to chemical energy
chemosynthesis - oxidation of inorganic substances such as sulfides
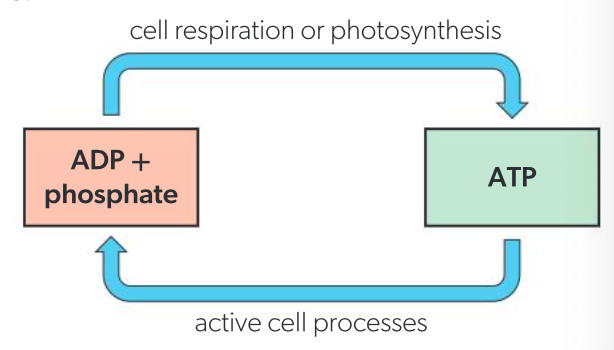
efficiency of interconversion of ATP and ADP
not 100%, some energy transformed into heat
gas exchange
carbon dioxide and oxygen move across the plasma membrane independently by simple diffusion (not one-for-one swapping), interdependent process with cell respiration
aerobic respiration in humans and many other animals and plants
C6H12O6 + 6O2 -(ADP→ATP)→ 6CO2 + 6H2O
anaerobic respiration in humans, other animals, and some bacteria
glucose -(ADP→ATP)→ lactate
anaerobic respiration in yeast and other fungi
glucose -(ADP→ATP)→ ethanol + carbon dioxide
features unique to aerobic cell respiration
oxygen used as electron acceptor in oxidation reactions
carbohydrates such as glucose, lipids including fats and oils, and amino acids after deamination can be used
CO2 and H2O are waste products
ATP yield of more than 30 ATP molecules per glucose
initial reactions are in the cytoplasm, more reactions in mitochondria
features unique to anaerobic cell respiration
oxygen is not used - other substances act as oxygen acceptors in oxidation reactions
only carbohydrates can be used
CO2 plus either lactate or ethanol are the waste products, water is not produced
ATP yield of only 2 ATP per glucose
all reactions happen in the cytoplasm, mitochondria not required
lactate/lactic acid
waste product of anaerobic respiration in muscles, limited tolerance in human body, breakdown requires oxygen
oxygen debt
demand for oxygen that builds up during a period of anaerobic respiration
oxidation reaction
loss of electrons and hydrogen atoms, release energy ex. C6H12O6 → 6CO2
reduction reaction
gain of electrons and hydrogen atoms, gain energy ex. 6O2 → 6H2O
electron carriers
substances that can accept and lose electrons reversibly, often linking oxidations and reductions in cells
NAD reduction
NAD+ + 2H+ + 2e- → NADH + H+
role of FAD as a carrier of Hydrogen
FAD → FADH2
stages of cellular respiration
glycolysis
link reaction
Krebs/citric acid cycle
electron transport chain and chemiosmosis
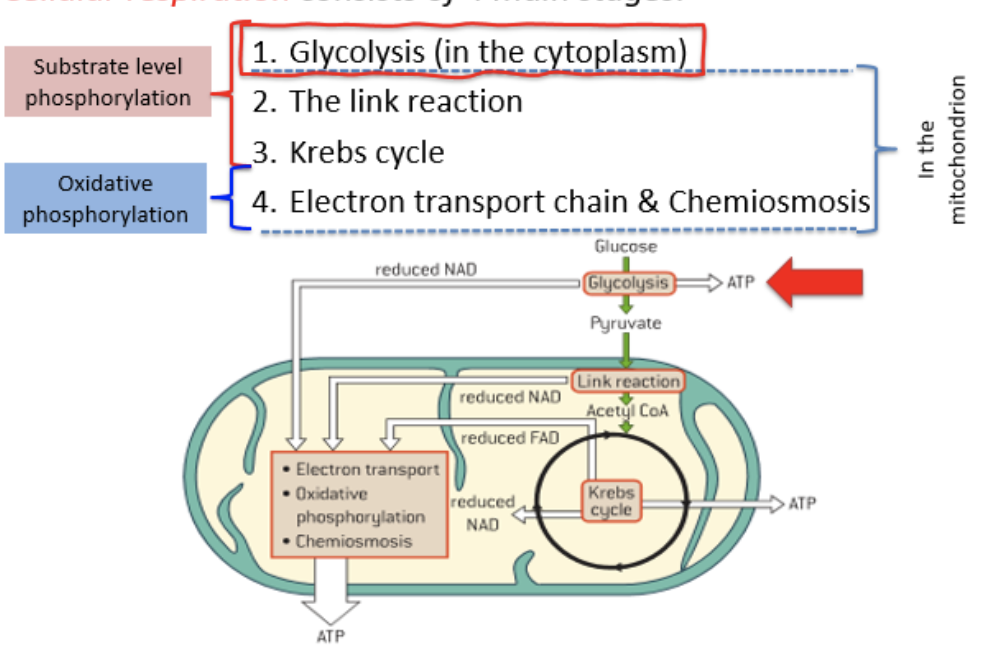
glycolysis
conversion of glucose to pyruvate in the cytoplasm of cells by aa chain of reactions, each catalyzed by a different enzyme, creating a small yield of ATP without oxygen consumption
phosphorylation of glucose
lysis
oxidation
ATP formation
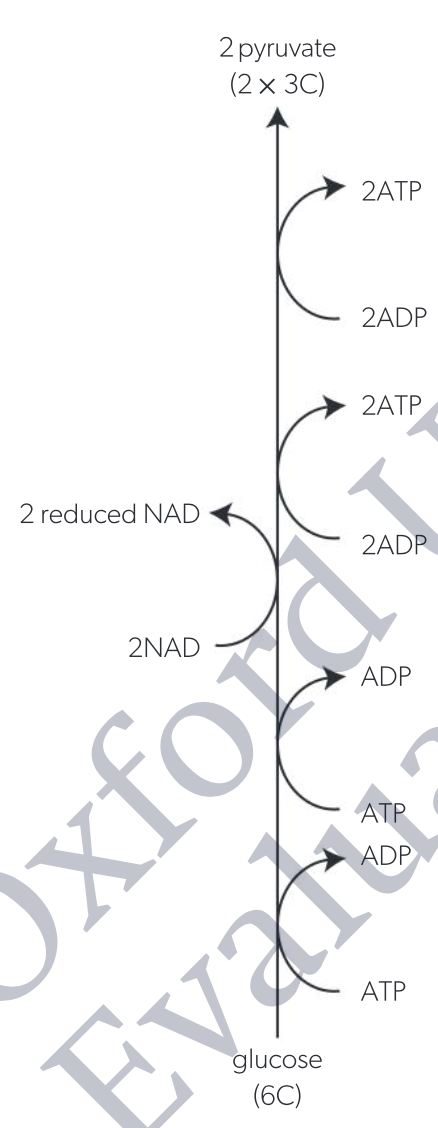
phosphorylation of glucose
addition of PO43- group(s), makes molecule more unstable and therefore more likely to participate in subsequent reactions
glucose -(ATP→ADP)→ glucose-6-phosphate (phosphorylation)
glucose-6-phosphate → fructose-6-phosphate
fructose-6-phosphate -(ATP→ADP)→ fructose-1,6-bisphosphate

lysis
fructose-1,6-bisphosphate → 2 triose phosphate

oxidation
triose phosphate + phosphate -(NAD→reduced NAD)→ bisphosphoglycerate
energy released by oxidation of triose allows second phosphate group to become attached
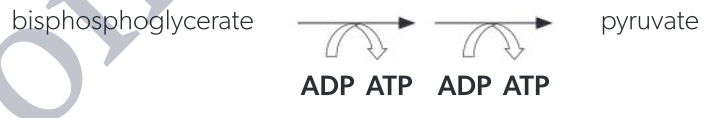
ATP formation
bisphosphoglycerate -(2ADP→2ATP)→ pyruvate
transfer of phosphate groups to ADP occurs twice because bisphosphoglycerate has two phosphates
4 ATPs produced per glucose
glycose (6C) converted to 2 pyruvate (3C each)
2 NAD converted to 2 reduced NAD
net yield of 2 ATPs
uses of pyruvate
anaerobic respiration (alcoholic fermentation, lactic acid fermentation) and aerobic respiration depending on oxygen availability
anaerobic respiration
NADH + H+ donates its H+ and e- to pyruvate during ethanol or lactate production, respectively
NAD+ becomes available again so that e- and H+ can continue to be transferred from glucose to NAD+ when oxidized
thus pyruvate synthesis during glycolysis continues
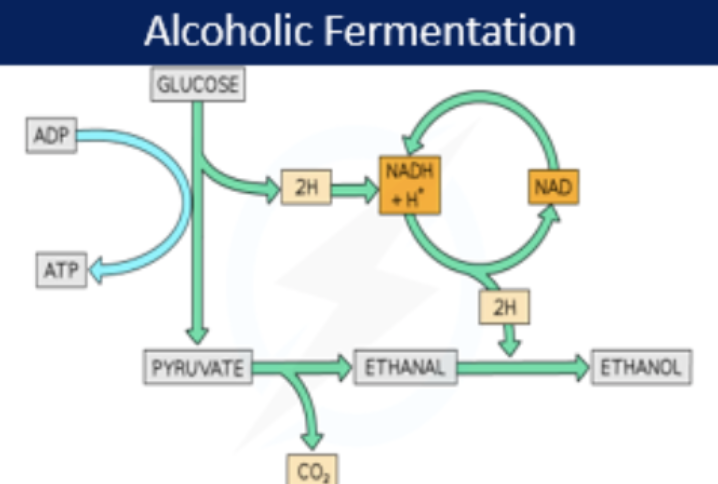
alcoholic fermentation
C6H12O6 → 2 pyruvate → 2 Ethanol + 2CO2 (net 2 ATP)
in yeast (eukaryotic cell) and some bacteria (prokaryotic cell)
cytoplasm
yeast as an example of anaerobic cell respiration
facultative anaerobe single-celled fungus, can respire aerobically or anaerobically
breaks down starch and sugars in dough by alcoholic fermentation to make CO2 and ethanol
CO2 released in fermentation trapped in dough, causing bread to rise
bread baked in oven to kill yeast and trap ethanol
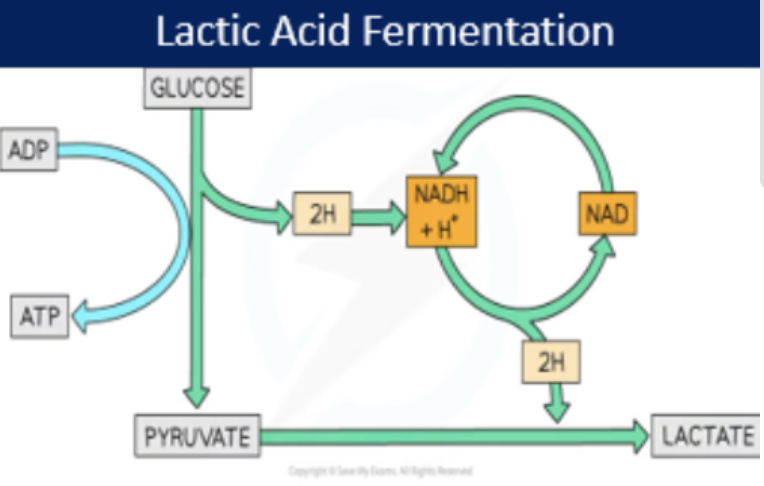
lactic acid fermentation
C6H12O6 → 2 Lactate (net 2 ATP)
some bacteria (prokaryotic cell) and some mammals
cytoplasm
aerobic respiration
C6H12O6 → 6CO2 + 6H2O (net 32-34 ATP)
before the link reaction
pyruvate/pyruvic acid enters mitochondrial matrix by facilitated diffusion to be processed further
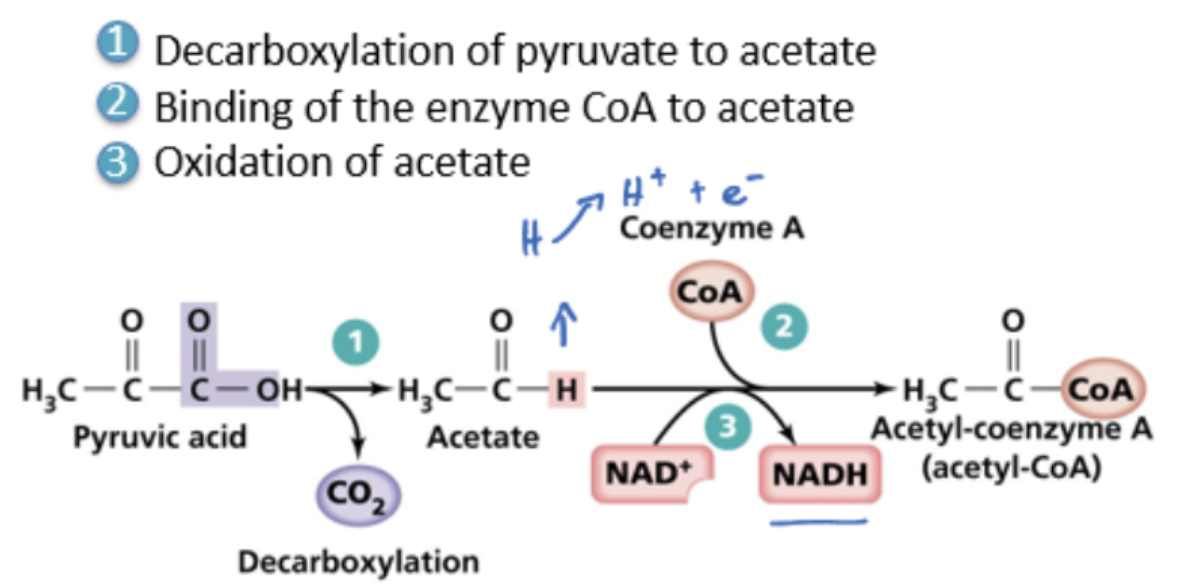
link reaction
decarboxylation of pyruvate (3C) to acetate (2C)
CO2 leaves cell, diffuses into the bloodstream and is expired
binding of enzyme CoA to acetate
oxidation of acetate into acetyl-coenzyme A
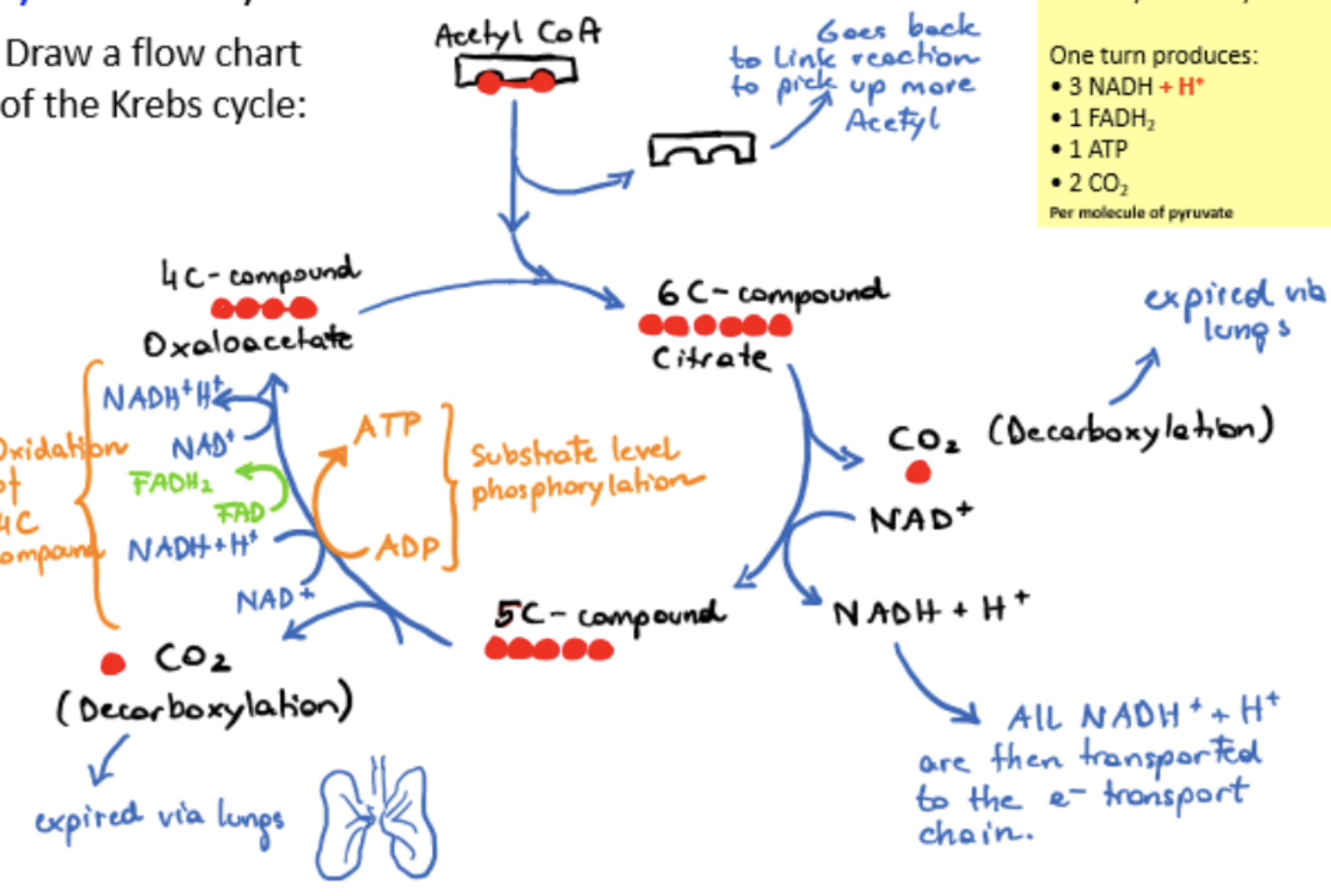
Krebs/citric acid cycle
oxidation and decarboxylation of acetyl groups
acetyl groups (2C) fed into cycle by transfer from coenzme A to oxaloacetate (4C) → citrate (6C)
citrate converted back to oxaloacetate by enzyme-catalyzed reactions, two carbons lost through decarboxylation reactions, creating CO2 waste product
4 oxidation reactions release energy, mainly held in electrons removed from oxidation and transferred through reduction of NAD+ and FAD
net products per acetyl-CoA molecule: 2CO2, 3NADH, 1 FADH2, 1 ATP
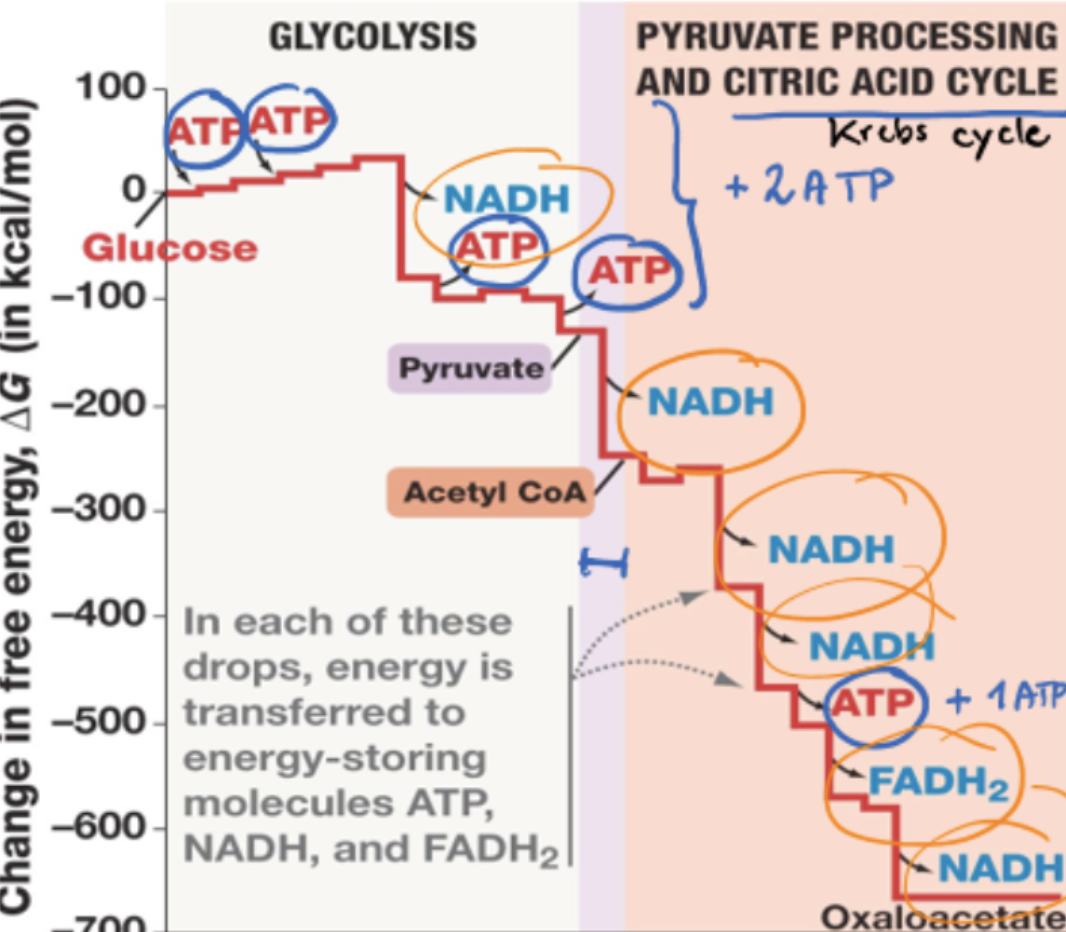
change in free energy graph in electron transport chain
in each drop, energy is transferred to energy-storing molecules NAD+ and FAD, which later become oxidized again in electron transport chain
energy gained from oxidation reaction used to make ATP
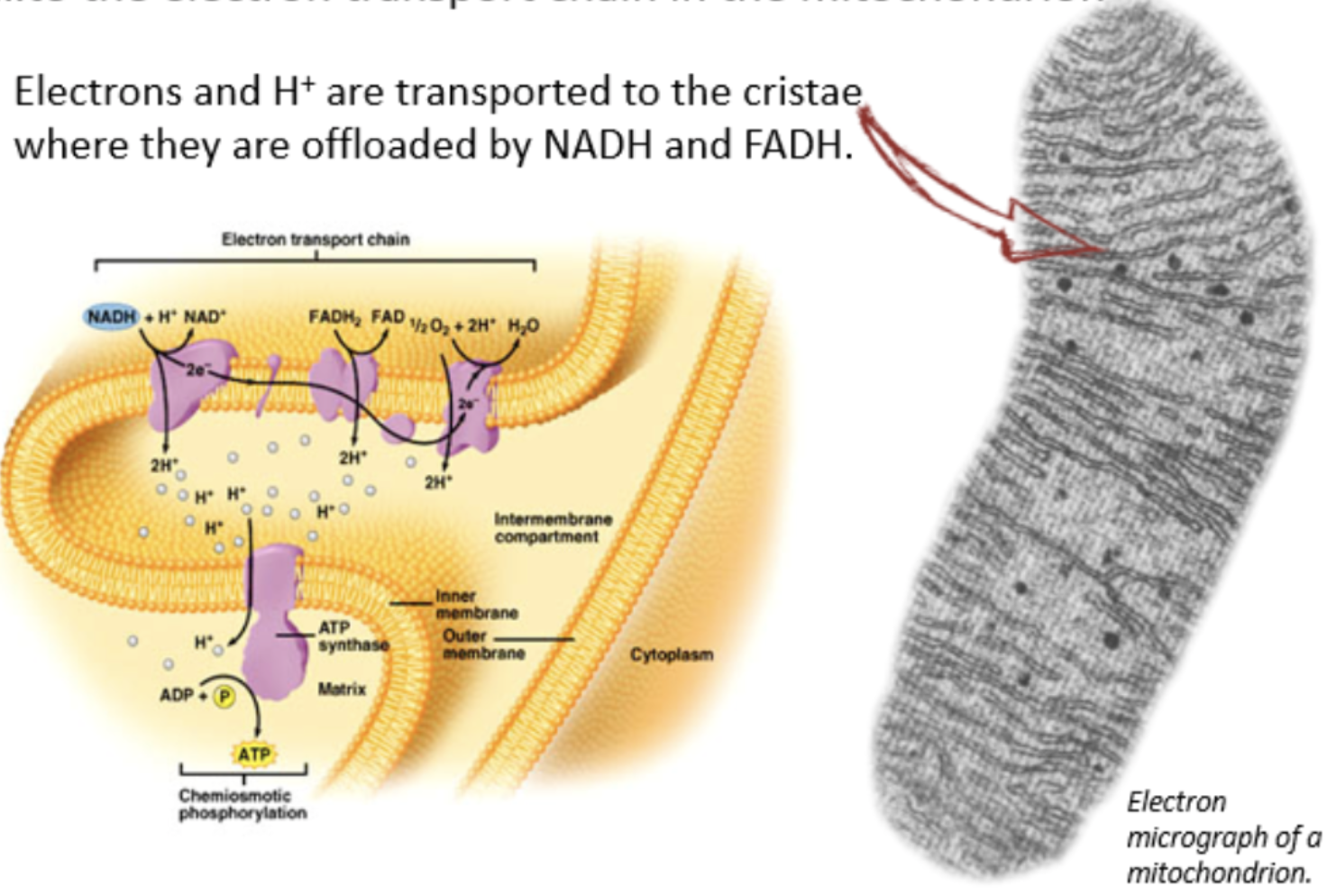
transfer of energy to the electron transport chain
electron carriers NAD+ and FAD bring electrons and hydrogen ions to electron transport chain in cristae of the mitochondria
electron transport chain
reduced electron carriers NADH + H+ and FADH2 from glycolysis and Krebs cycle move to inner mitochondrial membrane
membrane proteins accept electrons from NADH and FADH2
each carrier in chain has slightly higher electronegativity and therefore a stronger attraction for electrons than previous carrier
electrons passed down an energy gradient until they reach the end of the chain
electrons “fall” from higher levels to lower ones, energy released used to pump protons from matrix into intermembrane space against the concentration gradient
proton gradient drives chemiosmosis
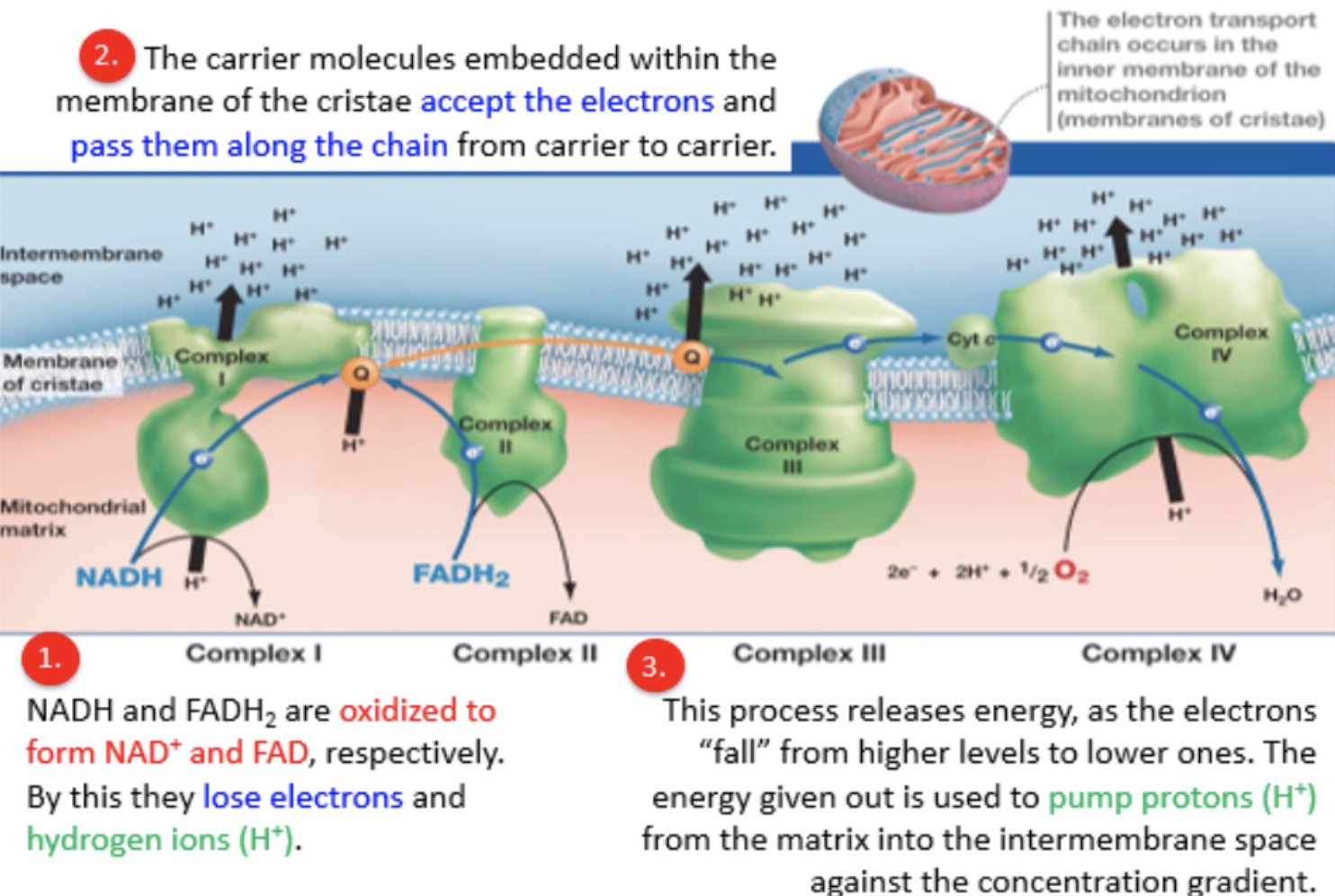
membrane proteins in electron transport chain
integral carrier and channel proteins embedded within phospholipid bilayer have a high tendency to become reduced by accepting electron
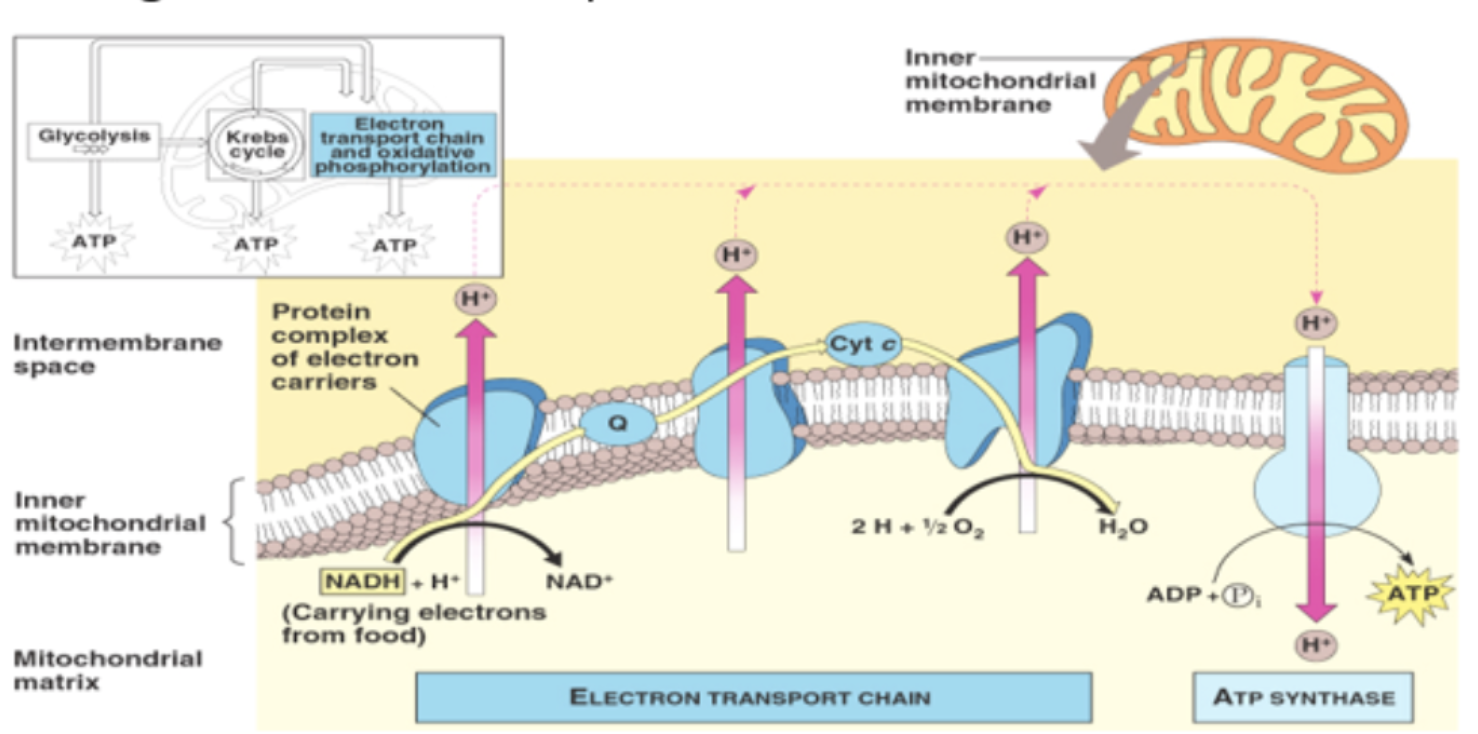
chemiosmosis
transfer of H+ sets up concentration gradient across the membrane as H+ accumulates in the intermembrane space
protons follow natural concentration gradient by moving through the ATP synthase into matrix
oxidative phosphorylation
oxygen as the terminal electron acceptor
oxidative phosphorylation in between chemiosmosis and ATP synthesis
movement of protons through ATP synthase down the concentration gradient releases energy used to phosphorylate ADP to ATP
ATP synthase
complex of integral proteins located in the mitochondrial inner membrane where it catalyzes the synthesis of ATP from ADP and phosphate, driven by a flow of protons
role of oxygen as terminal electron acceptor
in the reduction of the oxygen molecule, the O2 accepts electrons and forms a covalent bond with hydrogen to produce H2O
in the matrix, H+ combines with ½ O2 + 2e- to form water
carbohydrates as respiratory substances
simple sugars (glucose or fructose) can be used straight away in glycolysis and anaerobic respiration
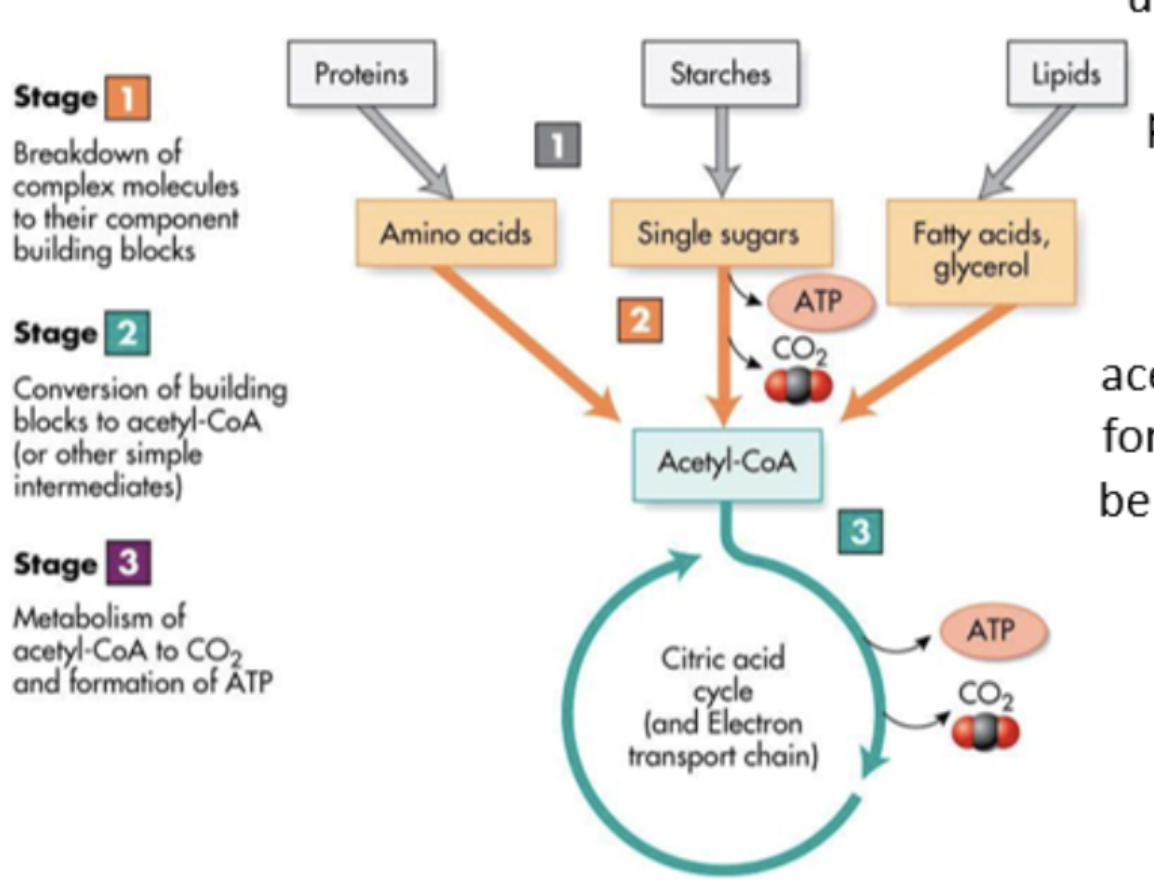
lipids as respiratory substances
lipids broken down into glycerol and fatty acids, fatty acids converted to acetyl groups to be used in Krebs cycle