principles - chemical formulae, equations and calculations
1/18
Earn XP
Description and Tags
without all the experiments from the moles test
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
19 Terms
what is Mr
relative molecular mass (sum of Ar)
what is Ar
the atomic mass - from periodic table - protons + neutrons
what is Mr
the molecular mass - sum of Ars
define mol
the unit for amount of a substance
mass = ___ x ___
mass = Mr x moles
percentage yield = ___ / ____ (x____%)
percentage yield = actual yield (g) / theoretical yield (g) x100%
why would you get less product than theoretically (3)
loss of reactants in transfer
side reactions
some reactions are reversible, they reverse
not everything reacted under heat ()
define empirical formula
the simplest whole number ratio of atoms in a compound
how to find empirical formula (Mg - 2.4, O - 1.6) the table
Mg | O | |
|---|---|---|
mass | 2.4 | 1.6 |
Ar | 24 | 16 |
moles | 0.1 | 0.1 |
ratio | 1 (divide all by smallest number) | 1 |
what is molecular formula
the actual number of atoms of each element in a molecule of the compound
general, how to find molecular formula
first find empirical formula
find Mr of empirical formula, compare that with given Mr of molecular thing
always a multiple of the empirical, what needs to be multiplied up
concentration moles formula
moles = concentration (mol/dm3) x volume (dm3)
gas volume moles formula
moles = gas volume (dm3) / 24
1 mole takes up 24dm3 at room tempw
how to go from mol/dm3 to g/dm3 and vice versa
the logic also
To go from g/dm3 to mol/dm3: Divide by the molar mass in grams
To go from mol/dm3 to g/dm3: Multiply by the molar mass in grams
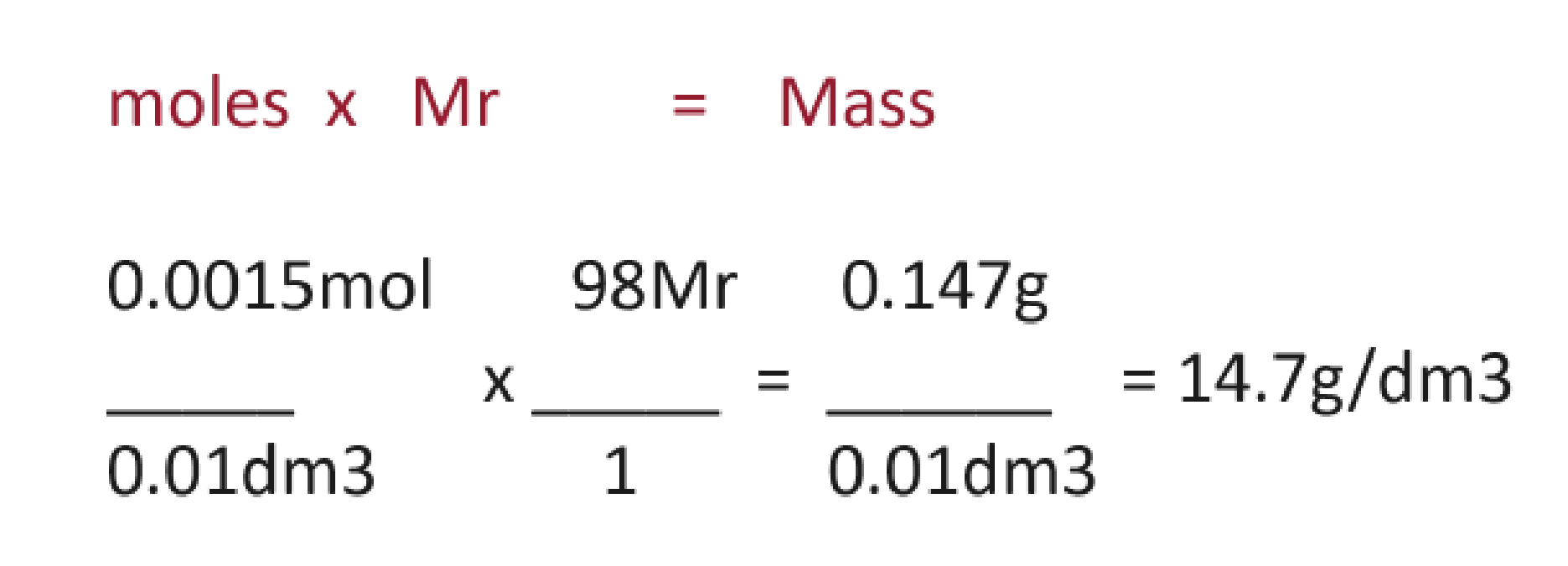
why do all gasses 1 mole = 24dm3
because the volume is essentially the same as the particles are so small their actual size is negligible, space in between creates the volume
24 dm3 in cm3
24 000 cm3
concentration formula with mass - g/dm3 conc.
how to find excess reagent, general steps
chemical formula is the correct one
compare formula moles with moles used in experiment
compare the ratios - where one reactant is ‘used more‘ in the experiment than in the formula, it is in excess
same if there is not enough of one irl reactant vs the ratio, that’s the limiting reagent so the other is in excess
Mg + 2Hcl → MgCl2 → H2O
0.3 mol reacts with 0.3 mol of Hcl, which is in excess
check notes
its Mg in Excess