Electronegativity and Polar Bonds
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
5 Terms
definition of non-polar (same charge)
there is an identical force of attraction between nucleus of each atom and shared pairs of electrons
definition polar (different charge)
a molecule with an uneven distribution of charge - one part of molecule is negative and one part of the other molecule is positive
smaller molecule - closer to the nucleus so stronger attraction rather than larger molecule
definition of electronegative
the relative ability of an atom to attract a pair of electrons to form a covalent bond
use FONClBrISCH - to know which element is the most and least electronegative
Explain instaneous dipole-induced dipole (mainly in nonpolar molecules)
electron movement leads to uneven distribution of charge
dipole is induced in a neighbouring molecule
very weak bonds are formed - little energy needed to break due to low melting/boiling points
larger molecules have more electrons so stronger bonds
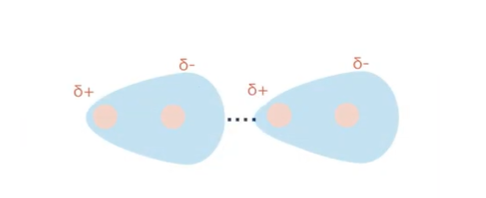
simple definition of instaneous dipole
a temporary dipole moment that arises due to random motion of electrons