Metabolism Part I
1/61
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
62 Terms
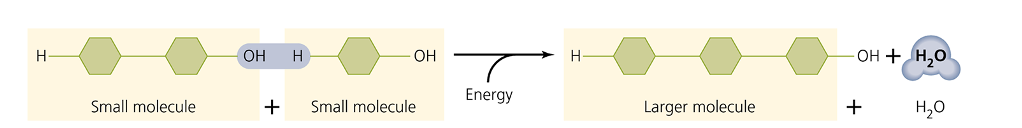
Synthesis
Formation of a larger, more complex molecule
Decomposition
Reverse of synthesis, wherein large molecules are broken down to smaller ones
Exchange
Combination of both synthesis and decomposition
Reactants
the matter that begin the reactions (atoms, molecules, etc)
Synthesis
Require energy to break bonds in reactants to form new bonds in products
endothermic reactions
Reactions that trap energy within new molecular bonds
Dehydration synthesis
Water molecule formed (removed)
anabolic, energy required rxn
anabolism
All the synthesis reactions in an organism are called
Decomposition
Break bonds within larger molecules to form smaller atoms, ions, and molecules
Exothermic
Release energy
hydrolysis
Ionic components of water are added to products
exothermic rxn
Exchange
•Involve breaking and forming covalent bonds
•Have endothermic and exothermic steps
•Atoms are moved from one molecule to another
Metabolism
sum of the chemical reactions that take place within each cell of a living organism and that provide energy for vital processes and for synthesizing new organic material.
Endergonic reactions
require energy
Exergonic reactions
release energy
Redox reactions
Transfer of electrons between two molecules or atoms
electron carriers
NAD+/NADH, FAD/FADH2, and NADP+/NADPH are all examples of what?
catabolism
________ in chemoheterotrophs:
NAD+/NADH
FAD/FADH2
anabolism
photosynthesis and ________:
NADP+/NADPH
chemical work
refers to synthesis of complex molecules
Transport work
refers to uptake of nutrients, elimination of waste and balance of ions across membranes
Mechanical work
refers to cell motility and movement of structures within or on the surface of a cell
energy
Chemical work, Mechanical work, and Transport work all require what?
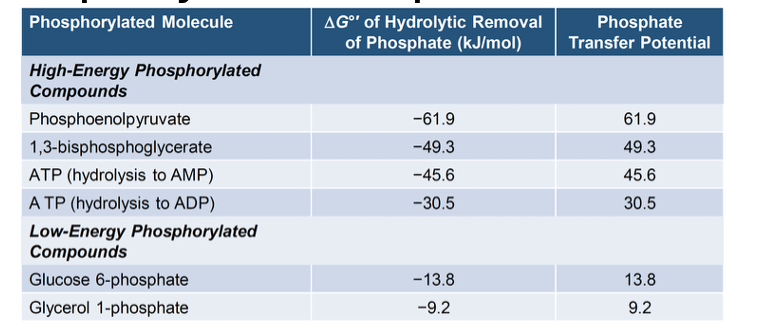
ATP (Adenosine triphosphate)
The molecule that stores energy in the cell
high
High or Low?
bonds between phosphate groups are ____-energy phosphate bonds
Exothermic reactions
form spontaneously
ΔG°’ is negative
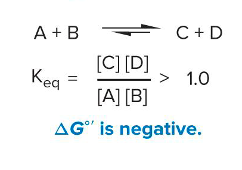
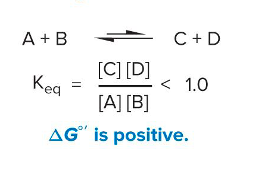
Endothermic reactions
Form non-spontaneously
ΔG°’ is positive
Delta G (ΔG°’)
Maximum amount of energy available from the system for useful work under standard conditions
Respiration
The final goal is to generate energy (ATP)
(aerobic or anaerobic)
ATP
Which molecule:
has a high phosphate transfer potential
Readily donates its phosphorylation group to other molecules
higher; spontaneous
The more negative ΔG°’, the _____ (higher/lower) phosphate transfer potential, which means it will be ______ (non-spontaneous/spontaneous).
Oxidation
loss of electrons
Reduction
gain in electrons
Glucose; aerobic
Which rxn is this? Is it aerobic or anaerobic?
C6H12O6 + 6 O2 + 6H2O → 6 CO2 + 12 H2O + energy (ATP)
O2 is reduced to H2O
In this reaction:
C6H12O6 + 6 O2 + 6H2O → 6 CO2 + 12 H2O + energy (ATP)
What is reduced?
C6H12O6 is oxidized to 6CO2
In this reaction:
C6H12O6 + 6 O2 + 6H2O → 6 CO2 + 12 H2O + energy (ATP)
What is oxidized?
Amylase
breaks down glycogen and starch
Cellulase
breaks down cellulose
Glucose
What are these polymers made out of? What will be released?
Starch
Glycogen
Cellulose
uses
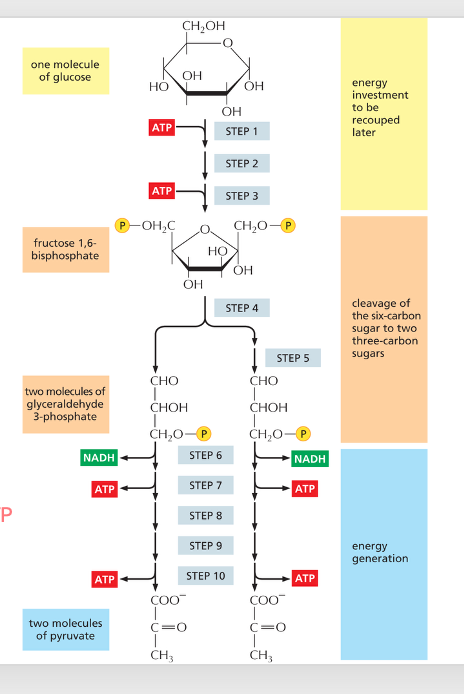
Glycolysis 1st phase:
uses or generates ATP?
generates
Glycolysis 2nd phase:
uses or generates ATP?
2; 4; 2
Glycolysis uses ___ ATP, produces ____ ATP and ___ NADH
Glycolysis
Produces energy and reduces electron carries and precursor molecules for metabolism
most common catabolism of glucose for bacteria, archaea and eukaryotes
Does NOT require oxygen
substrate level
ATP in glycolysis is synthesized in by which type of phosphorylation?
Pentose-phosphate pathway
gives it precursors for / important pathway for synthesis of:
nucleic acids, amino acids
NADPH
The Pentose-phosphate pathway produces 2 _____.
NADP+; NADPH/H+
Glycolysis and Pentose-phosphate pathway (PPP) share which two electron carriers?
nucleic acids; amino acids
Ribulose 5-phosphate is a precursor (helps create) for what?
pyruvate; Acetyl-CoA
After glycolysis, ________ is decarboxylated and converted into _________.
Eukaryote
Generates pyruvate and converts it into acetyl-CoA in the mitochondria
prokaryote
Generates pyruvate and converts it into acetyl-CoA in the cytoplasm
CO2; NADH
Pyruvate → Acetyl-CoA + ____ + ____
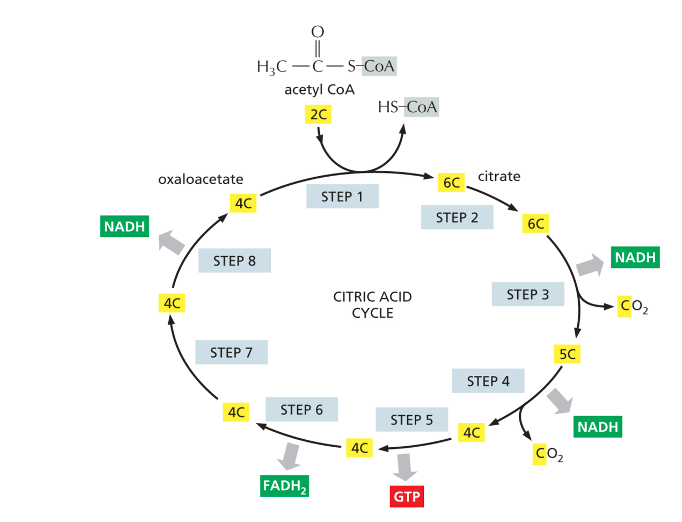
Krebs Cycle (Citric Acid Cycle) (tricarboxylic acid cycle (TCA))
Produces one ATP (or GTP), one FADH2, and three NADH
recall: they’re all electron carriers
TCA
Intermediates of ____ can be used for biosynthesis of amino acids, lipids, nucleotides etc.
ATP
glycolysis and Krebs cycle are mostly used to generate
catabolic; anabolic
Glycolysis and Krebs cycle are mainly ________, but also support ________ pathways.
anabolic; macromolecules
Glycolysis and the Krebs cycle provide intermediates for ________ pathways to produce ________.
substrate level phosphorylation
a phosphate group is removed from an organic molecule to ADP to make ATP
only 5 molecules are produced (4 by glycolysis and one by Krebs cycle)
oxidative phosphorylation
occurs through cellular respiration and the most ATP is generated
~28 molecules
Krebs cycle
Which cycle?
Glycolysis
Which cycle?