Lecture 1 & 2 - Radiation Absorption and Ionizing Radiation-Induced Cell Damage
1/59
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
60 Terms
What is radiation biology?
a field of clinical and basic medical sciences that involves the study of action of ionizing radiation on living things
Radiation’s interaction with matter can cause two things, what are they?
excitation
ionization
Excitation
raising of electrons into higher orbitals/energy states
BOND DOES NOT BREAK

Ionization
release of bound electron from the atom
ion is created
Two types of ionizing radiation
electromagnetic radiation
particulate radiation
X-Rays vs. Gamma rays
Energies are similar, but source is different
X-rays come from electron release
bremsstrahlung or characteristic
Gamma comes from nucleus energy release
photon released from decay
what are the three types of ionizing electromagnetic waves?
X-Rays
Gamma
high energy UV rays (sometimes)
Photon speed, charge and mass
Speed = c
mass = none
charge = none
pass undeflected through B field
Do photons interact with matter?
yes they can, but they can also pass through matter unimpeded and unchanged
How does the energy of electromagnetic waves relate to it’s frequency and wavelength
E=hv = E=h(c/wavelength)
energy is proportional to frequency, and inversely proportional to wavelength
What does quantum mechanics suggest
some physical quantities exist and can change and interact, but only by discrete amounts (quanta) and behave probabilistically rather than deterministically
wave particle duality
a particle can behave either like a wave or a particle depending on how it is observed
heisenberg’s uncertainty principle
states that it is impossible to simultaneously and precisely measure both the position and momentum of a particle.
why is quantum physics important for radiobiology?
if photons are absorbed in living material, the energy deposited in the tissues and cells is deposited in discrete packets
what is the difference between nonionizing and ionizing radiation?
the size of the individual energy packets
not total energy is involved
1 Gy of the dose is stronger than 10 fractions of 10 cGy
What is the lethal dose of ionizing radiation
4 Gy (4 J/Kg)
average man = 75 kg, so 300 J
300 J is the same amount of heat energy transferred to bodt after drinking a 3.1 mL sip of coffee at 60 degrees celsius
What are electronic orbitals
regions of highest probability of where the electron will most likely be
types of particulate radiation
electrons, protons, alpha particles, neutrons, etc
electron mass, charge, and radioactive decay
mass: 9.11 × 10-31 kg
charge: negative
radioactive decay: beta minus
How are electrons used in radiotherapy
due to their negative charge, electrons can be accelerated in an electromagnetic field to either be used to treat superficial tumors, or can be used to hit tungsten target to generate X-rays
Proton charge, mass, use in therapy
charge: positive
mass: 1836x mass of electron
therapy: proton therapy, bragg peak is more OAR sparing
Neutron charge, mass, radioactivity
charge: none
mass: slightly heavier than protons
radioactivity: can be used to collide with nuclear protons, ejecting them
can be used to produce ionizations and excitations with these protons
neutrons also used in fission reactions
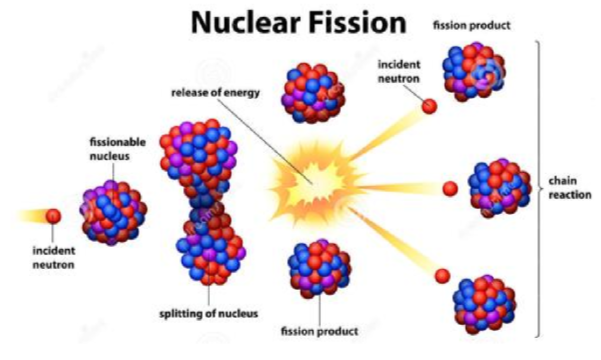
where can humans be exposed to neutrons more frequently?
flying
alpha particles
nuclei of helium atom (2 P, 2 N), postively charged
radium decay produces alpha particles
Examples of heavy charged particles that may be used in radiotherapy
carbon, neon, sillicon, argon
two types of ionizing radiation
directly ionizing
by charged particles with sufficient KE to break bonds
indirectly ionizing
by producing charged particles with high KE
What are the three electromagnetic interactions photons can undergo with subatomic structures?
photoelectric absorption
compton scattering
pair production
How is the probability of the interaction determined?
based on the energy of the photon and the atomic number of the target atom
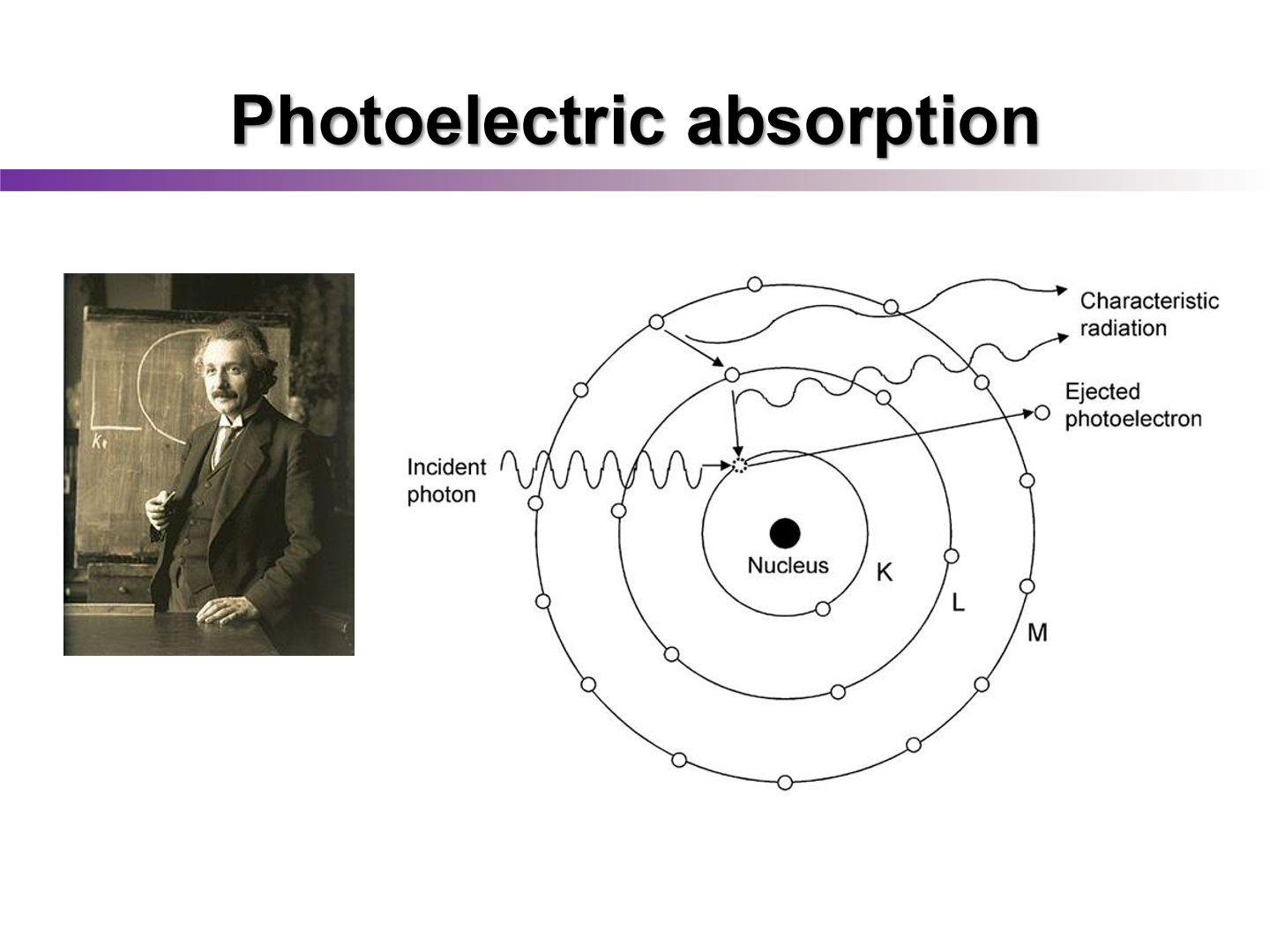
photoelectric absorption
compton scattering
inelastic scattering of a photon by a charged particle
energy of photon is decreased as part of energy is transferred to recoil e-
How does the compton effect vary with atomic number
compton cross section is independent of atomic number
how does the photoelectric effect vary with atomic number
cross section depends strongly on atomic number of absorbing material
Z3
At what energies (low or high) does compton and P.E occur
Compton: prevails at higher energies
P.E: lower energies
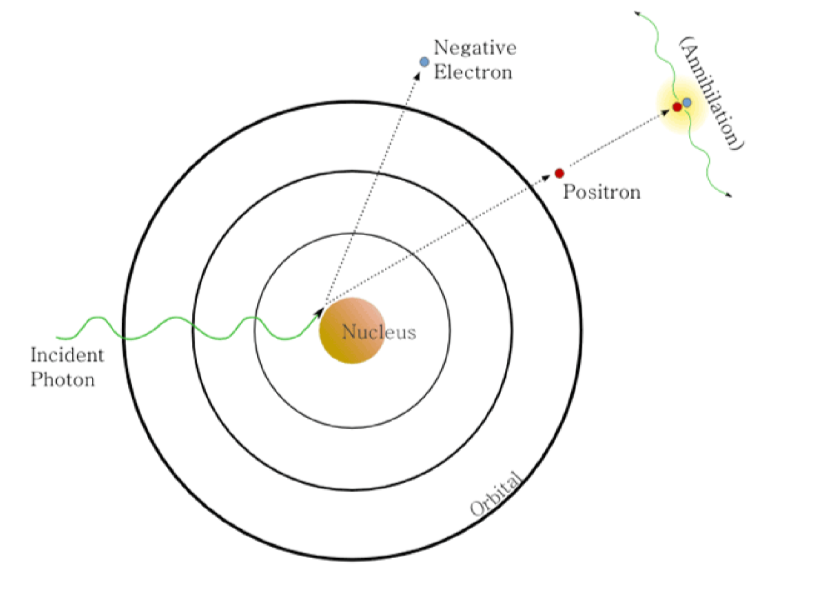
Pair Production
for photons above the baseline of 1.022 MeV, they can interact with the electric field of the nucleus and form an electron-positron pair
positron often annhilated with electron to release two more photons
photons energy is converted into mass of beta particles through mass-energy equivalence E=mc2
What photon energies would you use for X-ray radiation therapy, and what electromagnetic interactions would you see?
4-25 MeV
PE, Compton, and PP all possible
What photon energies would you use for X-ray CT scans, and what electromagnetic interactions would you see?
20-150 keV
mostly PE, potentially Compton, NO PP (E<1.022 MeV)
Direct Ionizing Radiation
dominant process for radiation with high linear energy transfer (LET)
Indirect ionizing radiation
radiation produces free radicals that interact with the target
free radical
a free radical is an atom, molecule, or ion that has unpaired valence electrons

hydroxyl radical, superoxide, and oxygen
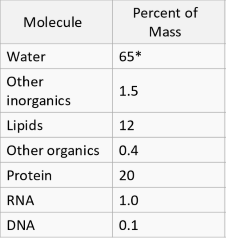
what percent mass of a human cell does water make up
65%
what percent molecules of a human cell does water make up?
98.73%
When an indirect ionizing event occurs, what is the most likely target?
Water
When an indirect ionizing event occurs, what possible targets are there
all are theoretically possible
DNA the least common
Radiolysis of Water Mechanism
H2O + x-ray —> H2O++ e-
e- pathway:
e- + H2O —> H2O-
H2O- —> H. + OH-
H2O+ pathway
H2O+—>H+ + OH.
end product of radiolysis e- pathway
H. + OH-
hydrogen free radical and hydroxide ion
end product of radiolysis H2O+ pathway
H+ + OH.
Proton and hydroxyl radical
Describe the chain of event in X-Ray Absorption (indirect pathway)
Incident X-Ray beam —> recoil electron —> ion radical —> free radical —> chemical changes from bond breakage —> biological effects
Time for initial ionization to occur
10-15 s
Ion radical life time
10-10 s
Free radical lifetime
10-5 s
enough time to float around and cause bond breakage in cells
Time scale of radiation physics
10-12 - 10-15 s
ionization, excitation, radical formation
time scale of radiation chemistry
10-6 - 10-9 s
DNA damage formation
time scale of radiation biology
103-106 seconds
repair replication, cell death, cancer, heritable mutations
Linear Energy Transfer (LET) definition
how much energy an ionizing particle transfers to material transversed per unit distance
What two factors does LET depend on
nature of radiation
nature of material
what travels deeper in tissue? high LET or low LET
Low LET
high LET gives more energy into material, so may run out of energy before getting deeper into tissue
low LET gives less energy to material, allowing it to go deeper into tissue
When could a high LET be used
to cause more severe damage to any microscopic structures near the particle tract
Gamma/X-Rays: high or low LET
Low LET, thus they need more shielding as they can penetrate deeper
alpha particles: high or low LET
high LET, less shielding needed