AP Bio Units 1-4
1/155
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
156 Terms
Active site
Site substrate attaches to on an enzyme
Allosteric site
Other location on an enzyme that an inhibitor can bind to
Catalyst
Speeds up chemical reactions without being consumed (or changed) by the reaction
Polypeptide
Polymer (chain) of amino acids
Primary structure
Sequence of amino acids; determined by genes
Secondary structure
Coils and folds in the polypeptide chain resulting from hydrogen bonds between amino group of one amino acid and the carboxyl group of another; alpha helices and beta-pleated sheets
Tertiary structure
Interactions among various R groups of a polypeptide

Quaternary structure
Interactions between multiple (key word) polypeptide chains
Monomer
Smaller, simpler molecules that are linked together to form larger, more complex molecules
Polymer
A large complex molecule formed from simpler molecules (monomers)
Dehydration synthesis reaction
A chemical reaction in which two molecules covalently bond to each other with the removal of a water molecule
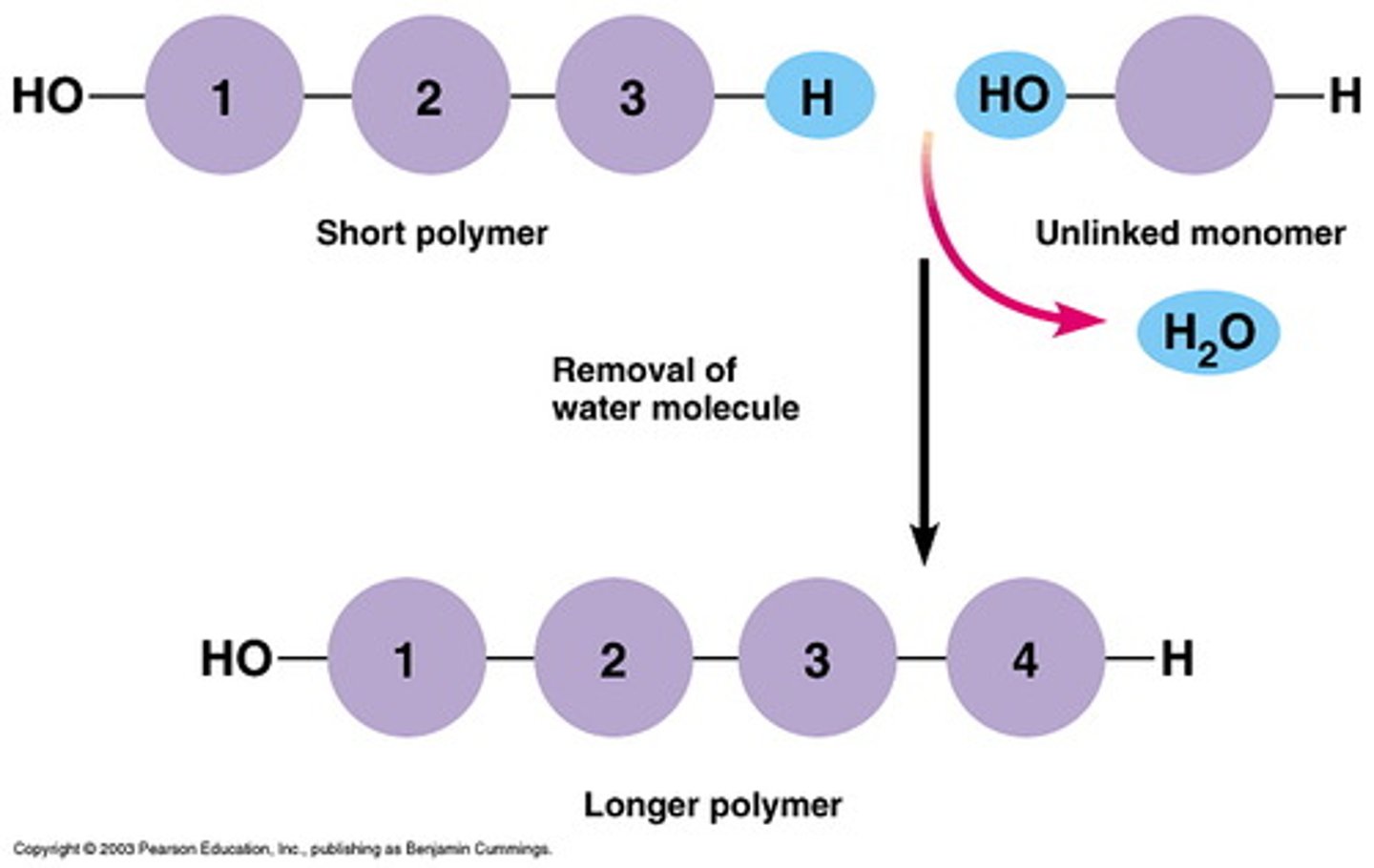
Hydrolysis
Breaking down complex molecules by the chemical addition of water

Denature
Unfold a protein's structure; occurs in extreme temperatures & pH ranges
Synthesis
To make or to build
Substrate
The molecule that is changed by the enzyme
Optimal/ optimum
Best or most favorable
Control treatment
Lacks (or does not receive) the specific factor being tested in an experiment
Dependent variable
Factor that is measured, which may change in response to the independent variable; always the y-axis on a graph
Independent variable
Factor that is purposely changed; this is what is being investigated
Hydrogen bond
A weak chemical bond between a hydrogen on one molecule and an electronegative atom on another molecule
Hydrophilic
"water loving"; substance can interact with water and is able to form hydrogen bonds; POLAR
Hydrophobic
"water fearing"; substance cannot interact with water and is unable to form hydrogen bonds; NON-POLAR
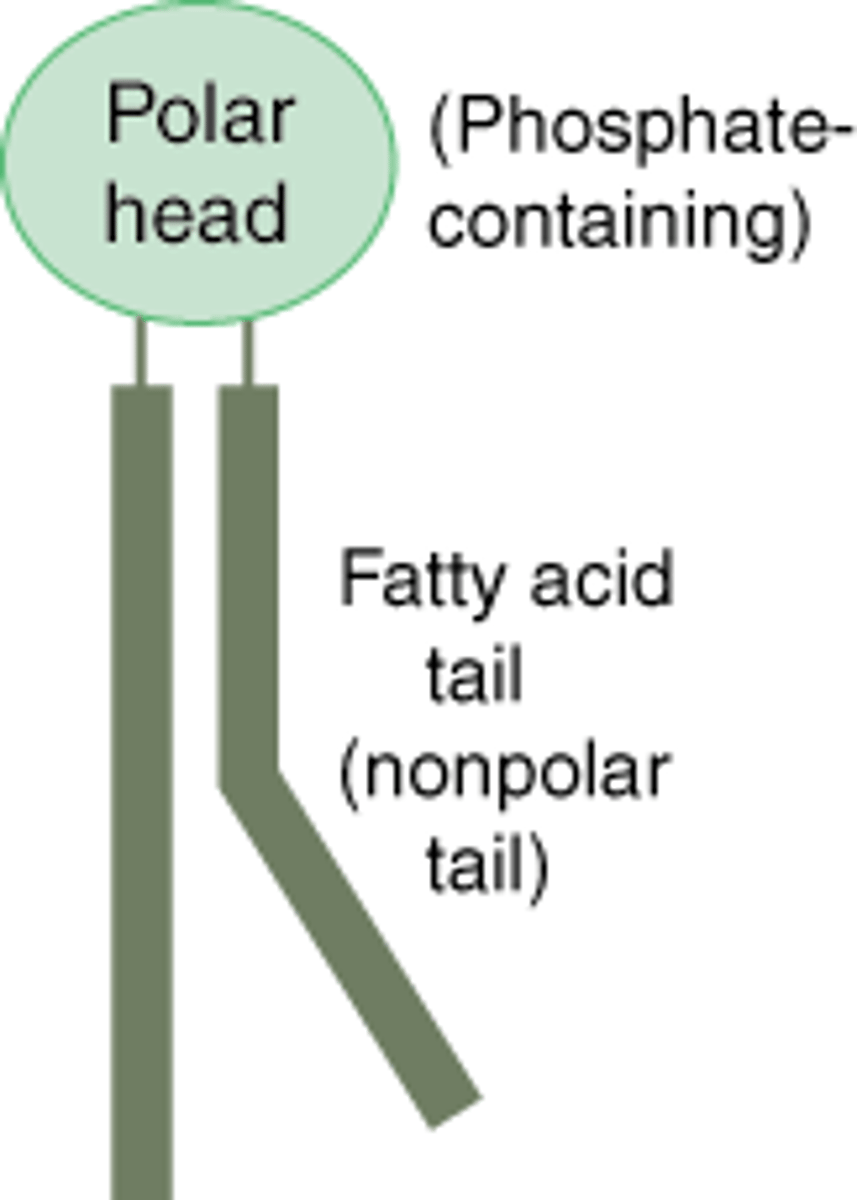
Phospholipid diagram
Polar covalent bond
One atom is more electronegative, and the atoms do not share the electron equally; Unequal sharing of electrons causes a partial positive or negative charge for each atom or molecule
Nonpolar covalent bond
The atoms share the electron equally
Example: oxygen molecule, carbon dioxide
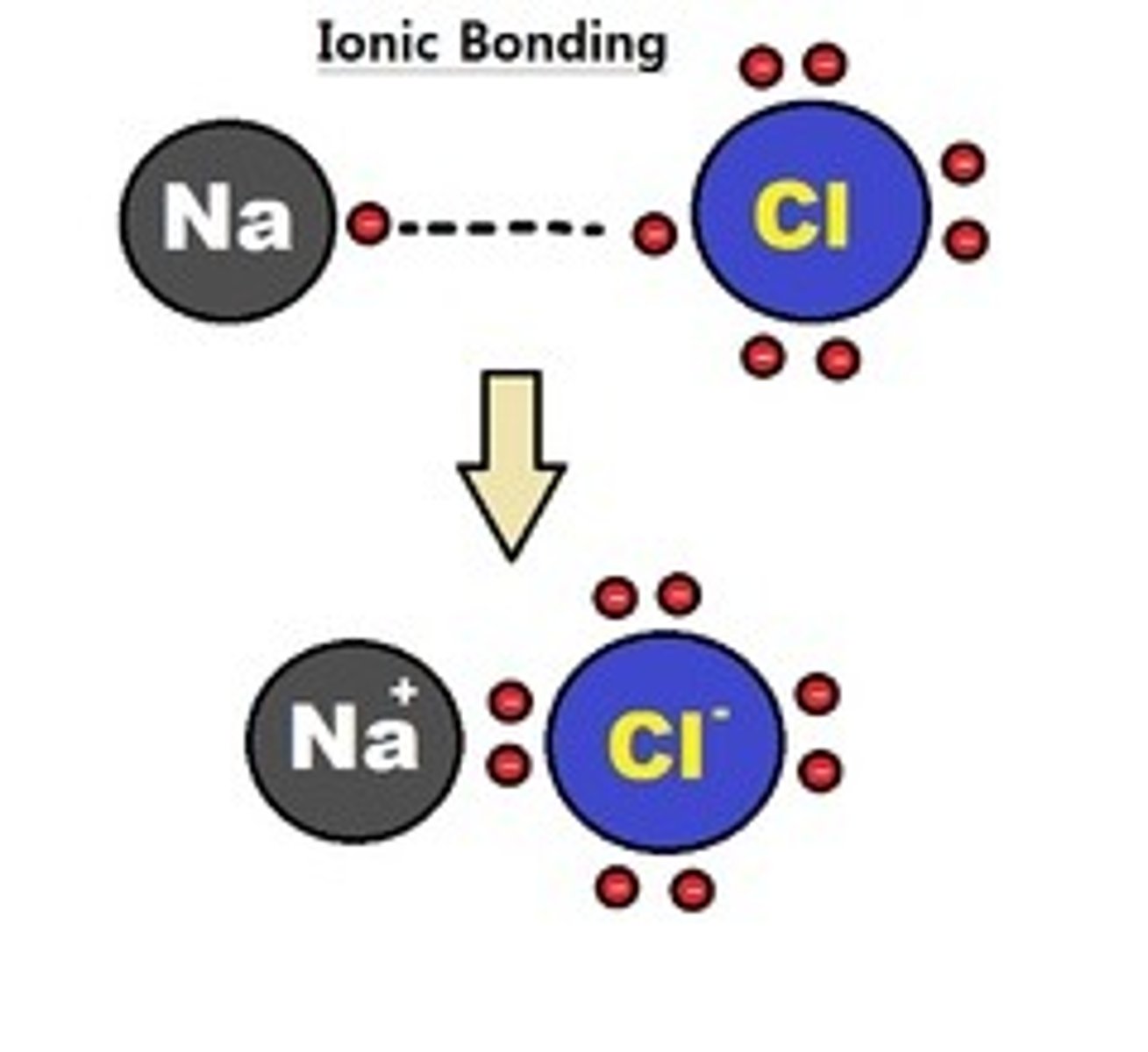
Ion
Electrically charged particle that forms when an atom gains or loses one or more electrons
Example: H+, Na+, K+
Ionic bond
Attraction between oppositely charged atoms, or ions due to transfer of electron from one atom to another
Acid
Any substance that increases the H+ concentration of a solution; acidic solutions have pH values less than 7
The more H+ = more acidic = the lower the pH
Base
Any substance that reduces the H+ concentration of a solution; basic solutions have pH values greater than 7
The lower H+ = more basic= the higher the pH
Concentration
A measure of the amount of dissolved substance contained per unit of volume
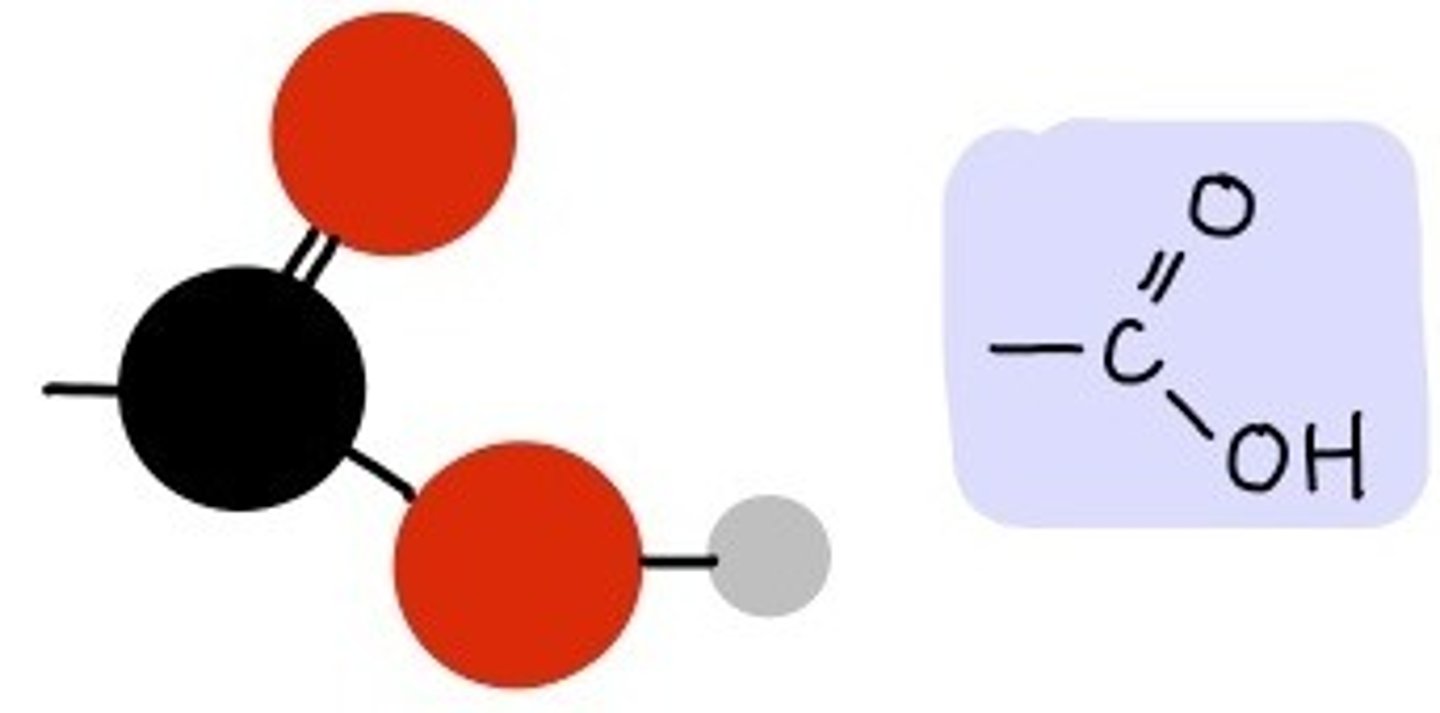
Carboxyl group
"Acid" in amino acids
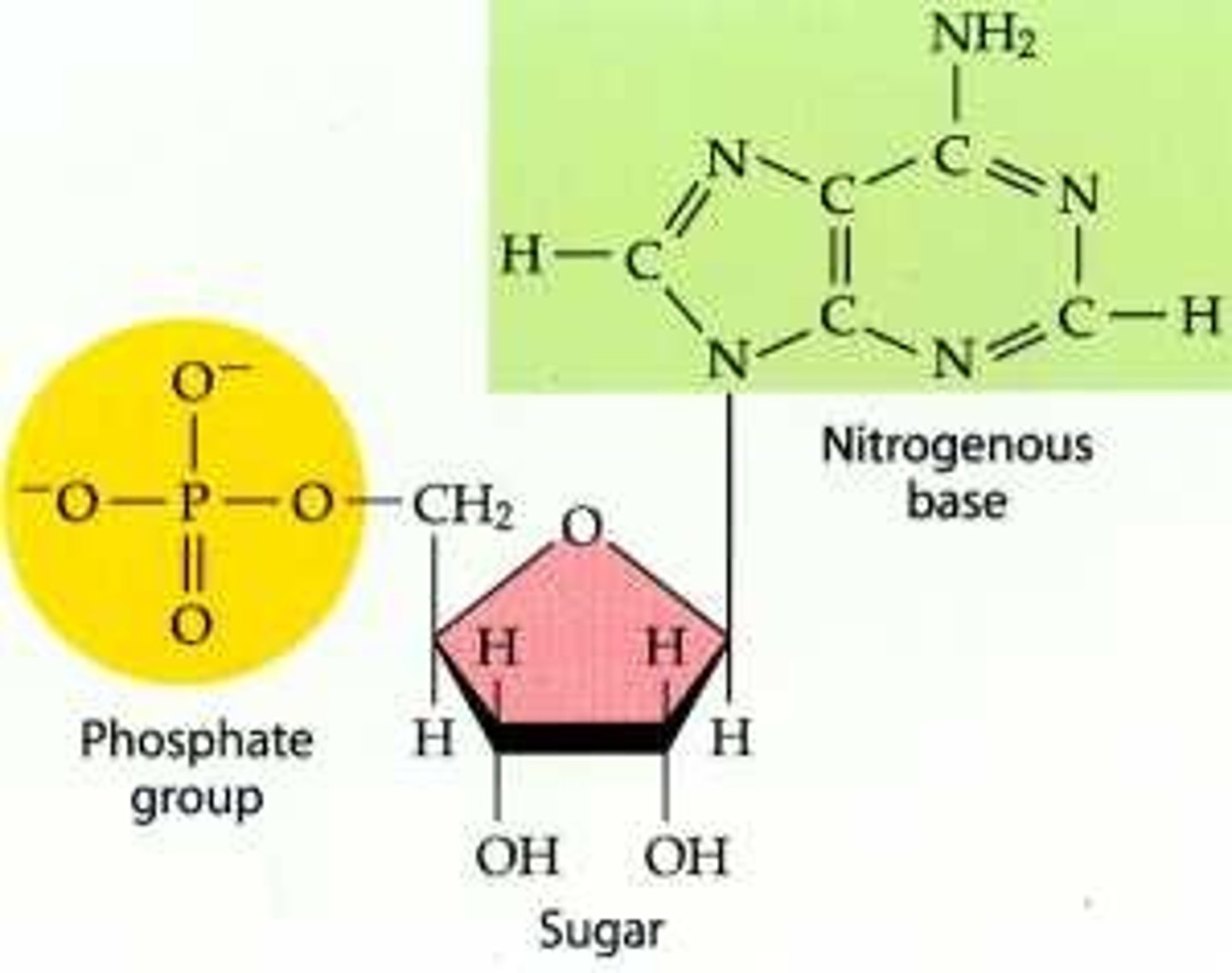
Phosphate group
Chemical group consisting of a phosphorus atom bonded to four oxygen atoms

Amino group
"Amino" in amino acids

Fatty acid
A carboxyl (carboxylic acid) group attached to a long carbon skeleton (hydrophobic)
Competitive inhibitor
Substance that reduces the activity of an enzyme by binding to the active site

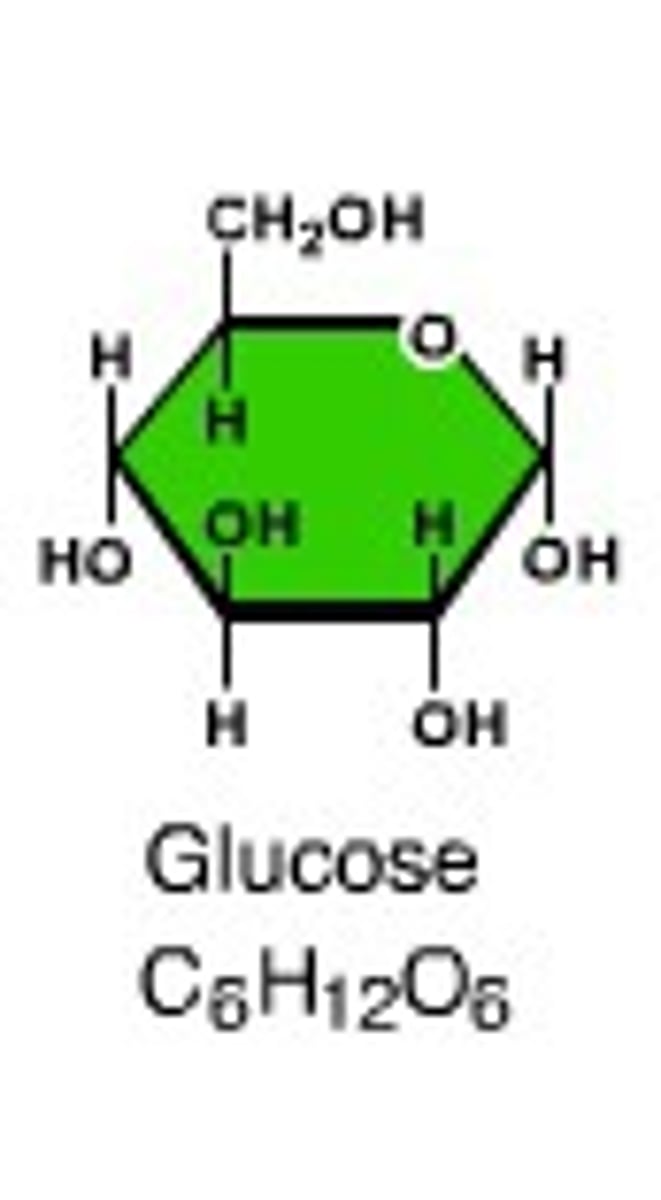
Monosaccharide
"One sugar"; the monomer used to build larger carbohydrates; molecular formula=C6 H12 O6
Examples: glucose, galactose, fructose
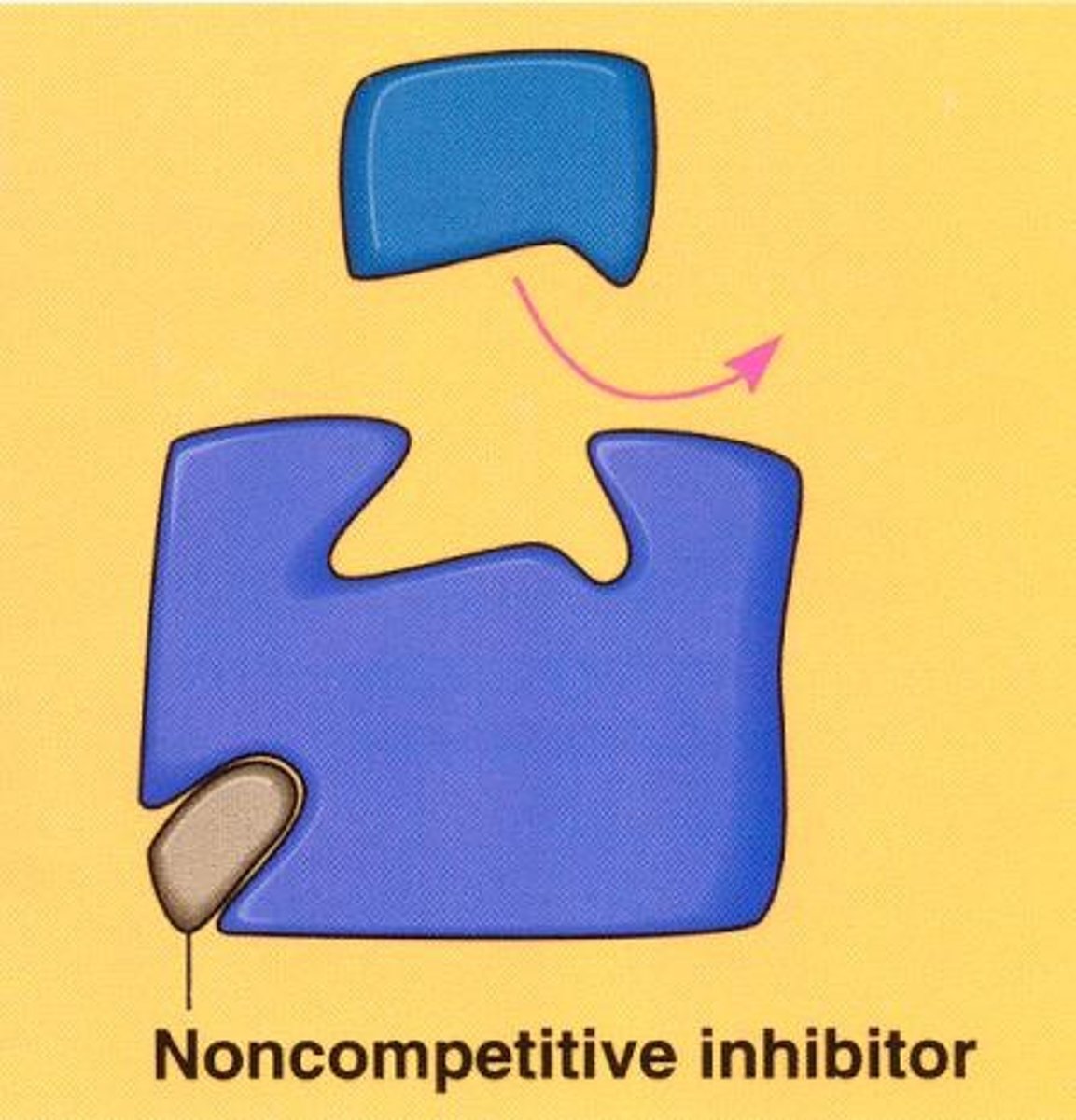
Noncompetitive inhibitor
Substance that reduces the activity of an enzyme by binding to an allosteric site, changing the enzyme's shape so that the active site no longer effectively catalyzes the conversion of substrate to product
Disaccharide
Two monosaccharides joined from a dehydration reaction
Examples: sucrose, lactose, maltose

Amino acid
Compound with an amino group on one end and a carboxyl group on the other end; building block of proteins
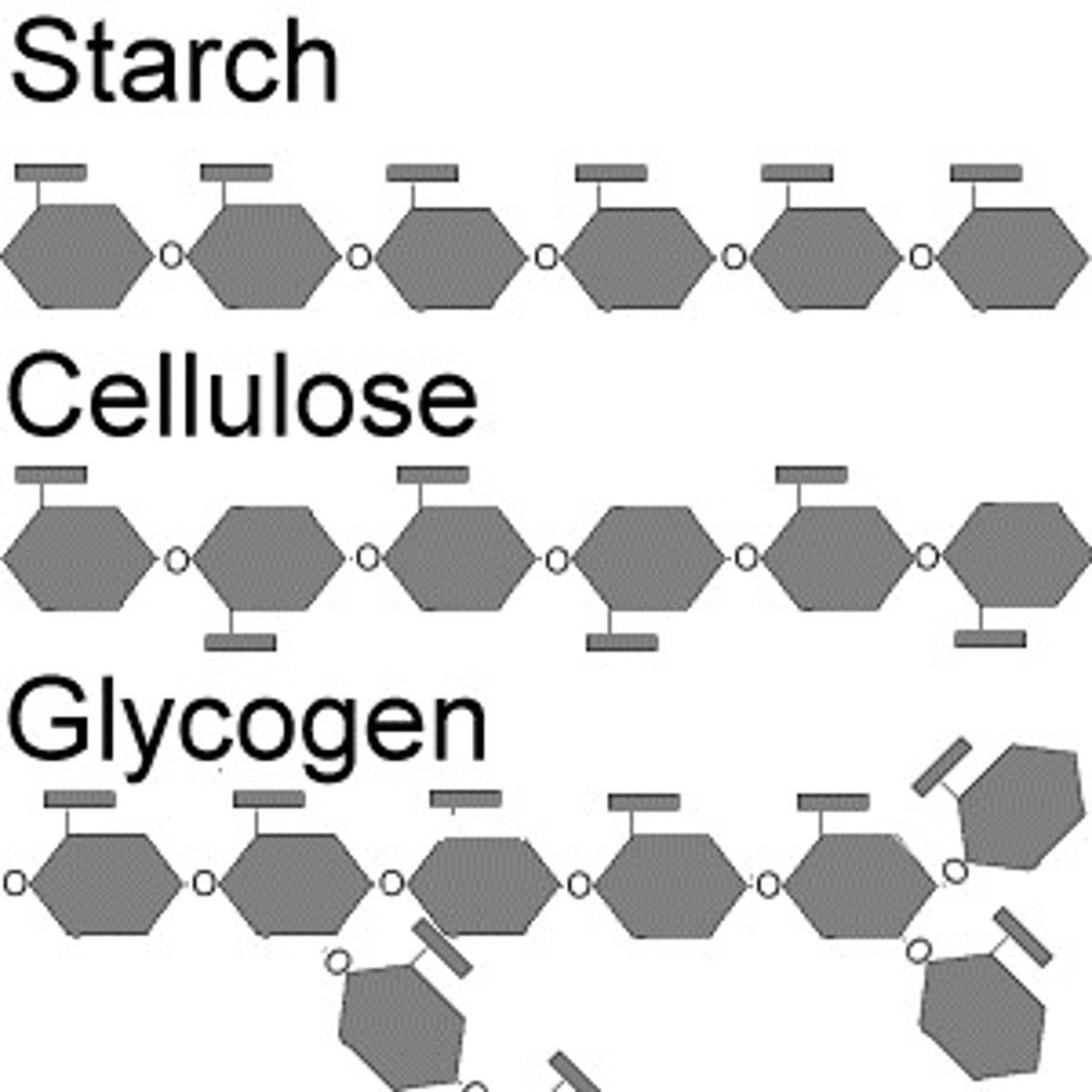
Polysaccharide
Polymers of sugars; have storage and structural roles; ratio of atoms= 1 carbon: 2 hydrogen: 1 oxygen
Ribose
Sugar in RNA
Lipid
Organic compound; common feature= HYDROPHOBIC
Consist mostly of hydrocarbon regions= nonpolar
Examples= fats, phospholipids, and cholesterol
R group
A functional group that defines a particular amino acid and gives it special properties
Deoxyribose
Sugar in DNA
Nucleotide
Monomer of nucleic acids; contains: nitrogen containing base, sugar, & phosphate group
Cohesion
Attraction between molecules of the same substance
Example: water to water
Adhesion
Attraction between molecules of different substances
Example: water to penny
Surface tension
The attractive intermolecular forces at the surface of a liquid
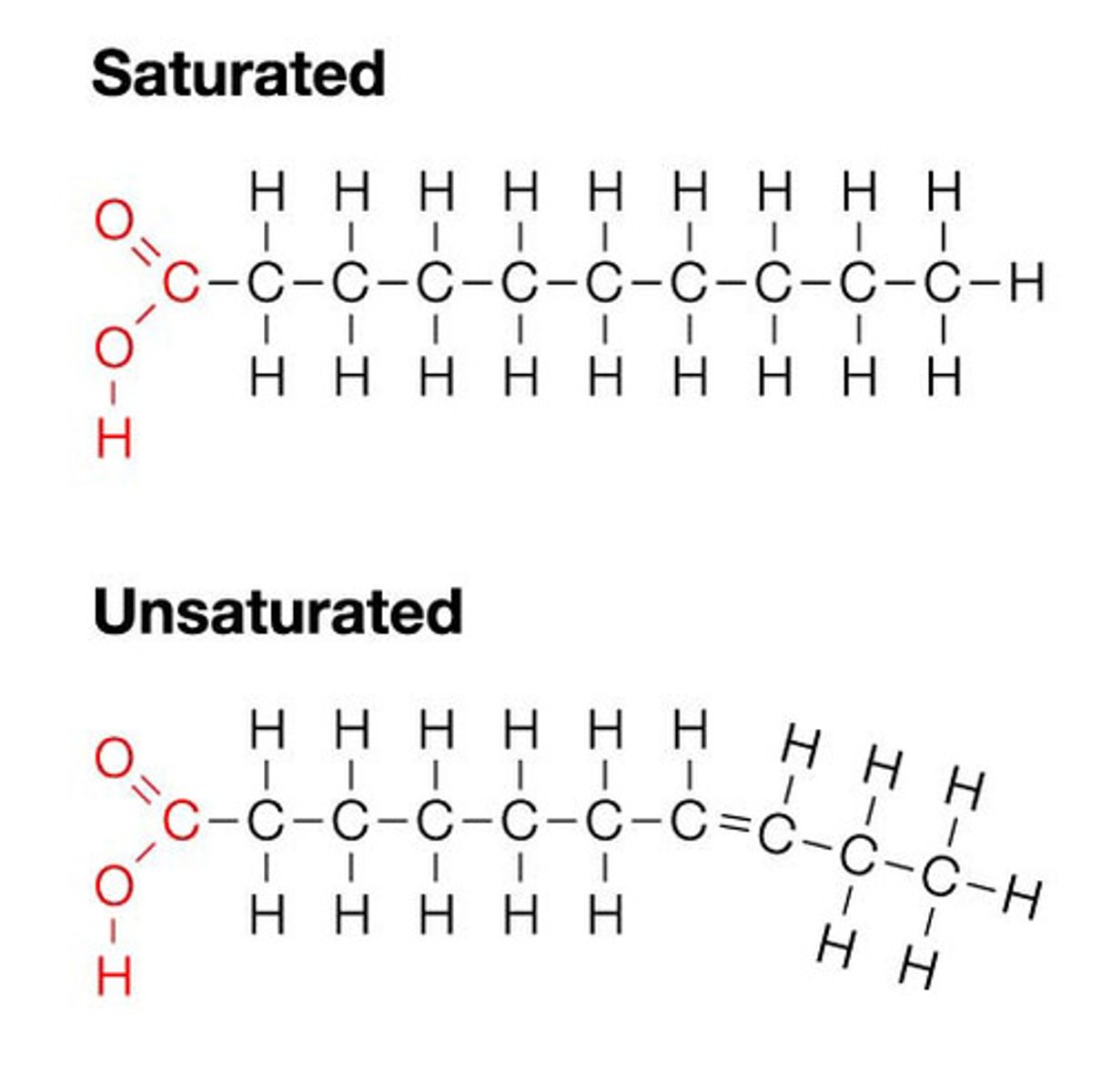
Saturated fatty acid
A fatty acid in which all carbons in the hydrocarbon tail are connected by single bonds, thus maximizing the number of hydrogen atoms that can attach to the carbon skeleton

Unsaturated fatty acid
A fatty acid possessing one or more double bonds between the carbons in the hydrocarbon tail

Prokaryote
Cells that do NOT contain a nucleus or other membrane bound organelles; bacteria
Eukaryote
Cells that contain a nucleus and other membrane bound organelles (ER, GA, lysosome, vacuole, mitochondria, etc)
Fluid mosaic model
Model that describes the plasma membrane as a mosaic (mixture) of protein molecules bobbing in a fluid (moves) bilayer of phospholipids
Vesicle
Membrane surrounded container used to ship or store materials for the cell
Endomembrane system
System of organelles that are continuous or connected through the transfer of vesicles; example: ER --> GA --> plasma membrane
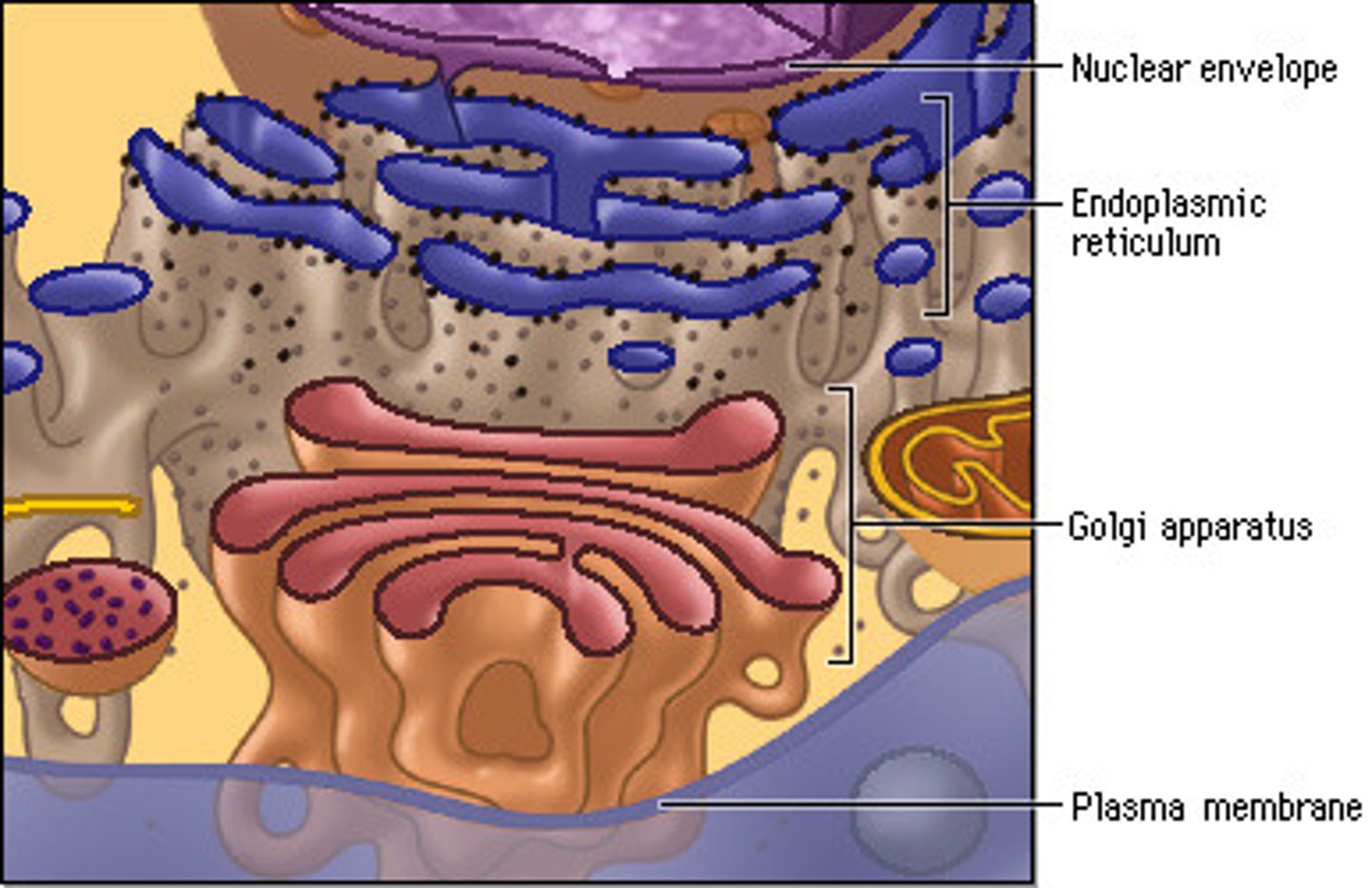
Nucleus
Contains most of the DNA in a eukaryotic cell
Asexual reproduction
The generation of offspring from a single parent, without the fusion of reproductive cells
Plasma membrane
The membrane at the boundary of every cell that acts as a selective barrier, regulating the cell's chemical composition; a phospholipid bilayer with embedded proteins
Selectively permeable
Allows only some substances to cross
Free ribosomes
Uses mRNA to build proteins that remain in cytosol (such as cytoskeleton or motor proteins); NOT attached to ER
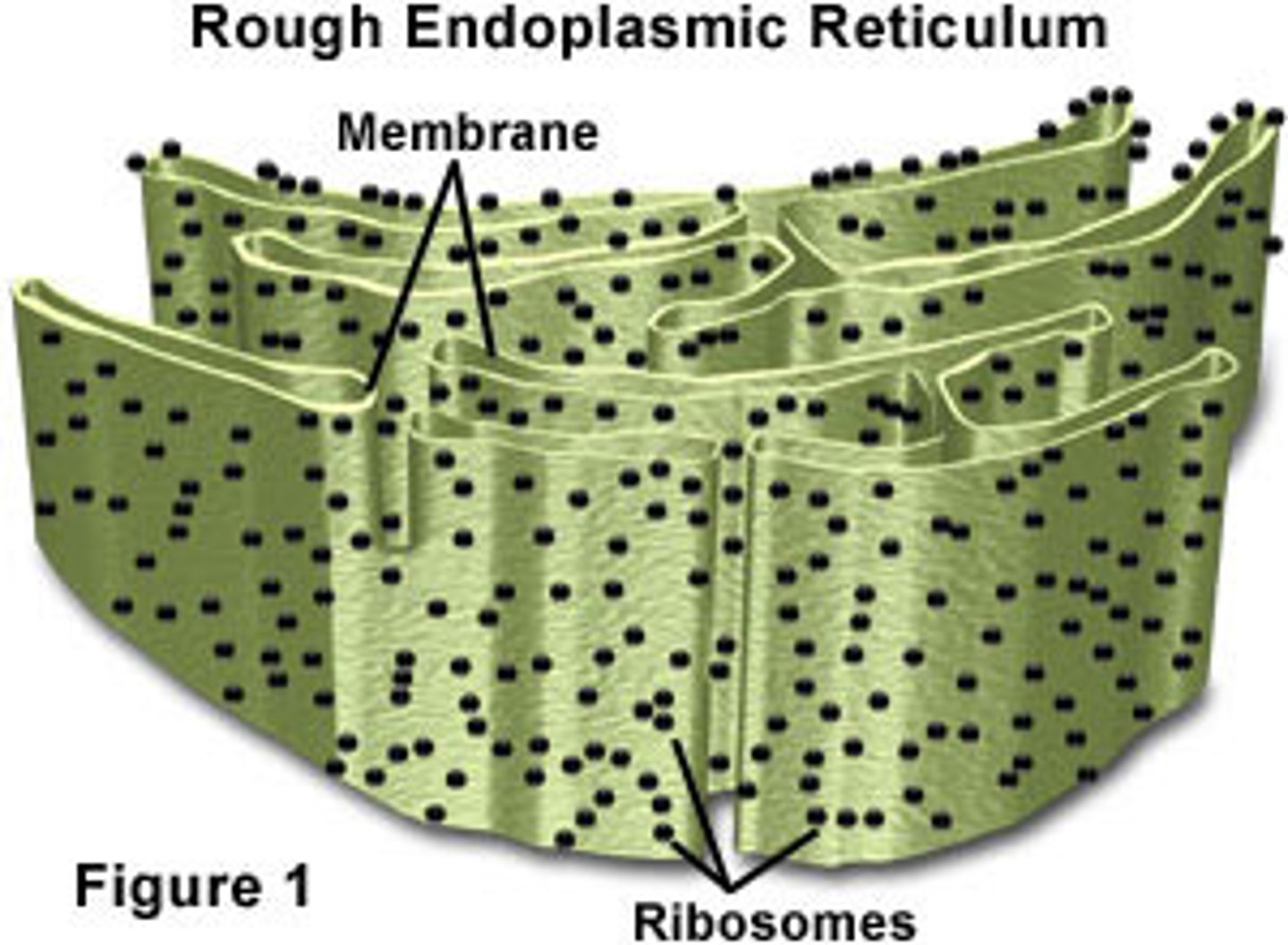
Bound ribosomes
Attached to endoplasmic reticulum; builds proteins that need to enter the endomembrane system (such as excreted from the cell, integrated into the plasma membrane, found inside an organelle)
Smooth ER
Organelle that synthesizes lipids, metabolizes carbohydrates, detoxifies drugs and poisons, stores calcium ions; no ribosomes
Rough ER
Has attached ribosomes that make proteins found in membrane & secreted proteins, distributes transport vesicles, and is a membrane factory for the cell
Golgi apparatus (GA)/ Golgi complex
Modifies certain products of the ER;
Manufactures certain macromolecules;
Sorts and packages materials into transport vesicles, adds molecular "ID tags" for proper delivery

Lysosome
Vesicle containing hydrolytic enzymes that can digest macromolecules
Vacuole
Organelle that store molecules, mainly water
Mitochondria
Organelle that uses sugar & oxygen to generate ATP (process called cellular respiration)
Contains circular DNA, ribosomes, double phospholipid membrane, cristae (folds)

Chloroplast
Organelle that uses light energy to generate oxygen & sugars (process called photosynthesis)
Contains circular DNA, ribosomes, double phospholipid membrane, thylakoids, stroma

Endosymbiosis theory
The theory that mitochondria and chloroplast originated as prokaryotic cells engulfed by host cells
Evidence includes that they both contain:
Phospholipid membrane
Free-floating circular DNA
Ribosomes
Similar size (1-10 mm)
Similar reproduction
Diffusion
The tendency for molecules to spread out evenly into the available space
A substance will move from where it is MORE concentrated to where it is LESS concentrated

Osmosis
Diffusion of water
Lysis
To break; the disintegration of a cell by rupture of the cell wall or membrane; example: when an animal cell is placed in a hypotonic solution
Salinity
Measure of all the salts dissolved in water
Hypertonic solution
Solute concentration is greater than that of the other solution; cell placed in a hypertonic solution will shrink

Hypotonic solution
Solute concentration is less than that of the other solution; cell placed in a hypotonic solution will expand

Isotonic solution
Solute concentration is the same as inside the cell; no net water movement across the plasma membrane

Osmoregulation
The control of solute concentrations and water balance; example: paramecium in freshwater (hypo) have contractile vacuoles to pump incoming water back out
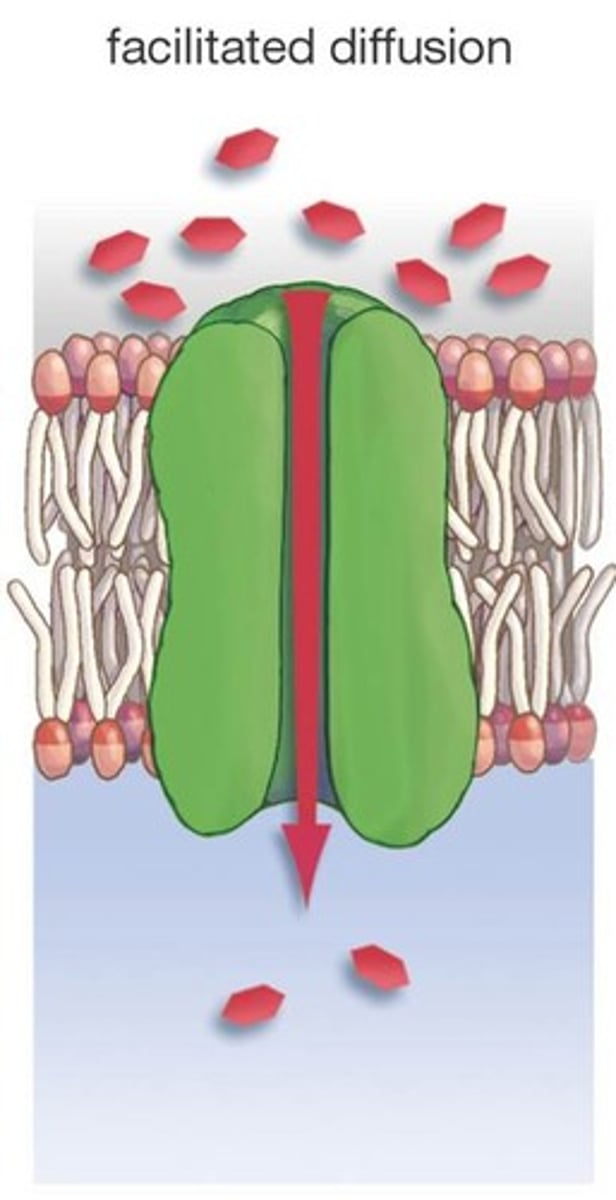
Facilitated diffusion
Movement of specific molecules across cell membranes through protein channels; passive transport process (no energy required); example includes aquaporins
Water potential
Tendency of water to diffuse from one area to another;
= Water's potential energy;
Always moves from regions of higher water potential to regions of lower water potential
Exocytosis
transport vesicles migrate to the membrane, fuse with it, and release their contents
EXO= EXIT

Endocytosis
Cell takes in molecules and particulate matter by forming new vesicles from the plasma membrane
ENDO= ENTER

Phagocytosis
A type of endocytosis in which large particulate substances or small organisms are taken up by a cell
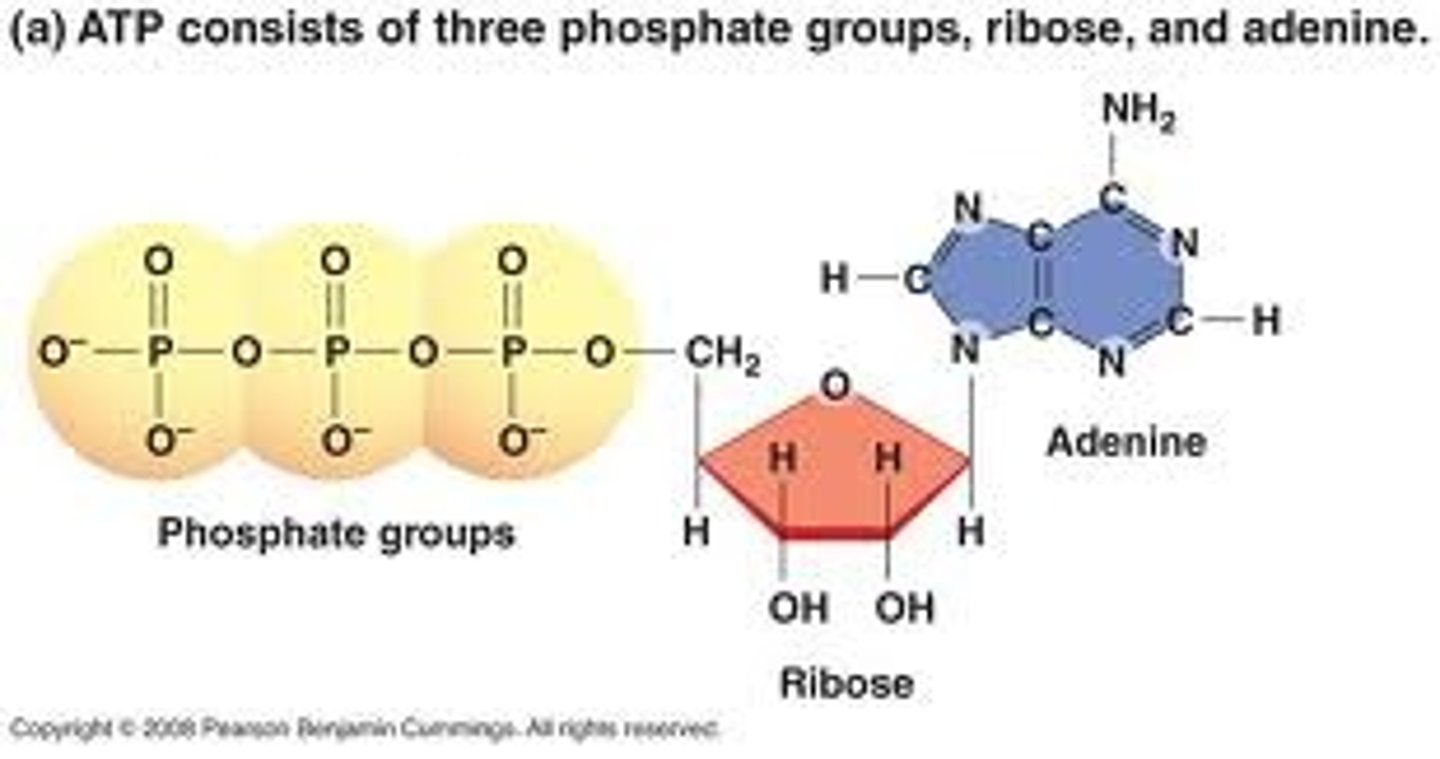
ATP
Adenosine triphosphate; Main energy source that cells use for most of their work
An organic molecule called adenosine attached to a string of three phosphate groups
Active transport
Moves substances against their concentration gradients
Requires energy, usually in the form of ATP
Sodium-potassium pump
A carrier protein that uses ATP to actively transport sodium ions out of a cell and potassium ions into the cell
Result= Higher Na+ concentration outside cell; positive charge outside, negative inside cell
Solute
A substance that is dissolved in a solution.
Osmolarity
Total concentration of all solute particles in a solution
Homeostasis
Process by which organisms maintain a relatively stable internal environment
Proton pump
An active transport protein in a cell membrane that uses ATP to transport hydrogen ions against their concentration gradient, generating a membrane potential in the process.
Metabolism
Sum total of an organism's chemical reactions
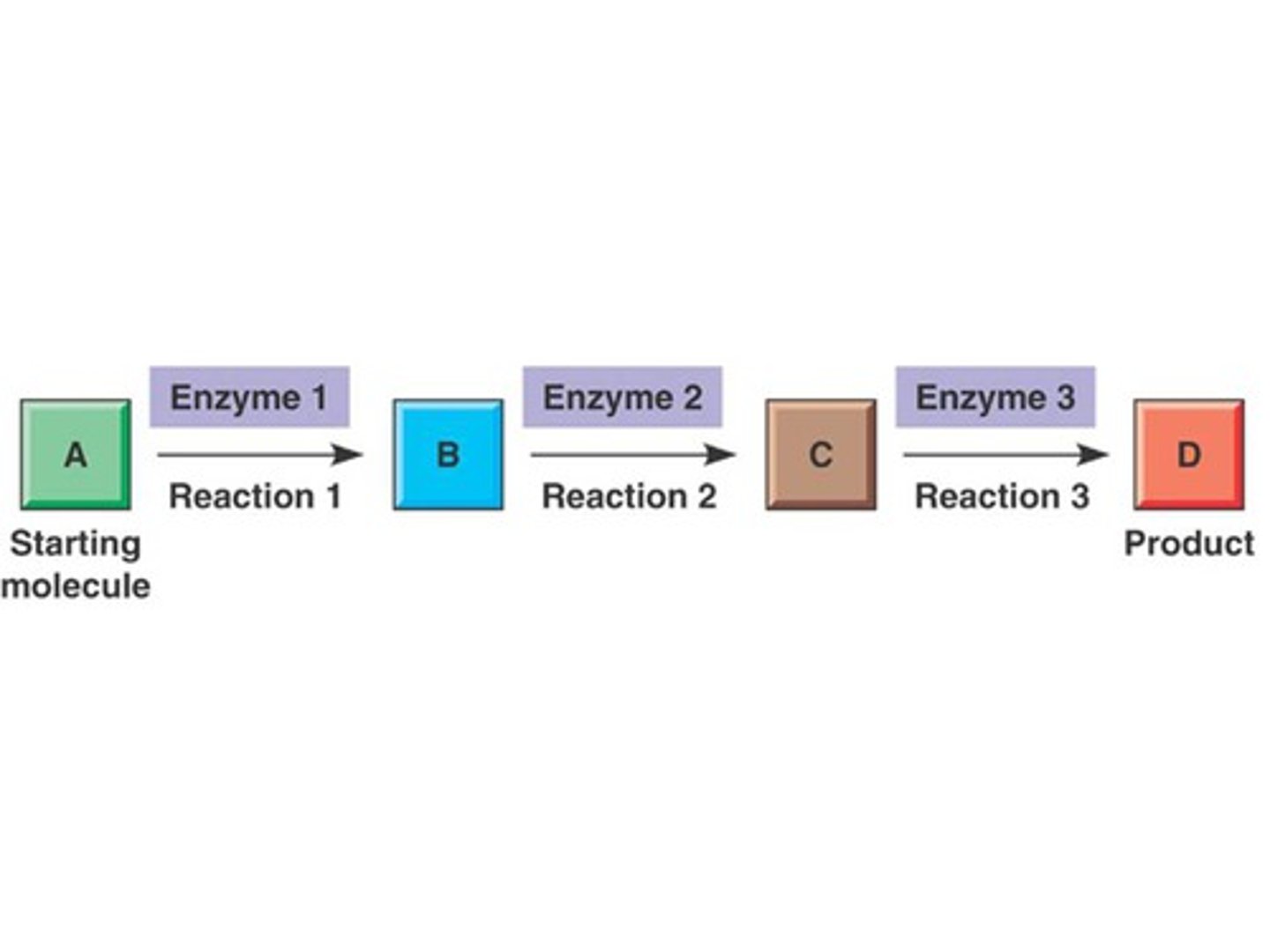
Metabolic pathway
A series of chemical reactions that are catalyzed by enzymes; two major pathways are cellular respiration and photosynthesis
1st law of thermodynamics
Energy can be transferred and transformed, but it cannot be created or destroyed
2nd law of thermodynamics
During every energy transfer or transformation, some energy is unusable and is often lost as heat
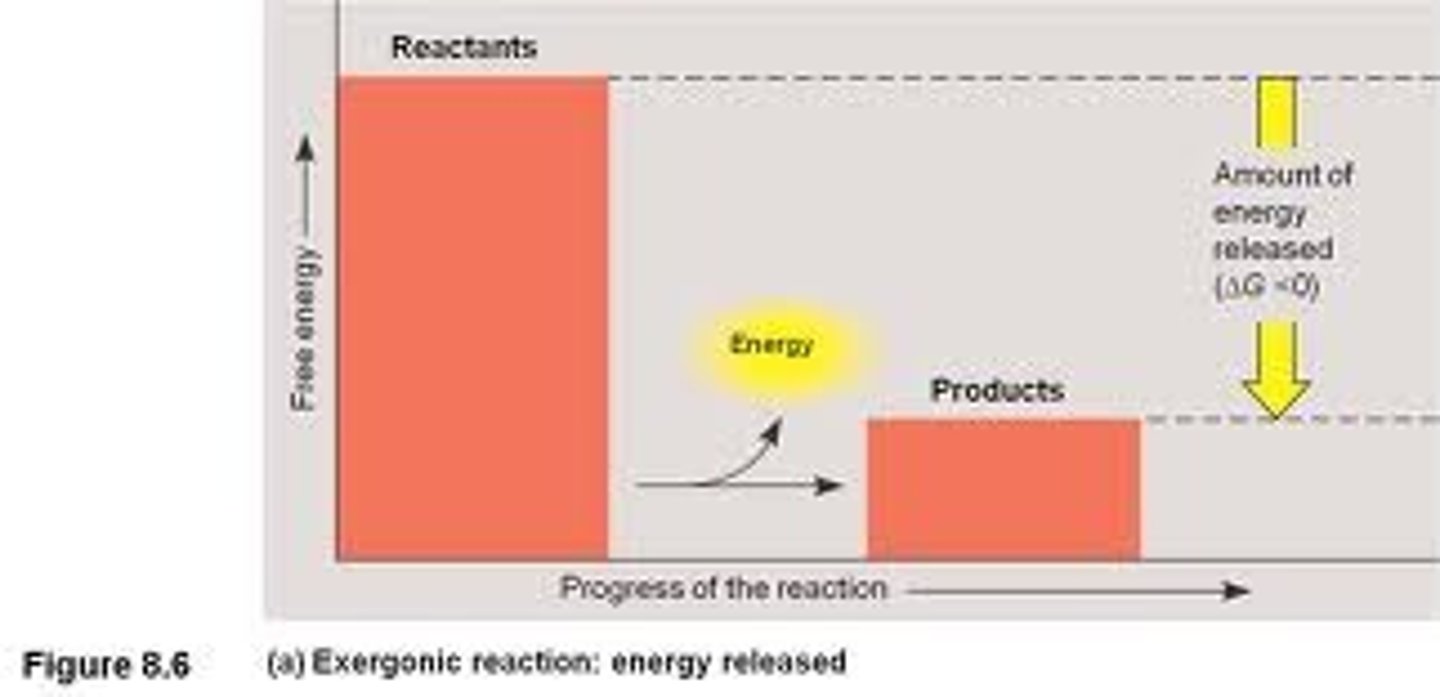
Exergonic reaction
Breaks molecules apart and releases energy; is spontaneous (no energy input)
Endergonic reaction
Builds molecules and requires energy input; is not spontaneous

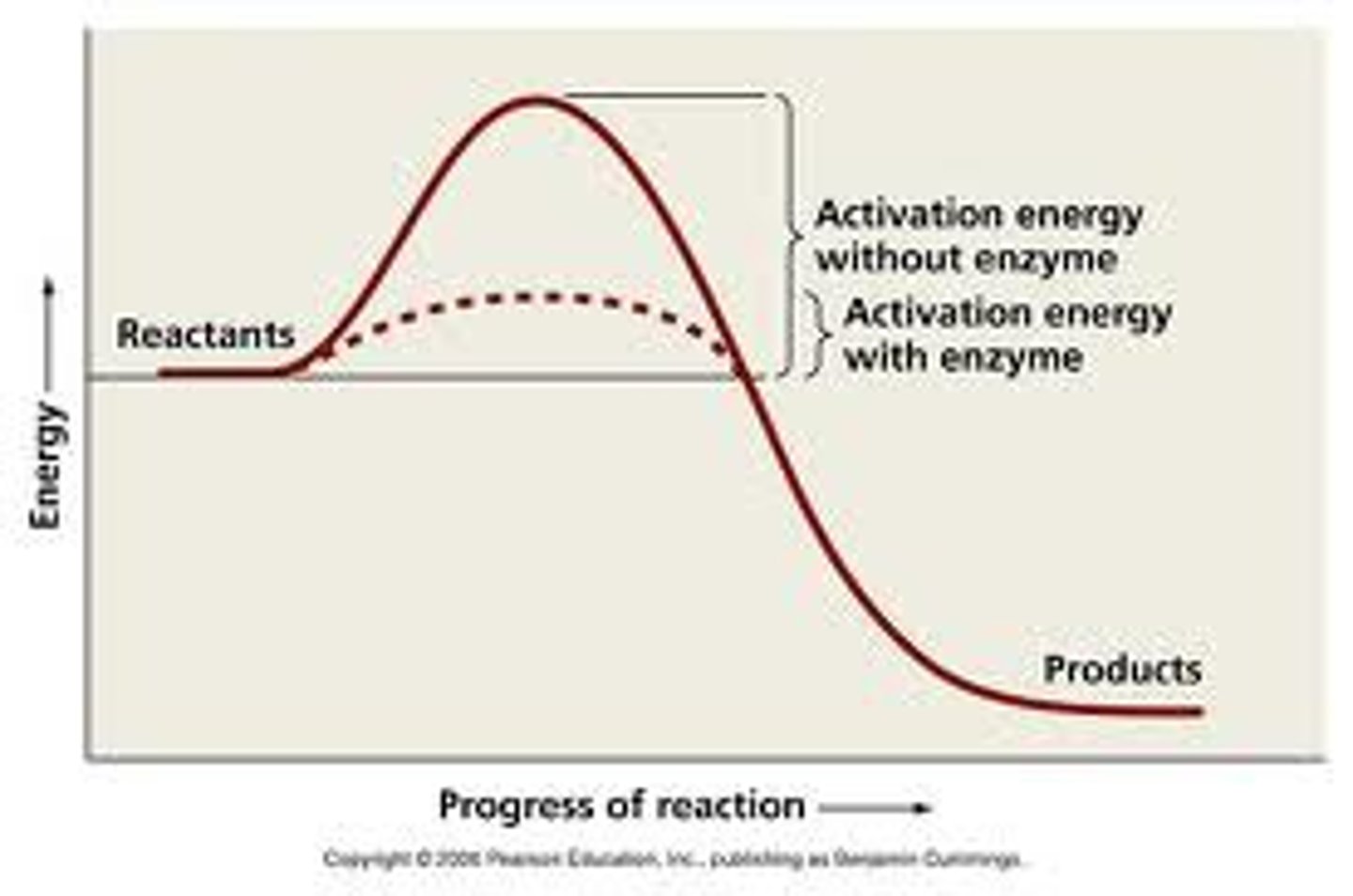
Activation energy
Initial energy needed to start a chemical reaction
Activation energy diagram
Feedback inhibition
The end product of a metabolic pathway shuts down the pathway

Adenosine triphosphate (ATP)
An organic molecule called adenosine attached to a string of three phosphate groups; primary energy-transferring molecule in the cell
Phosphorylation
Removing phosphate group from ATP and adding it to another molecule to activate it