12 alkenes
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
sigma and pi bond in an alkene
sigma: head on overlap of orbitals (strong)
pi: side on overlap of p-orbitals (weak)
stereoisomerism (e/z, cis trans)
same structural formula, but different arrangement of the atoms in space
E/Z occurs when there’s C=C bond, and 2 different groups
stereoisomers around double bond arises because rotation of the DB is restricted and the groups attached to C atoms are fixed relative to each other
cahn ingold prelog rules
the atoms attached to each C atom is a DB are given priority by atomic number
if the groups of higher priority are on the same side of the DB, the compound is a Z isomer
if groups of higher priority are diagonal, it is a E isomer
electrophile
atom/group accepts a electron pai (partial + charge)
heterolytic bond fission
a atom/group takes both shared electrons
bond enthalpy
take place when breaking by homolytic fission 1 mol of given type of bond in the molecules of a gaseous species
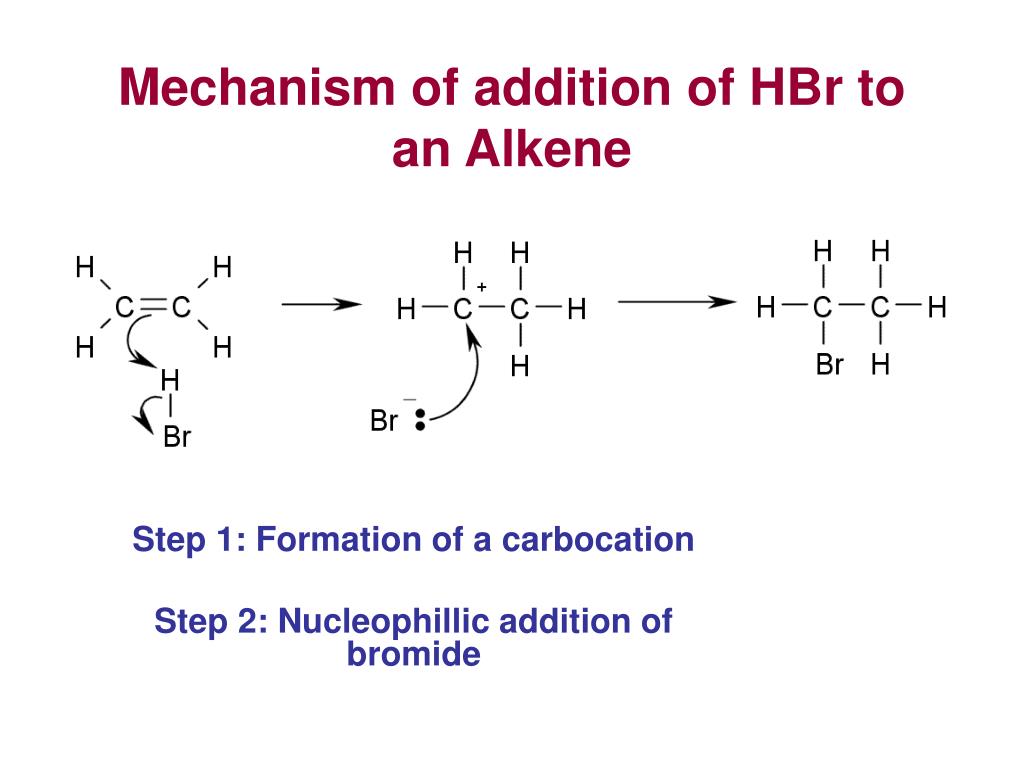
addition of HBr to alkenes - hydrogen halide
alkene + hh (HCl) → haloalkanes (2 possible products)
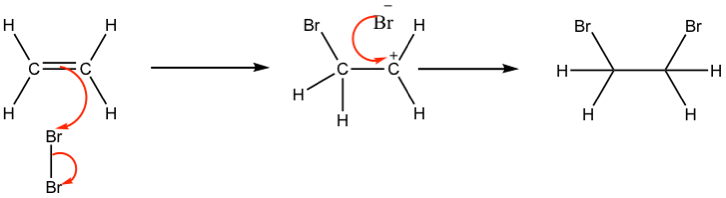
addition of Br2 to alkenes - halogenation
alkene + chlorine/bromine RTP → haloalkane
hydration of alkenes
alkenes + steam + phosphoric acid catalyst H3PO4 → major and minor product of alcohol
the mechanism of addition to alkenes
electrohphillic addition
markownikoff’s rule of stability of cation intermediate
the addition of HX to an unsymmetrical alkene, the hydrogen atom attaches to the carbon that already has more hydrogens, forming the more stable carbocation intermediate.
addition polymerisation of alkenes
unsaturated alkene molecules undergo addition polymerisation to produce saturated chains with no DB
high temps and pressure with long chains of alkenes = polymerisation
eg.polyethene : toys, shampoo bottles (most common polymer)
sustainability of waste polymers
recycle: can be reused, reducing polymers on landfills
disposing: non biodegradable, kills marine life
PVC recycling: hazardous - releases hydrogen chloride (toxic)
use as fuel: can be incinerated to be heated into steam and generates turbines
feedstock recycling: reclaims monomers