CBNS101 Lecture 09 Enzyme-coupled receptors
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Enzyme-coupled receptors
-- have a cytosolic domain that has enzyme activity, or directly associates with an enzyme
-- single-pass transmembrane proteins that mostly dimerize/autophosphorylate to activate
Receptor tyrosine kinases (RTKs)
- Phosphotyrosines serve as docking sites via SH2 / PTB domains on signaling proteins
- Ras GEF (Sos) -> Ras -> MAP kinase cascades
- PI 3 kinase -> PI(3,4,5)P3 -> Akt (PH domain) -> growth/survival
- Rho GTPases -> cytoskeletal rearrangements
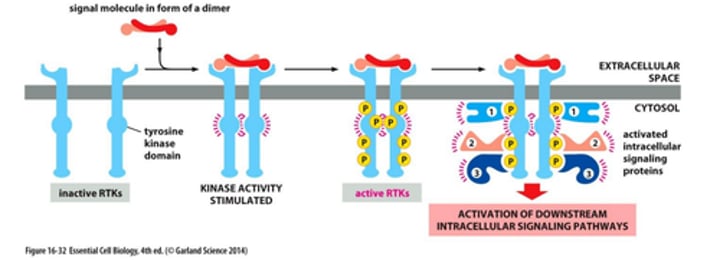
explain how RTKs is activate by dimerization / autophosphorylation
Ligand binding on the extracellular side leads to activation in the following way:
- Unbound RTKs = inactive, monomer conformation
- Ligand binding causes dimerization of the receptors
- Dimerization brings the two cytoplasmic tyrosine kinase domains into close proximity, so they phosphorylate each other, which leads to further activation
- Activated domains further phosphorylate themselves, providing docking sites for downstream signaling protein
explain how RTKs is activate by dimerization / autophosphorylation in the case of a EGF receptor
In some cases (EGF receptor), dimerization brings the two kinase domains into an asymmetrical dimer, where one "activator" domain turns on a "receiver" domain, which then phosphorylates both domains
what are Phosphorylated tyrosines on RTKs and how do Proteins dock to RTKs?
docking sites for intracellular signaling proteins
via SH2 (Src homology 2) or PTB domains (phosphotyrosine binding)
what are they types of proteins that dock to RTKs
-- enzymes: phospholipase C-γ (PLCγ); (like PLCβ activated by GPCRs)
-- soluble proteins: ex., tyrosine kinases (ex., Src) that phosphorylate other signaling proteins
-- phosphoinositide 3- kinase (PI3-kinase)
-- inhibitory, via negative feedback loop
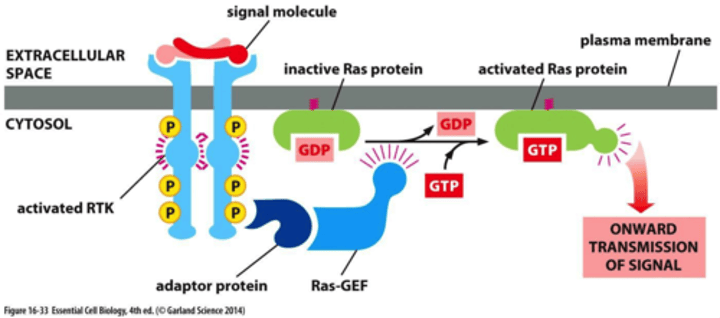
how are signals transmitted downstream after docking of proteins?
- monomeric GTPase Ras
-Ras and Rho families relay signals from cell-surface receptors
- Ras and Rho family GTPases are activated by Ras/Rho GEFs downstream of RTKs: Ras is activated by Sos (Son-of-sevenless), recruited by Grb2 to RTKs
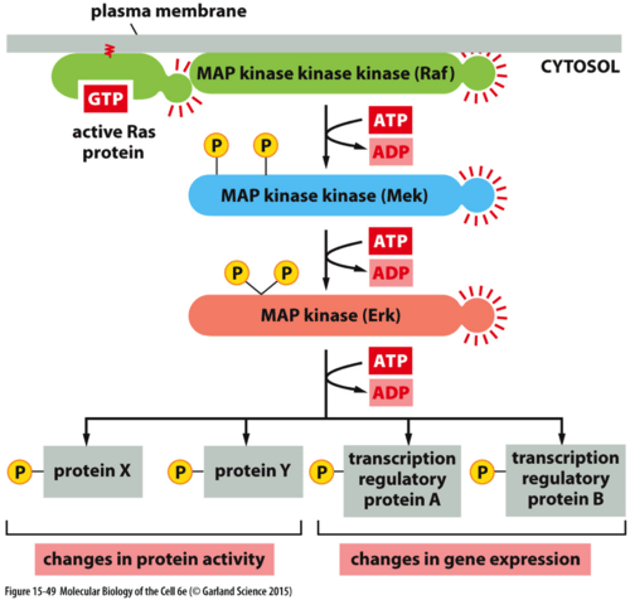
What does Ras activate?
Ras activates a MAP kinase signaling module/cascade:
1) Ras activates MAP kinase kinase kinase (MAP KKK, Raf)
2) MAP KKK phosphorylates MAP KK (Mek)
3) MAP KK phosphorylates MAP kinase (Erk)
4) MAP kinase phosphorylates target proteins important for cell proliferation,
How are scaffold proteins important for MAP kinase modules
Scaffold proteins separate different MAPK modules
Different scaffold proteins are used to assemble different sets of kinases, avoiding unwanted cross-activation
T/F Ras is a key oncogene
T.
30% of tumors have hyperactive mutant forms of Ras
~1/3 of all cancers have mutations in Ras genes. what is the mutation? what does it effect?
- Most mutations occur in just one of three aminoacids
- Mutant Ras loses ability to hydrolyze GTP -> always in the GTP-bound, ON state
- Incoming signals (ex., EGF) are no longer necessary, Ras signals downstream regardless of growth factor presence
- Results in uncontrolled cell growth, etc
How are Activated RTKs and Ras turned off
- Tyrosine phosphorylations are removed by tyrosine-specific protein phosphatases
- Ras is inactivated by Ras GAPs
what experiment demonstrates Ras inactivation? explain the procedure.
FRET
Ras is fused to a YFP protein; GTP with a fluorescent label is introduced
After receptor activation, FRET signal from YFP to the red dye on GTP is observed transiently
Ras is frequently mutated in human cancers
~1/3 of all cancers have mutations in Ras genes
Most mutations occur in just one of three aminoacids
Mutant Ras loses ability to hydrolyze GTP -> always in the GTP-bound, ON state
Incoming signals (ex., EGF) are no longer necessary, Ras signals downstream regardless of growth factor presence
Results in uncontrolled cell growth, etc
how Ras activates a MAP kinase signaling module
- A key signaling cascade activated by Ras is the mitogen-activated protein kinase module/pathway (MAP kinase module)
- A conserved cascade of three Ser/Thr kinases that is used in different signaling contexts:
- Ras activates MAP kinase kinase kinase (MAP KKK, Raf in mammals)
- MAP KKK phosphorylates MAP KK (Mek in mammals)
- MAP KK phosphorylates MAP kinase (Erk in mammals)
- MAP kinase phosphorylates target proteins important for cell proliferation, including immediate early genes
how Scaffold proteins separate different MAPK modules
- Humans have 12 MAPKs, 7 MAPKKs and 7 MAPKKKs, that work in at least five separate MAPK modules transmitting different signals with different outcomes
- Different scaffold proteins are used to assemble different sets of kinases, avoiding unwanted cross-activation
What do Rho family GTPases do?
they transmit signals to the cytoskeleton
specifically: they transmit signals from cell-surface receptors to regulate actin and microtubule cytoskeleton function - control cell shape, polarity, motility and adhesion
who are the main players for how Rho GTPases transmit signals to the cytoskeleton
Rho, Rac and Cdc42
Rho GTPases transmit signals to the cytoskeleton. explain how it does so in this Ex.,: during motor neuron axon outgrowth / migration to its muscle target
-- Ephrin signaling proteins on the surface of "incorrect" cells activate Eph RTKs
-- RTK signaling activates the Rho GEF ephexin, which activates RhoA
-- Activated RhoA regulates target proteins -> actin reorganization, collapse of axon growth cone -> repelling axon from growing in this direction
Explain how RTKs signal through membrane-bound PI(3,4,5)P3
- PIPs (phosphoinositides), particularly PI(3,4,5)P3, serve as signaling molecules
- Phosphoinositide 3-kinase (PI 3-kinase) is activated by RTKs (and GPCRs!) and adds a phosphate
- PTEN phosphatase dephosphorylates PIPs
- PI(3,4,5)P3 serves as a docking site for signaling proteins with pleckstrin homology (PH) domains - ex., Akt
PI-3-kinase / Akt is a major survival/growth pathway
- Pro-survival/growth signals activate RTKs - > PI 3 kinase -> PI(3,4,5)P3 -> Akt
- Activated Akt phosphorylates many targets, inactivating them
- The end result is pro survival/growth: ex., Akt phosphorylates and inactivates Bad, turning on Bcl2
what does PI3K / Akt activate? what does it control?
mTORC1 complex, a key regulator of growth
-- it controls growth by responding to growth factor signaling (RTKs) and amino acid availability through Rheb and Rag GTPases
-- it is a protein kinase that phosphorylates many downstream proteins
Tyrosine-kinase-associated receptors
function similar to RTKs, except the kinase domain is a separate cytoplasmic protein
Receptors for: antigens (BCR, TCR), interleukins / cytokines, integrins, hormones
activation via dimerization
Some work through the Src family (SH2 and SH3 domains)
How associated Cytokine receptors activate the JAK-STAT pathway
1) cytokine receptor family binds cytokines, some hormones
2) after ligand binding and dimerization, the associated Janus (JAK) kinases phosphorylate/activate themselves and tyrosines on the receptors
3) Signal transducers and activators of transcription (STATs) are phosphorylated by JAKs, dissociate and form dimers, which act as transcription activators in the nucleus
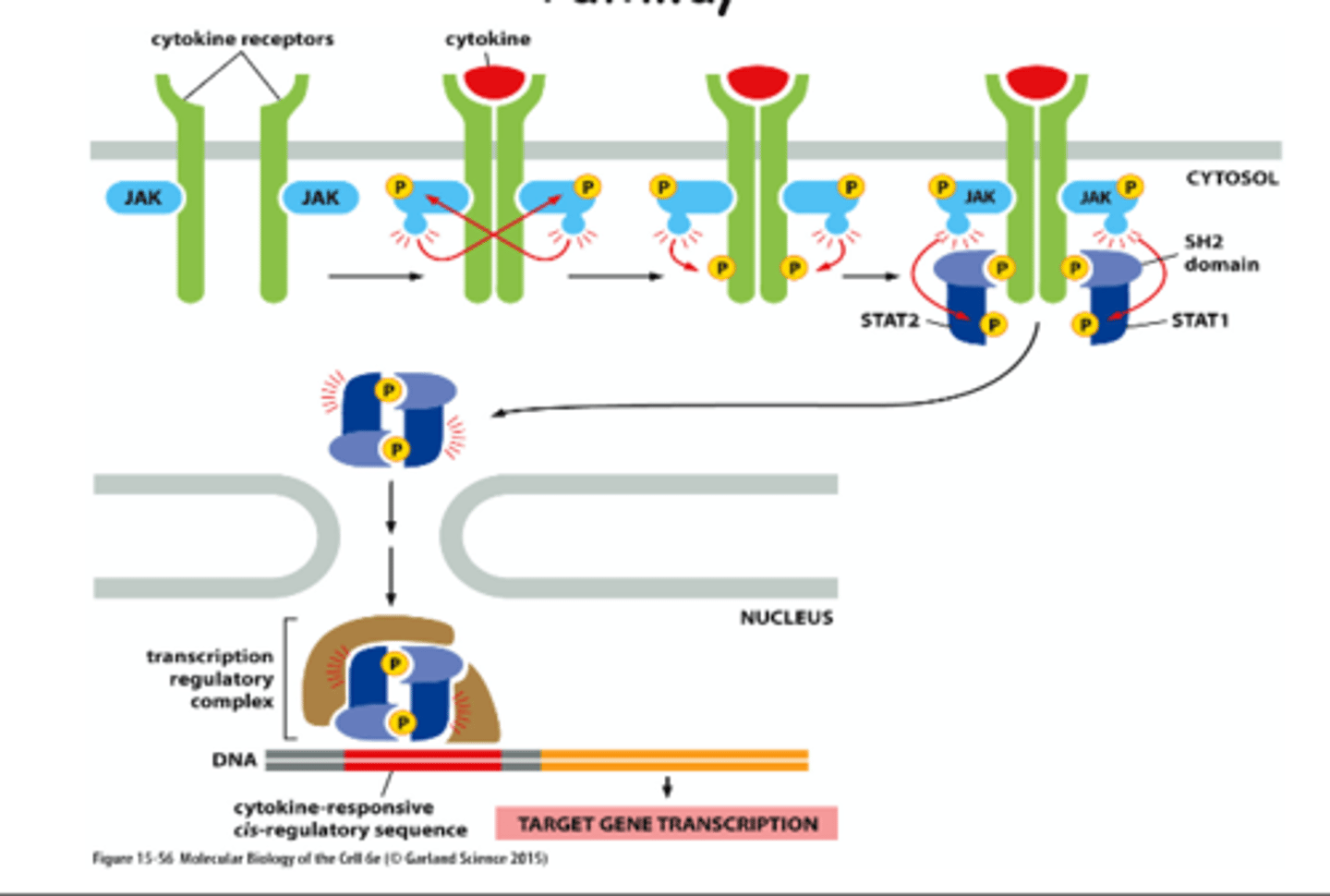
Tyrosine kinase associated receptors
Cytokine receptors -> JAKs -> STATs -> transcription
explain how transforming growth factor-β (TGFβ) proteins activate receptor Ser/Thr kinases
1) The TGFβ are dimeric signaling proteins that bind and cause multimerization of receptor Ser/Thr kinases
2) The receptors phosphorylate themselves and Smad proteins, which regulate transcription in the nucleus
3) Endocytosis of TGFβ receptors = further activation (in early endosomes) and/or degradation (through caveolae)
Receptor serine/threonine kinases
TGFβ signaling to Smads, transcriptional activation