Action potential generation
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
reaching threshold
- when a depolarizing stimulus arrives, the open probability of sodium channels will increase.
- the open channels pass unitary currents that build into an inward macroscopic current.
voltage threshold
- if the inward current is large enough, it will trigger a positive feedback loop and fire a spike.
complex behavior of threshold
it is dynamic and can change, even disappearing at times
experimental determination of threshold
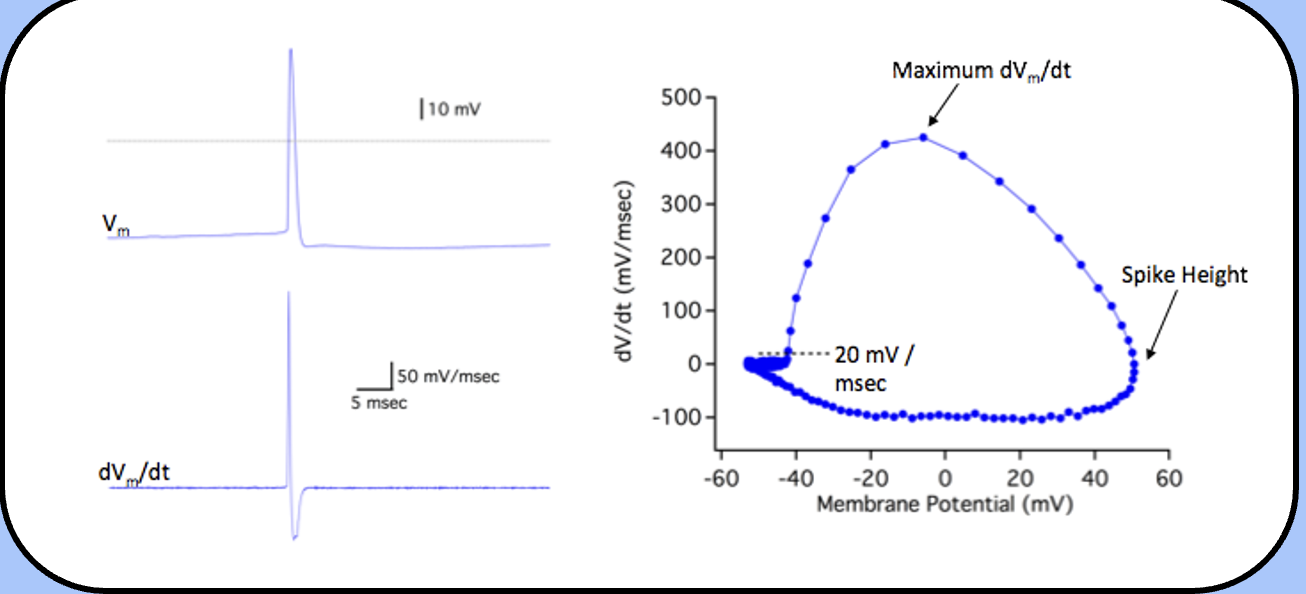
find the potential where the membrane potential increases faster than 20 mV / msec.
the first step is to differentiate the action potential—the dV/dt,
phase-plane analysis
the voltage where the dV/dt crosses 20 mV / msec is usually defined as threshold, the “top of the circle” is the maximum dV/dt, and the right-most side of the circle is the spike height.
the balancing act determining threshold
inward currents pushing to Ena and outward currents pushing to Ek
action potentials begin when the inward current temporarily dominates the membrane current, and end when the restoring outward currents bring the system back into balance
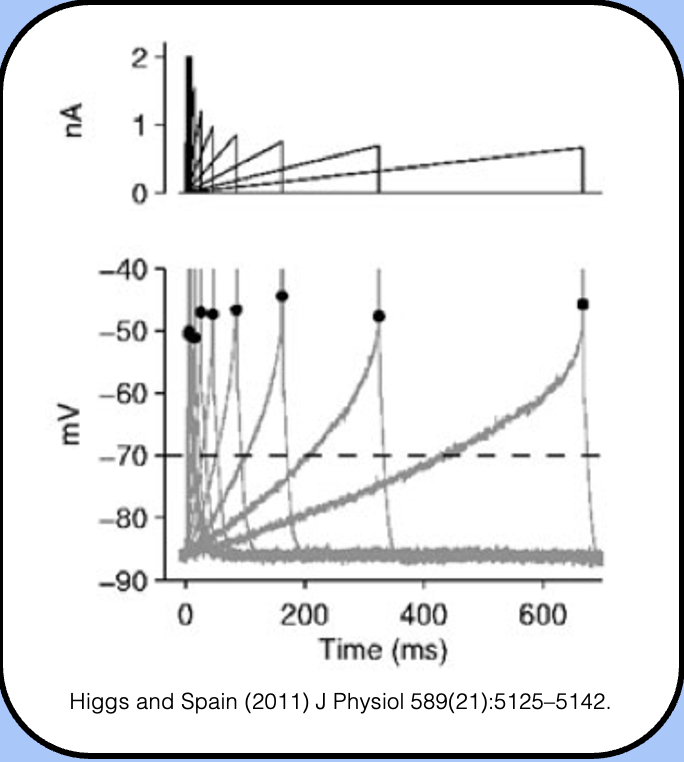
ramp stimuli
ramp currents as stimuli.
- here, the experimenter controls the rate of current injection as well as its final magnitude.
- interestingly, slower ramp speeds are associated with a depolarization of threshold—and if the ramp is too slow, it might be impossible to elicit an action potential.
slow depolarization—sodium channels
- the large sodium currents that flow during fast voltage ramps (400 mV/s) all but disappear during slow voltage ramps.
- this is because slow ramps give the channels enough time to inactivate—sodium channels do not have to open before they inactivate
slow depolarization—potassium channels
here, we see an outward current flowing during the voltage ramp.
because many potassium channels don’t inactivate (or inactivate slowly) outward currents become larger as the ramp potential increases.
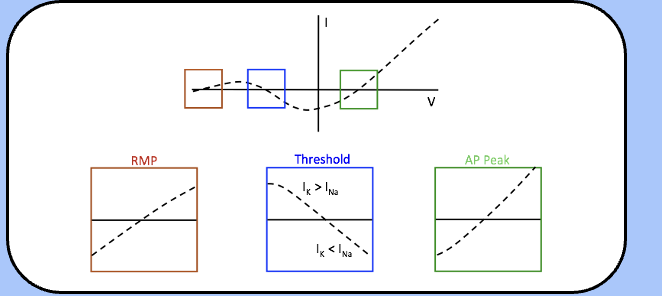
Noble’s model of excitation
here, we assume that m is extremely fast and reaches steady-state almost instantaneously, and that the instantaneous potassium current is linear.
the total membrane current Im is equal to INa + IK—note the three important physiological parameters associated with an Im of zero.
Noble’s model of excitation—passive domain
- here, the slope of the I–V plot is positive, which means that this is a stable point—any small perturbation of the membrane potential away from this point will elicit a current response that brings the membrane potential back to this point.
- this means that RMP is a stable point—in the language of dynamical systems, this is called a stable attractor.
stable attractor
RMP is a stable point
Noble’s model of excitation—active domain
the slope of the I–V plot near the second crossing is negative, and any stimulus that depolarizes the membrane potential beyond this point will elicit an action potential.
just below the threshold, Im is outward, and will drive the membrane potential back to RMP—beyond the threshold, there is an inward current that will further depolarize the membrane potential toward the third crossing through positive feedback.
Noble’s model of excitation—Action potential
- once we pass threshold (also called a bifurcation), the membrane potential races toward the third crossing—a theoretically stable point at the peak of the action potential.
- this point is really not stable, however, because potassium channels will activate and drive the membrane potential back toward RMP. (this is where the model fails bc the upward trend will be very fast)
threshold accomodation
- as sodium channels inactivate and potassium channels activate (i.e., we allow n and h enough time to change) there will be more IK and less INa.
- this shifts the whole I–V relationship upward, thereby raising the threshold.
- if the depolarization is slow enough, threshold disappears.
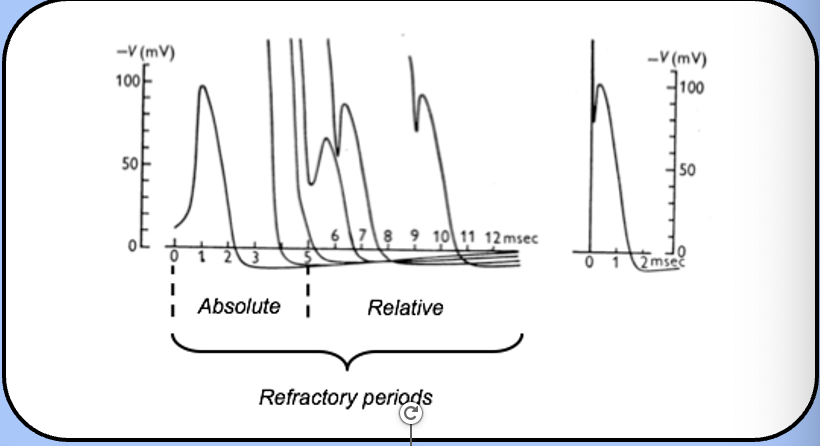
refractory periods
- when an action potential occurs, it sets in motion a series of events that affect the neuron’s ability to fire a second action potential.
absolute refractory period
the period when eliciting a second action potential is impossible.
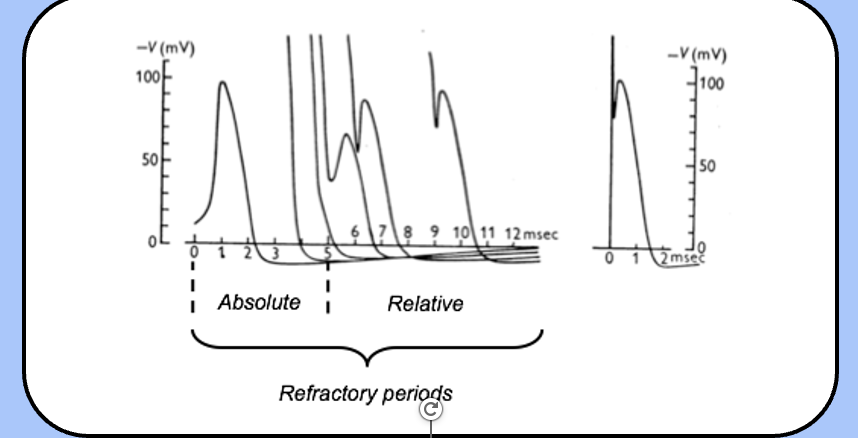
relative refractory period
the period where a second action potential can be generated, but only with a stronger stimulus
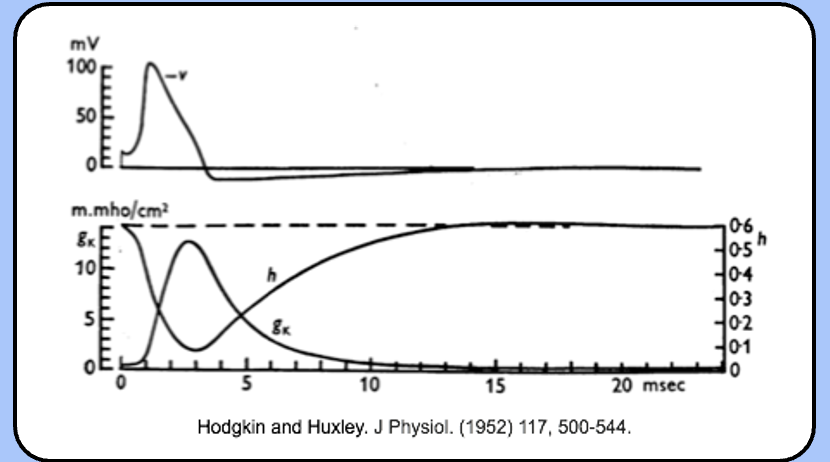
events of the refractory period
not surprisingly, the refractory periods are defined by the values of h and n.
immediately after the action potential, h is low, so there are few available sodium channels, and n is high, so IK is large.
over time, h recovers and n decreases, as the neuron approaches normal excitability.
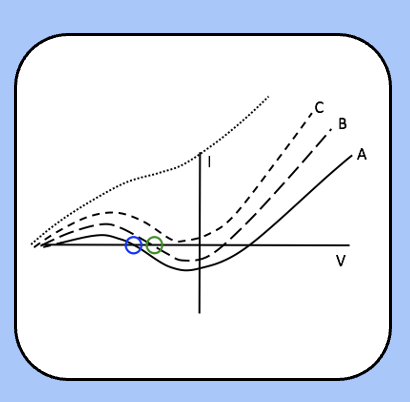
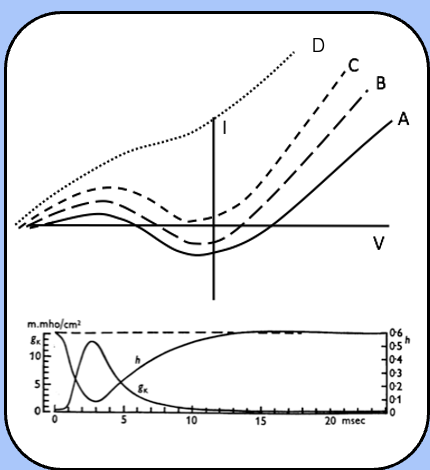
threshold during the refectory period
immediately after the action potential (the absolute refractory period) Im jumps up to curve D or C, where there is no threshold.
over time, h recovers and n decreases, so the Im follows curve B, where their threshold exists, but is higher than usual (the relative refractory period).
finally, the h and n reach their steady-state values as Im follows curve A (normal excitability).
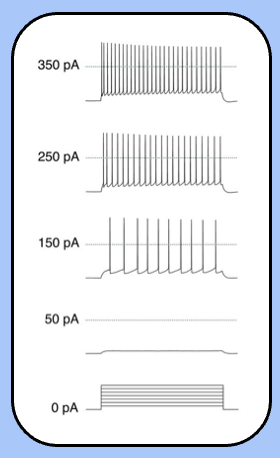
action potential trains
neuron responds to current steps by firing action potentials throughout the stimulus.
it might seem obvious, but this neuron is encoding magnitude of the current step into action potential frequency
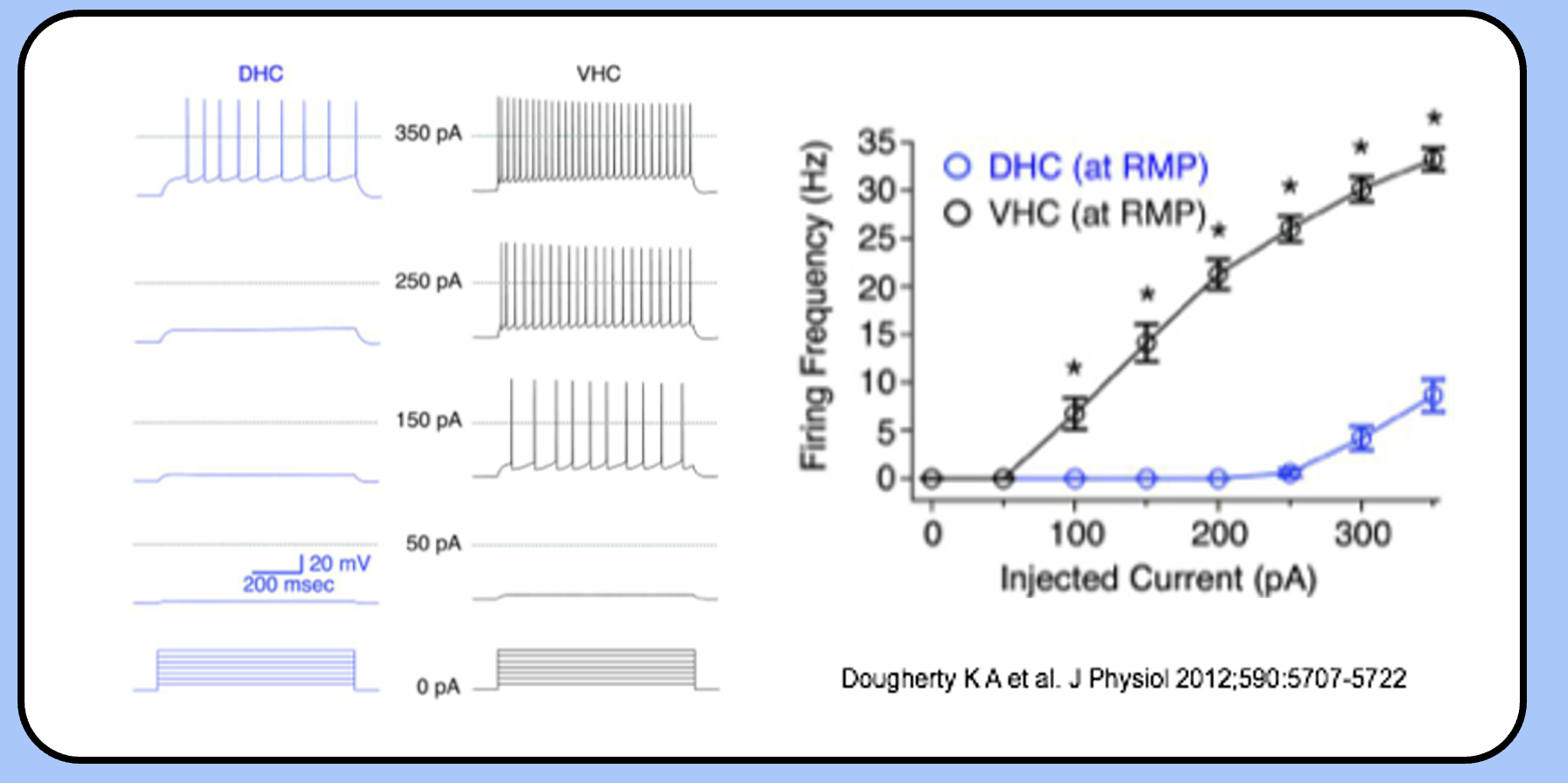
the firing frequency vs current relationship
- one simple way to describe the firing properties of a neuron is to plot the firing frequency against the injected current (the F–I plot).
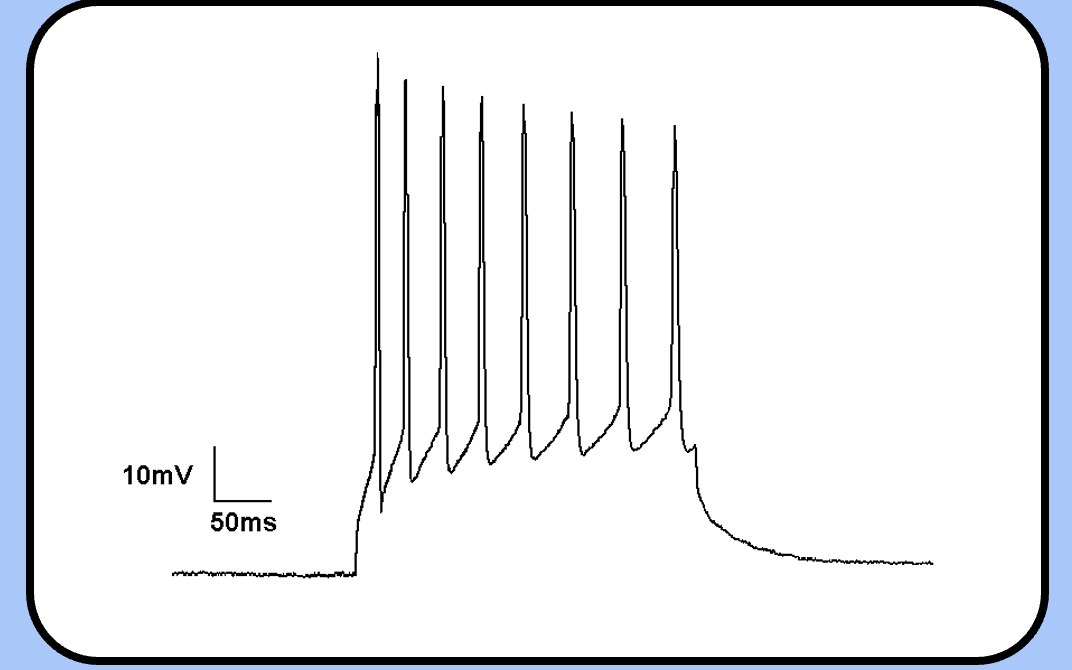
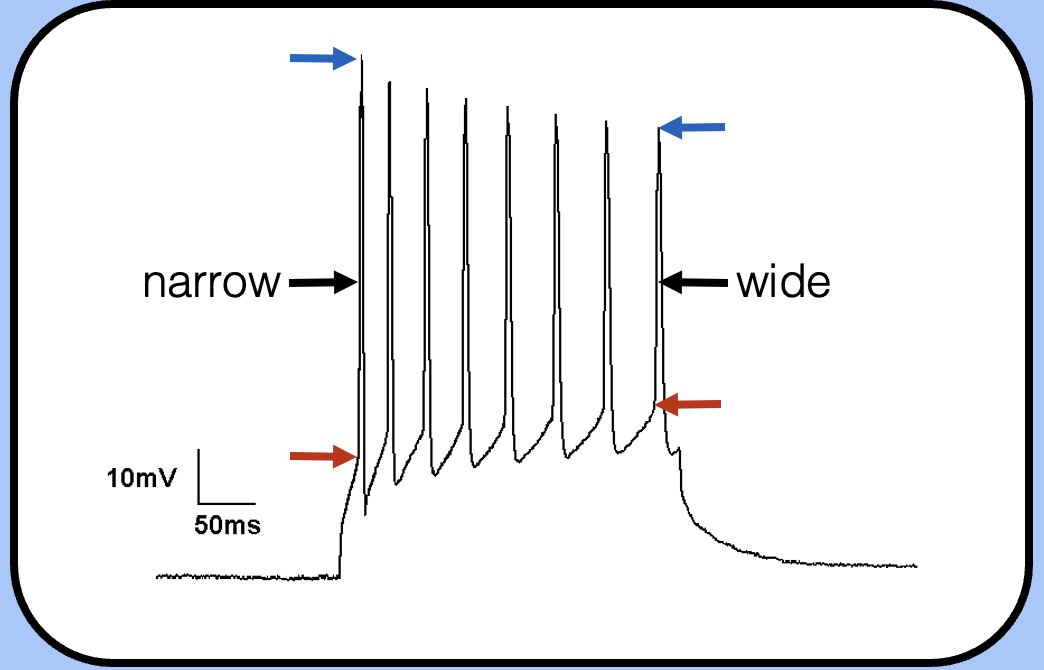
changes during an action potential train
—the threshold has increased, the spike height is shorter, and the action potential is wider (this is harder to see though).
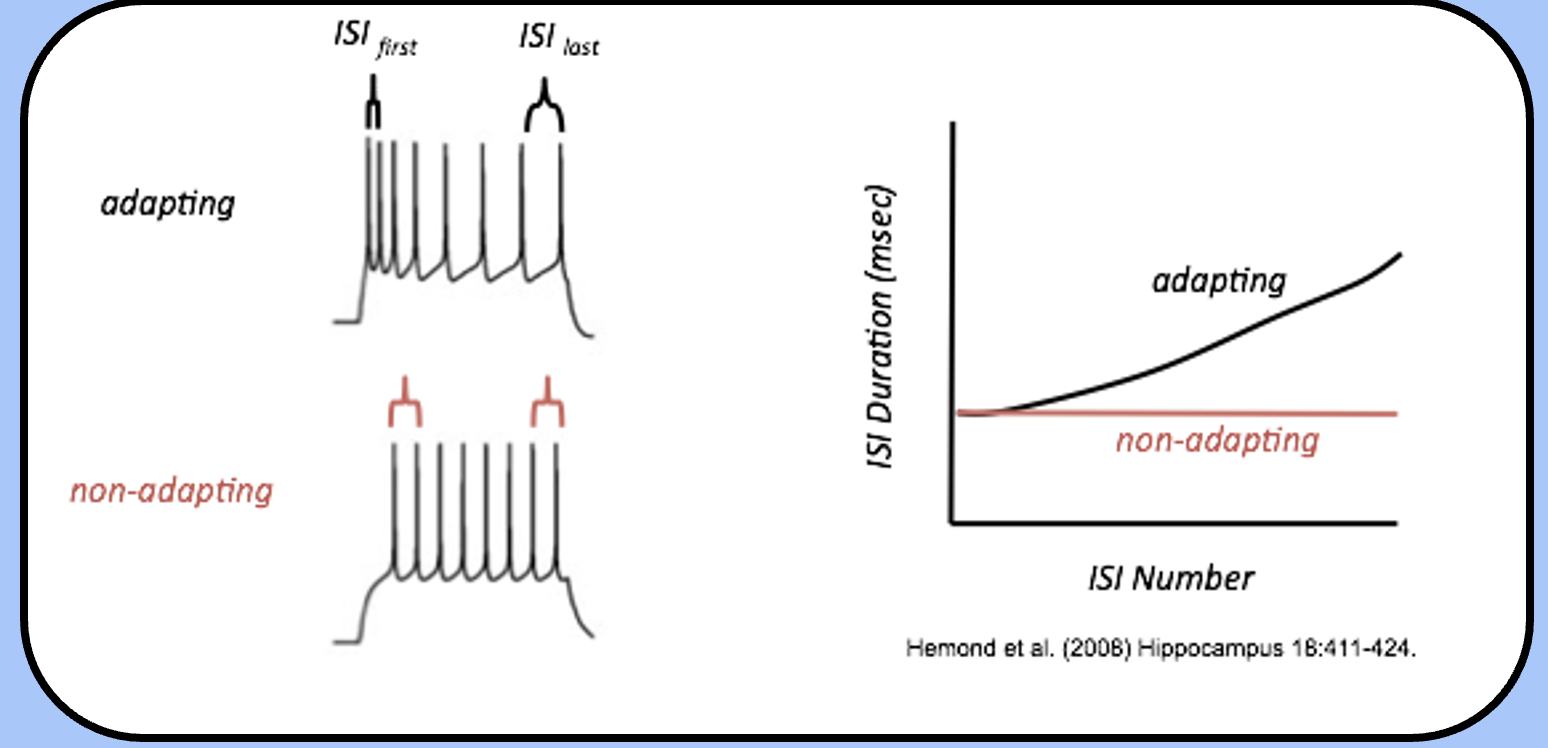
spike frequency adaptation
another property that we often see during action potential trains is spike frequency adaptation (SFA).
trains from an adapting neuron will “space themselves out,” during the train, so that the interspike interval (ISI) between the last two spikes is larger than the ISI between the first two spikes.
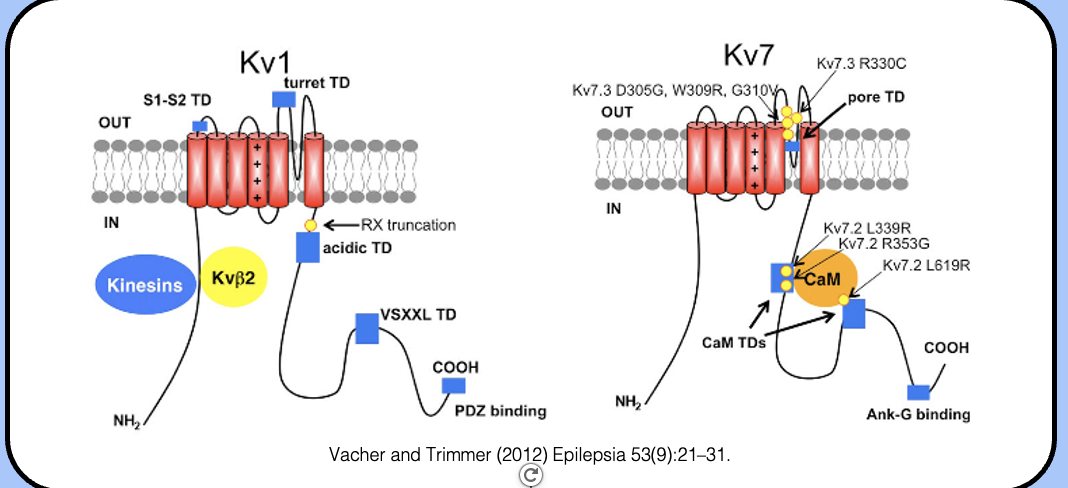
KCNQ channels
the channels underlying the M-current are a class of potassium channels called KCNQ channels (or Kv7.x channels).
they are just one of many types of potassium channel, and share the same topology and structure with their potassium channel cousins.
KCNQ2/3-mediated currents
- in the nervous system, the M-current is mediated by heterotetramers made of KCNQ2 and KCNQ3 subunits (KCNQ2/3 channels).
- the defining feature of this chalk of channels is their slow activation—note that these channels activate at least an order of magnitude more slowly than the H&H potassium channel.
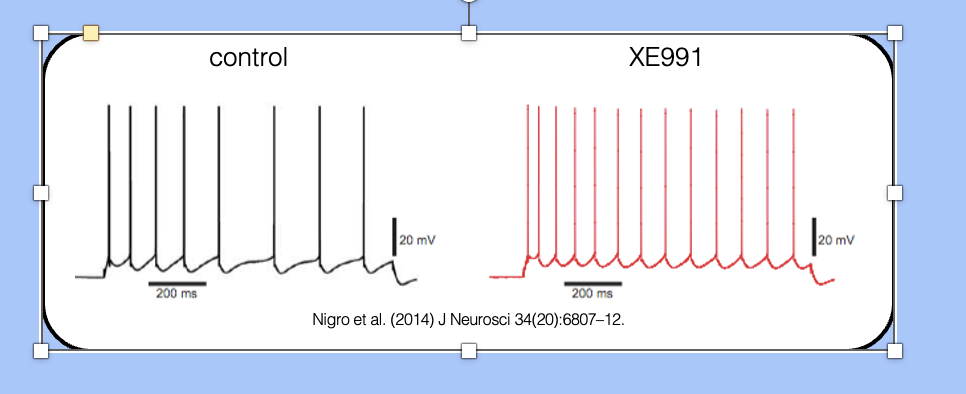
spike frequency adaptation—blocking IM
XE991 is a drug that blocks KCNQ2/3 channels, which can therefore be used to investigate the role of the M-current in SFA.
- here, we see an adapting neurons before and after application of XE991—the adapting neuron has been converted to a non-adapting neuron.
spike frequency adaptation—potentiating IM
- retigabine is a recently approved anti-epileptic drug that potentiates KCNQ-mediated currents.
-here, we can see that a non-adapting neuron can be converted into an adapting neuron by potentiating the M-current with retigabine.
- the slowly activating M-current makes it harder for later action potentials to reach threshold—think about what this does to Noble’s model of excitation.
where does AP originate?
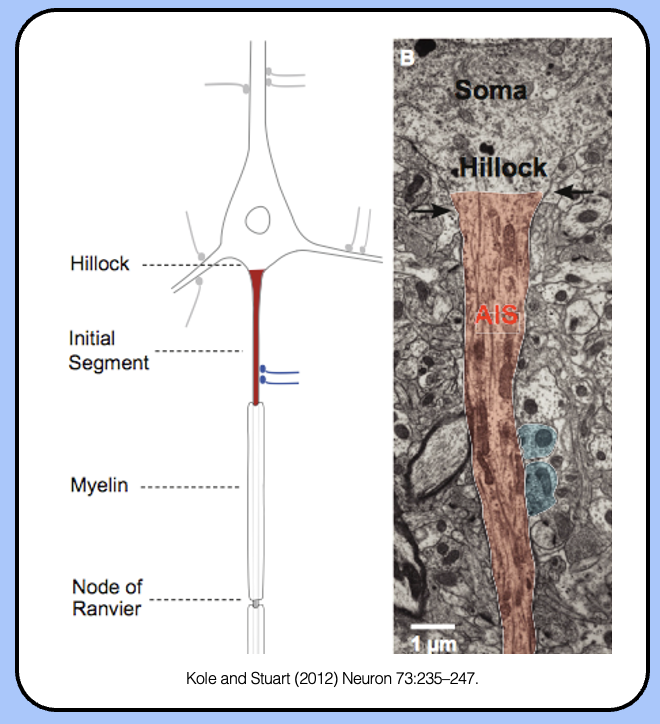
axon initial segment
axon initial segment
the AIS is the region of the neuron dedicated to generating action potentials—threshold is lowest in the AIS.
sodium channels of the AIS
- the AIS is loaded with sodium channels—one way to identify the AIS is to stain for Nav1.6. density going from 110 to 300 channels/um²
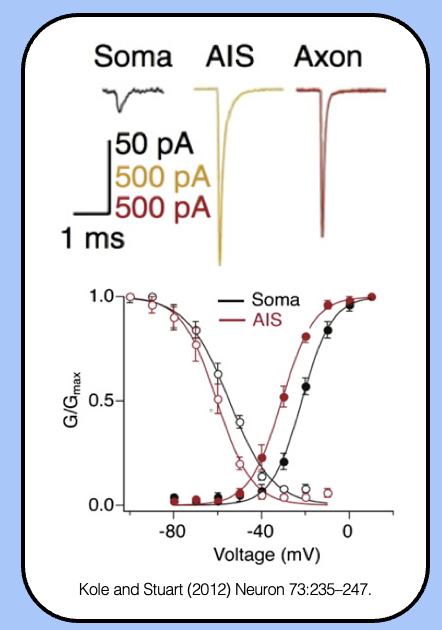
sodium currents of the AIS
- the G-V curve of AIS sodium channels is left-shifted, which means that they’ll open in response to a smaller depolarization.
- the high density of channels combined with their enhanced biophysical properties makes the AIS the most excitable region of the neuron.
potassium channels of the AIS
- the sodium channels of the AIS aren’t alone—there is also a high density of potassium channels at the AIS.
- the major potassium channels are Kv1.2 (which is comparable to the H&H potassium channel) and Kv7.2 / Kv7.3 (aka KCNQ2/3, which mediates the M-current).
cytoskeleton of the AIS
- another defining feature of the AIS is its actin cytoskeleton network, which helps sodium and potassium channels to accumulate in this region and serves as a “filter” to prevent somatodendritic proteins from entering the axon.
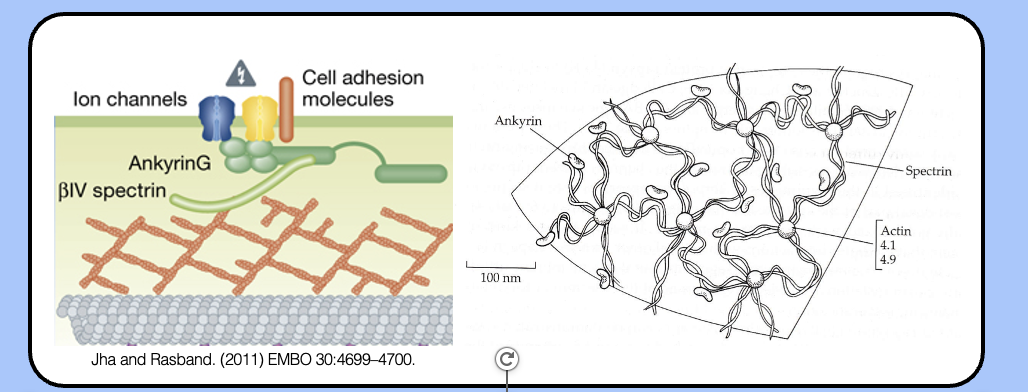
the actin–spectin network
- the actin cytoskeleton of the AIS is organized into a sub-membrane network by the actin-binding protein βIV spectrin.
- a similar sub-membrane network exists beneath the membrane of red blood cells, where spectrin was discovered.
- through its many protein–protein interacts, the actin–spectrin network is the glue that holds the AIS together.
ankyrinG
- sodium and potassium (Kv7) channels are attached to the AIS cytoskeleton by a protein called ankyrinG (AnkG).
- AnkG is the most common molecular marker of the AIS.
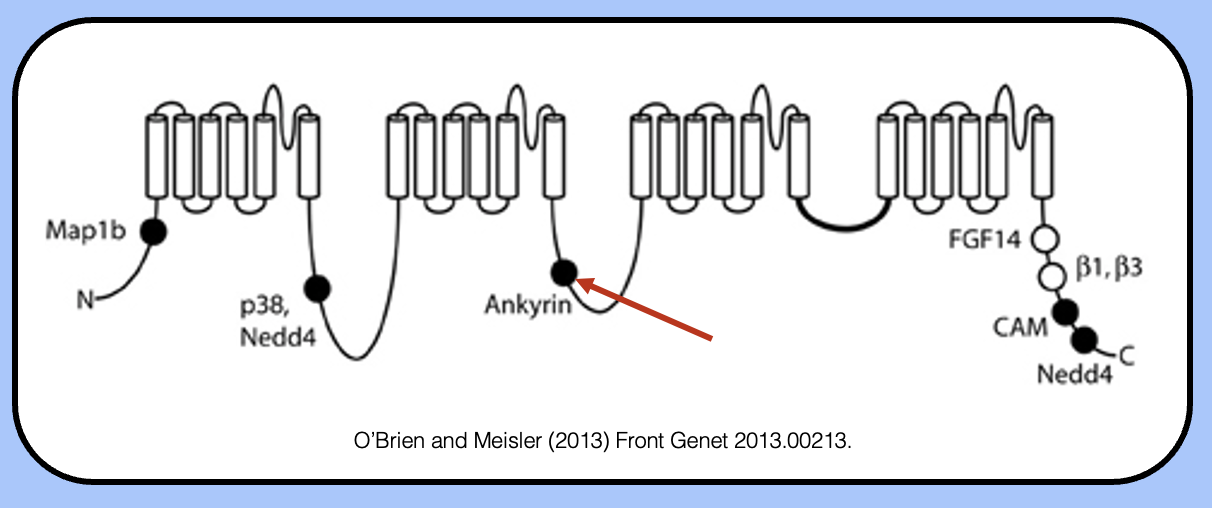
the ankyrin binding site
- sodium channels contain a nine-amino acid motif in their intracellular DII–DIII linkers that allows them to interact with AnkyrinG.
- when engineered onto other channels, this motif is sufficient to localize somatodendritic potassium channels to the AIS.
localizing potassium channels tho the AIS
- like sodium channels, Kv7 (KCNQ) channel have an ankyrinG-binding motif in their C-terminal region that localizes them to the AIS.
- the factors that localize Kv1 channels to the AIS are poorly understood, although the N-terminal tetramerization domain facilitates an interaction with kinesins that seems to be important for their axonal targeting.