Exam 1 Review
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
48 Terms
HCl
-7
H3O+
0
-COOH
5
NH4+
9
phenol
10
R-SH
11
R-NH3+
11
H2O
14
R-OH
17
R-C - - - C-H
26
NH3
36
CH4
60
less than 0
protonated alcohol, carboxylic acid, water
~5
carboxylic acid
~10
protonated amine
~15
alcohol, water
~40
amine

Alkene
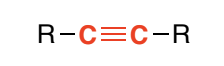
Alkyne
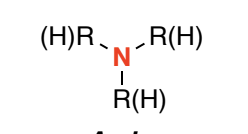
Amine
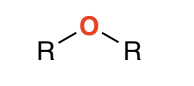
Ether
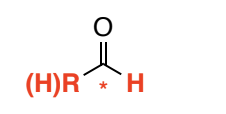
Aldehyde
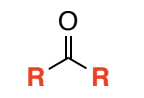
Ketone
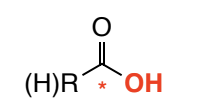
Carboxylic acid

Ester
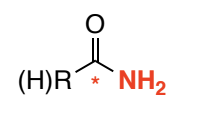
Amide
sp3 no lone pairs
109.5, tetrahedral
sp3 1 lone pair
107, trigonal pyramidal
sp3 2 lone pairs
104.5, bent
sp2 no lone pairs
120, trigonal planar
sp2 1 lone pair
<120, bent
sp
180, linear
B/Al/C+
make 3 bonds, have empty p orbital
Does molecule H-bond with water?
O/N atom w/ lone pair
Does molecule H-bond with itself?
O/N atom w/ lone pair, O-H or N-H bond
Chair conformation energy (lowest to highest)
Chair, twist boat, boat, half chair
Types of gauche interactions
cis-1,2; trans-1,2-diequatorial
Meso checklist
even number of chiral carbons, constitutional symmetry, opposite R/S
Method for determining relationships
constitution, stereochem, conformation
Relative stability of resonance contributors
full octet, electronegativity, minimize charge separation
What is the hybridization state of a vinyl carbocation, carbon radical, and carbanion?
sp, sp, sp2
Gauche interactions
when large groups are 60 degrees apart
1,3-diaxials can be between what types of molecules?
two large molecules or one large molecule and H
How to identify cis/trans stereocenters
1,4 substituted cyclohexane or 1,3 substituted cyclobutane rings with 2 distinct groups
Assign R/S when lowest priority group is in the plane
Assign priority, swap #4 with dashed group, render configuration, reverse
what are conformational isomers
formed from rotation around single bonds
Reaction favors side with _____ pKa
higher
The most stable base is the _______ base, is the ________ acid
weakest, strongest