MIDTERMS STUDY GUIDE: Bio
1/158
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
159 Terms
Explain why carbon has an ability to form large, diverse molecules
Carbon can make many different molecules because it can bond with four other atoms at once. This lets it form long chains and shapes, making a huge variety of molecules.
Define inorganic compounds
chemicals that don’t primarily come from living things and usually don’t contain carbon-hydrogen (C-H) bonds. Ex: water, salts, and metals.
Define organic compounds
chemicals that mostly contain carbon and hydrogen. They are usually found in living things. Ex: sugar, fats, and proteins.
List the four main classes of macromolecules important to life
Carbohydrates (ex. sugars, starches), Proteins (ex. enzymes, muscles), Lipids (ex. fats, oils), and Nucleic Acids (ex. DNA, RNA)
Explain the relationship between monomers and polymers
Monomers are small building blocks, and polymers are long chains made by linking many monomers together. For example, individual sugar molecules (monomers) can join to form a sugar chain (polymer).
Compare dehydration synthesis and hydrolysis (simplified version)
Dehydration makes bigger molecules, while hydrolysis breaks them down
Define dehydration synthesis
when two molecules join together, and a water molecule is removed
Define hydrolysis
when a water molecule is added to break a bond between two molecules
Name the monomers of carbohydrates
monosaccharides (like glucose and fructose)
Describe the composition and structure of carbohydrates
Carbohydrates are made of sugar molecules, which are made of carbon, hydrogen, and oxygen. They can be simple sugars (like glucose) or long chains of sugars (like starch). The basic formula for most carbohydrates is CH₂O.
examples of disaccharides
sucrose (table sugar), lactose (milk sugar), maltose
examples of monosaccharides
glucose, fructose, galactose
examples of polysaccharides
starch, glycogen, cellulose
what are the two roles of carbohydrates?
Being an energy source and storage
explain and provide examples for carbohydrates function of being an energy source
Carbs provide quick energy. Ex: glucose is used by our cells for energy
explain and provide examples for carbohydrates function of being storage
Carbs can be stored for later use Ex: starch in plants and glycogen in animals store energy for when it’s needed
Classify the polysaccharide: starch
found in plants; stores energy
Classify the polysaccharide: chitin
found in animals and fungi; provides structure
Classify the polysaccharides: cellulose
found in plants; provides structure (cell wall)
Classify the polysaccharides: glycogen
found in animals; stores energy
what carbohydrates are found in the human diet?
sugars, starches, and fibers
what do sugars do in the human diet?
provide quick energy and are found in fruits and sweets (like glucose and sucrose)
what do starches do in the human diet?
store energy and take longer to digest (like in potatoes, bread, and rice)
what do fibers do in the human diet?
helps with digestion and keeps the digestive system working well, but our body can’t digest it (found in vegetables, fruits, and whole grains)
a carbohydrate
Name the subunits of lipids
fatty acids and glycerol
Describe the composition and structure of triglycerides
…are made up of one glycerol molecule and three fatty acids. The glycerol is a small molecule, and the fatty acids are long chains of carbon, hydrogen, and oxygen atoms. They are connected in a way that forms a "backbone" of glycerol with three fatty acid tails attached.
Explain saturated fatty acids
have no double bonds between carbon atoms, and all carbon atoms are "saturated" with hydrogen. They are usually solid at room temperature (e.g., butter)
Explain unsaturated fatty acids
have one or more double bonds between carbon atoms, which means they have fewer hydrogen atoms. They are usually liquid at room temperature (e.g., olive oil)
Explain why fats are hydrophobic
they are made of long chains of carbon and hydrogen, which don’t mix well with water. Water is a polar molecule, while fats are nonpolar, so they repel each other. This is why fats don’t dissolve in water.
Describe the structures, functions, properties, and types of lipid molecules
include fats, oils, phospholipids, and steroids. Fats and oils store energy and provide insulation. Phospholipids form cell membranes, while steroids, like cholesterol, act as hormones. Lipids are hydrophobic, energy-rich, and play key roles in protection, regulation, and structure in the body.
Describe the structural properties of phospholipids that make them essential for cell membranes.
…have a unique structure with a hydrophilic (water-loving) head and two hydrophobic (water-hating) tails. This allows them to form a bilayer in cell membranes, where the heads face outward toward water, and the tails face inward, away from water. This structure creates a barrier that protects the cell while allowing certain substances to pass in and out.
a lipid: saturated
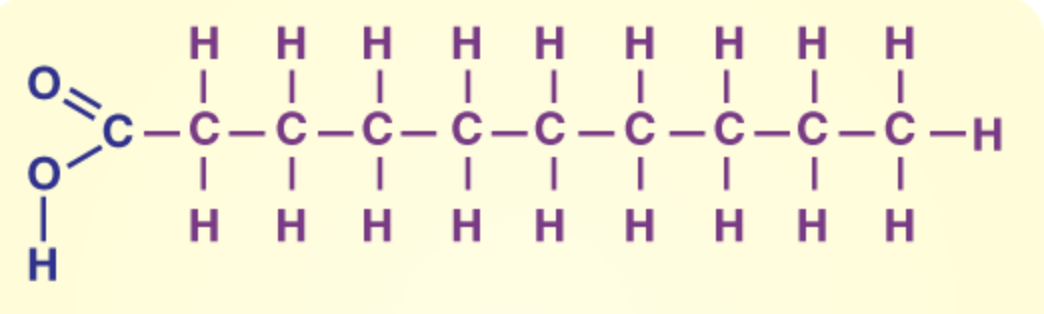
a lipid: unsaturated
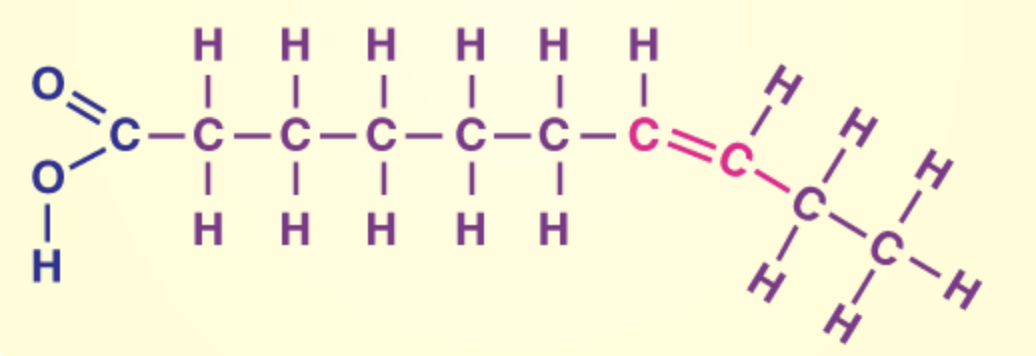
name the monomers of proteins
amino acids
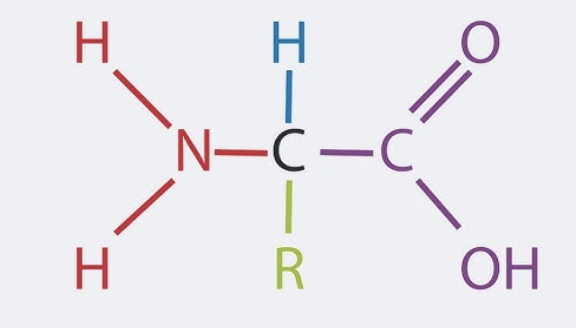
Describe the structure of an amino acid
has a central carbon atom bonded to a hydrogen atom, an amino group (–NH₂), a carboxyl group (–COOH), and a unique side chain (R group)
explain the relevance of the R group in an amino acid
makes each amino acid unique, affecting how it interacts in proteins
Describe the structures, functions, properties, and types of proteins.
Proteins are made of amino acids and have different shapes. They do jobs like speeding up reactions (enzymes), building structures (collagen), and carrying molecules (hemoglobin). Types include enzymes, structural proteins, transport proteins, antibodies, and hormones. Their properties depend on their amino acids.
name the 4 levels of protein structure
primary, secondary, tertiary, and quaternary
Describe the primary structure and identify the types of chemical bonds and interactions that play a role
The sequence of amino acids in a chain. Peptide bonds link the amino acids.
Describe the secondary structure and identify the types of chemical bonds and interactions that play a role
Local folds like alpha helices and beta sheets. Hydrogen bonds help stabilize these folds.
Describe the tertiary structure and identify the types of chemical bonds and interactions that play a role
The overall 3D shape of the protein. Hydrophobic interactions, hydrogen bonds, ionic bonds, and disulfide bonds (strong covalent bonds between cysteine amino acids) hold the shape together
Describe the quaternary structure and identify the types of chemical bonds and interactions that play a role
Multiple protein chains (subunits) coming together. Non-covalent interactions (like hydrogen bonds and hydrophobic interactions) or covalent bonds (like disulfide bonds) hold the subunits together
Explain why a change in the amino acid sequence of a protein can impact that proteins function
can alter a protein's shape, making it unable to function properly, like failing to bind or catalyze reactions. Even small changes can disrupt its activity.
Explain how denaturation affects protein structure
Denaturation is when a protein’s structure unravels, losing its shape. This happens due to factors like heat or pH changes. Without its proper shape, the protein can't function correctly.
amino acids
name the monomers of nucleic acids
nucleotides
describe DNA
double-stranded, has the sugar deoxyribose, and uses the bases A, T, C, G. It stores genetic information.
describe RNA
single-stranded, has the sugar ribose, and uses A, U, C, G (with uracil instead of thymine). It helps in protein synthesis and carries genetic information from DNA.
nucleotides
endergonic reaction
chemical reaction that requires energy to occur, absorbs energy from surroundings
energy coupling
process where energy from an exergonic (energy-releasing) reaction is used to drive an endergonic (energy-absorbing) reaction
exergonic reaction
chemical reaction that releases energy, the products have less energy than the reactants, so energy is given off, often as heat or light
metabolic pathway
series of chemical reactions in a cell, where the product of one reaction becomes the reactant for the next
metabolism
chemical reactions that happen in a cell or organism to maintain life; breaking down molecules for energy (catabolism) and building new molecules (anabolism)
endergonic reactions (graph)
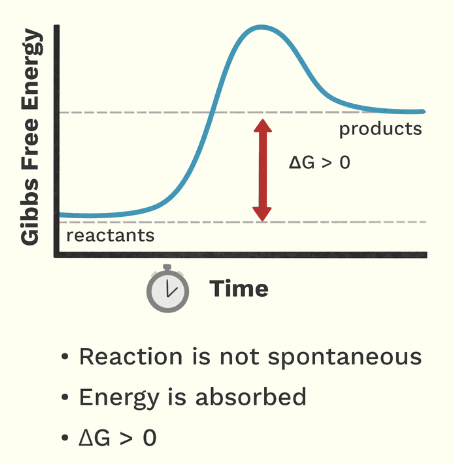
exergonic reactions (graph)
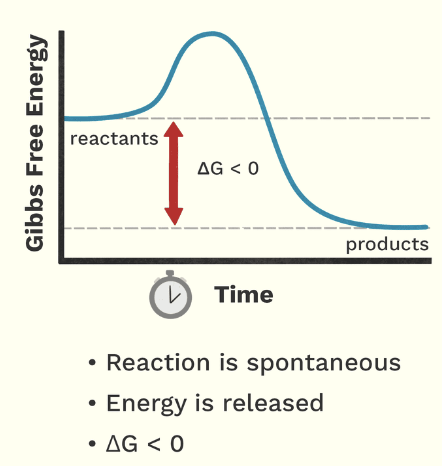
Describe the relationship between catabolic reactions with endergonic or exergonic reactions
break down molecules and release energy, so they are exergonic
Describe the relationship between anabolic reactions with endergonic or exergonic reactions
build molecules and require energy, making them endergonic
catabolic is equals…
= exergonic (energy-releasing)
anabolic is equals…
= endergonic (energy-consuming)
activation energy
energy needed to start a chemical reaction. It’s like a "push" that helps molecules react with each other
catalyst
substance that speeds up a chemical reaction without being used up in the process. It lowers the activation energy needed for the reaction to occur
energy barrier
amount of energy needed to start a chemical reaction. It’s the "hurdle" that reactants must overcome for a reaction to happen
enzyme
protein that speeds up chemical reactions in the body. It lowers the energy needed for a reaction to occur, making processes like digestion and metabolism happen faster
Explain why cells need enzymes
they speed up chemical reactions, allowing important processes to happen quickly and efficiently at the right temperature. Without enzymes, reactions would be too slow to support life
Describe the relationship between enzymes and activation energy
Enzymes lower the activation energy needed for a reaction to start. This makes reactions happen faster and more easily
graph labeled
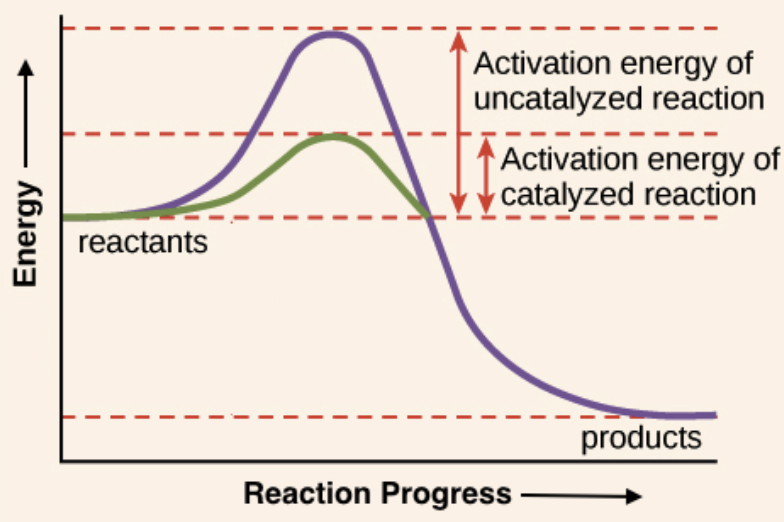
active site
the region on an enzyme where the substrate binds and a chemical reaction is catalyzed
allosteric site
different part of an enzyme where molecules can bind, changing the enzyme's shape and affecting its activity. It’s like a “control” switch for the enzyme
cofactors
small molecules or ions that help enzymes work properly
coenzymes
type of cofactor, usually organic molecules, that help enzymes by carrying chemical groups or electrons during reactions
induced fit
model describing how an enzyme's active site changes shape when the substrate binds
substrate
the molecule that an enzyme acts on to help start a chemical reaction
Describe how protein structure relates to specificity of enzymes
…are proteins with a unique shape. This shape allows them to only bind to certain molecules (substrates), like a key fitting into a lock. This is why each enzyme works with specific substances
Explain the steps of a catalyzed reaction (the catalytic cycle)
binding - The substrate (molecule) attaches to the enzyme’s active site.
reaction - The enzyme helps convert the substrate into a product by speeding up the reaction.
release - The product is released from the enzyme.
reset - The enzyme is ready to bind to a new substrate and repeat the proces
Identify factors that influence the optimal conditions for enzyme function
Enzyme activity depends on temperature, pH, substrate concentration, enzyme concentration, and salt levels. Each enzyme has ideal conditions, and if these conditions are off, the enzyme may not work well
Predict the effects that a change in one of these factors would have on enzyme activity
Changes in temperature, pH, substrate, enzyme concentration, and salt levels can slow down or stop enzyme activity. Each factor has an optimal range for the enzyme to work best.
Describe the structure of a plasma membrane and relate these parts to the functions of the plasma membrane
a phospholipid bilayer with proteins, cholesterol, and carbohydrates. It controls what enters and exits the cell, supports the cell, and helps with communication and recognition.
describe the structures of prokaryotic cells
…are simpler, smaller, and lack a nucleus. Their DNA is in a region called the nucleoid. They also don't have membrane-bound organelles, like mitochondria or the endoplasmic reticulum. Examples include bacteria and archaea.
describe the structure of eukaryotic cells
…are larger and more complex, with a defined nucleus that holds their DNA. They have membrane-bound organelles, such as the mitochondria, Golgi apparatus, and endoplasmic reticulum. Examples include plant, animal, and fungal cells
Explain why compartmentalization is important in eukaryotic cells
helps separate different functions into specific areas, making processes more efficient and controlled. It allows the cell to perform complex tasks without interference between them.
Describe the structures and functions of the four basic functional compartments of eukaryotic cells.
the nucleus (controls the cell), the cytoplasm (holds organelles), the endomembrane system (processes proteins and waste), and the mitochondria (produce energy). These parts help the cell function efficiently.
Compare the structures of plant and animal cells. Note the function of each cell part
Plant and animal cells both have a nucleus, cytoplasm, and mitochondria. Plant cells also have a cell wall, chloroplasts for photosynthesis, and a large vacuole for storage. Animal cells have lysosomes for waste breakdown and centrioles for cell division. These structures help each cell type do its specific job.
Describe the parts of the cell theory
First, all living things are made of cells. Second, the cell is the basic unit that carries out all life functions. Third, all cells come from existing cells through cell division. These ideas are essential for understanding how life works at the cellular level.
cell theory
states that all living organisms are made of cells, the cell is the basic unit of life, and all cells come from pre-existing cells
cell wall
a rigid outer layer that surrounds plant, fungal, and bacterial cells. It provides support, protection, and helps maintain the shape of the cell.
cellular metabolism
refers to all the chemical reactions that occur within a cell to maintain life. It includes two main processes of catabolism and anabolism
central vacuole
a large, fluid-filled space in plant cells. It stores water, nutrients, and waste products, and helps maintain the cell's shape by providing turgor pressure
centrosome
a region in animal cells that organizes microtubules. It contains centrioles and helps control cell division by forming the mitotic spindle that separates chromosomes during cell division
chloroplasts
are organelles in plant cells that capture sunlight and use it to make food (glucose) through photosynthesis. They contain a green pigment called chlorophyll, which helps absorb light
chromatin
a complex of DNA and proteins found in the nucleus of eukaryotic cell
chromosome
a long, thread-like structure made up of DNA and proteins that carries the genetic information in cells
cilia
tiny, hair-like structures that protrude from the surface of some cells
crista
a fold or ridge inside the mitochondria
cytoplasm
the jelly-like substance inside a cell that fills the space between the cell membrane and the nucleus
cytosol
the fluid part of the cytoplasm in a cell
cytoskeleton
a network of fibers inside a cell that gives the cell its shape and helps it maintain structure
endomembrane system
a group of related organelles inside a cell that work together to make, modify, and transport proteins and lipids (fats)
endoplasmic reticulum
a network of membranes inside a cell that helps make and transport proteins and lipids (fats)