Organic Chemistry Exam 3
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
36 Terms
Halolkane
C-X

Ketone
O=C(-C)
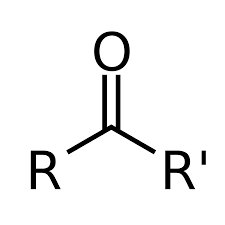
Aldehyde
O=C-H
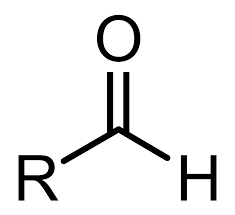
Carboxylic Acid
O=C-O-H
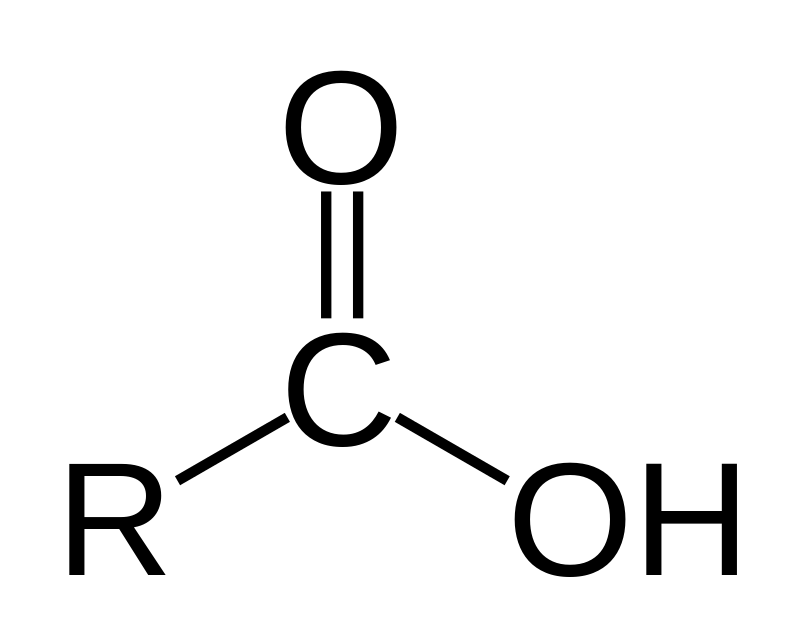
Ester
O=C-O-C
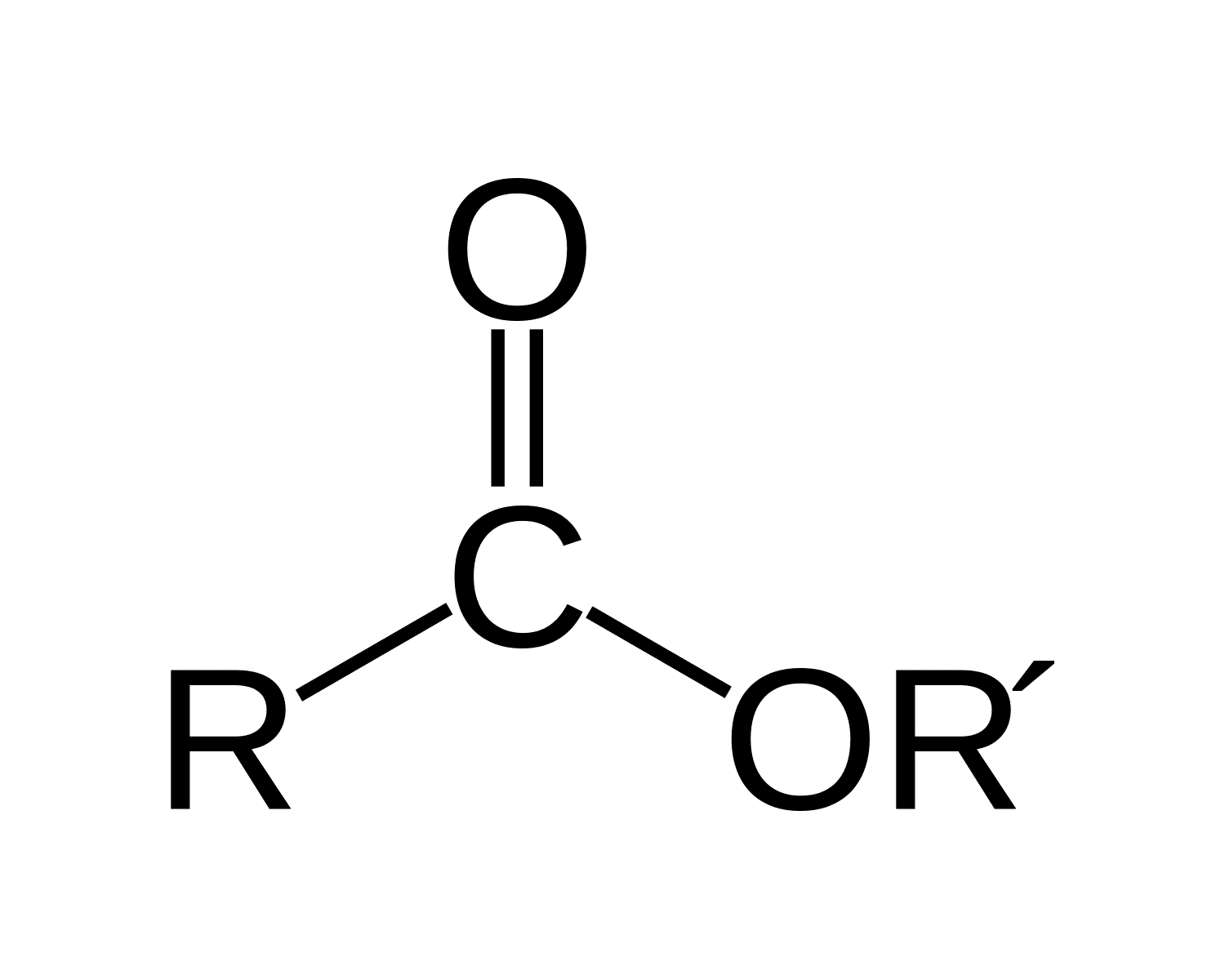
Amide
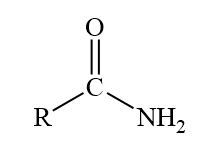
O=C-N
Amine
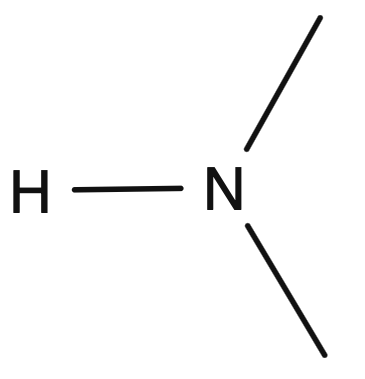
C-N
Ether
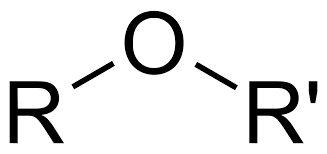
C-O-C
Alcohol
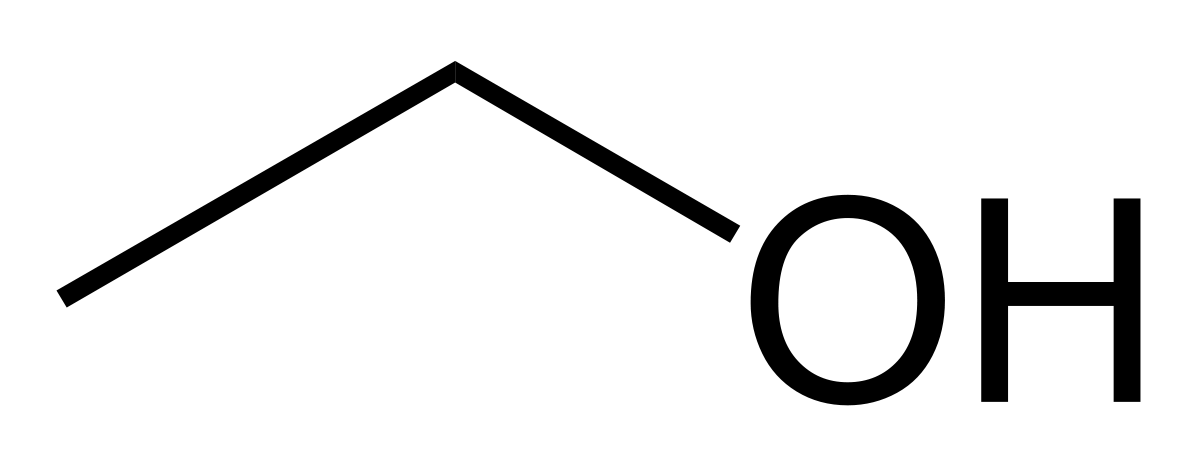
C-O-H
F
Fluoro
I
Iodo
Cl
Chloro
Br
Bromo
Alcohol Suffix
-ol
Common Good LG (More Basic)
I>Br>Cl; O+R2, OSO2R (OTs, OMs)
Common Good Nucleophiles (Less sterically crowded, stronger bases)
Br⊖, CN⊖, N3⊖, I⊖, ⊖SH, ⊖SR, ⊖OH, ⊖OR
Common Average Nucleophiles
NH3, Cl⊖, RCO2⊖
Common Poor Nucleophiles
H2O, ROH (CH3OH or MeOH, EtOH or CH3CH2OH), CH3COOH (AcOH), HSO4⊖
Common Protic Solvents (Anions)
H2O, CH3OH(MeOH), EtOH (CH3CH2OH), CH3COOH (AcOH) (Contains OH)
Common Aprotic Solvents (Metals)
DMF, DMSO, acetone, acetonitrile
Double Bonds
changes ending from an to en, add number of start of bond
E/Z
e is trans z is cis
Me
SN2 only
1
SN2 and E2 possible
3
E2 very fast with a strong base (E1: + H2SO4) (E2: + POCl3, Py)
2
E2 fast with a strong base, but all mechanisms possible (SN2+ SN1: + HCl, HBr, HI) (SN2: + PBr3, SOCl2) (E1: + H2SO4) (E2: + POCl3, Py)
vinylic
E2 possible
Weak Base
E2 Slow
Strong Base
E2 Speeds Up
Poor nucleophile
SN2 very slow
Good Nucleophile
SN2 speeds up
Not Polar
SN1/E1 slow down; SN2 and E2 less so
Polar Aprotic
SN2 speeds up more than SN1
Polar Protic
SN1
Vinylic
one of the two atoms that form the double bond
Allylic
carbon atom that is next to the double-bonded carbon atom