AP Chemistry ALL UNITS (1-9) Ultimate Study Guide [INCOMPLETE]
1/161
Earn XP
Description and Tags
okay so ik this is a little late but if anyone else has procrastinated to the point of last minute cramming like me then here you go have this i guess. i have another one of these of just units 1-5 i made for my midterm and just kinda copied over here with the new units added so... yeah lol. rn i have units 1-6 done, 7 is started but ill finish tmmr probably
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
162 Terms

Theory which predicts the geometry of molecules, founded on the idea that valence electrons repel from one another, so they will be as far away from each other as possible in a molecule’s shape. Although this is neglectable, the more delocalized lone pairs there are, the more they will repel. The angle of an electron’s repulsion will depend on the amount of electron domains present. Also, while lone pairs don’t physically contribute to molecular geometry, their repulsion still does.
For different electron and molecular geometries, go here: https://knowt.com/flashcards/841bf0c5-ebab-461b-8672-ab2f158e957b
Molarity
The concentration, typically shown with brackets ‘[]’, of a solution as it is directly related to moles, represented by the equation M = n × V (where M is molarity, n is moles of solute, and V is volume of solvent in liters).
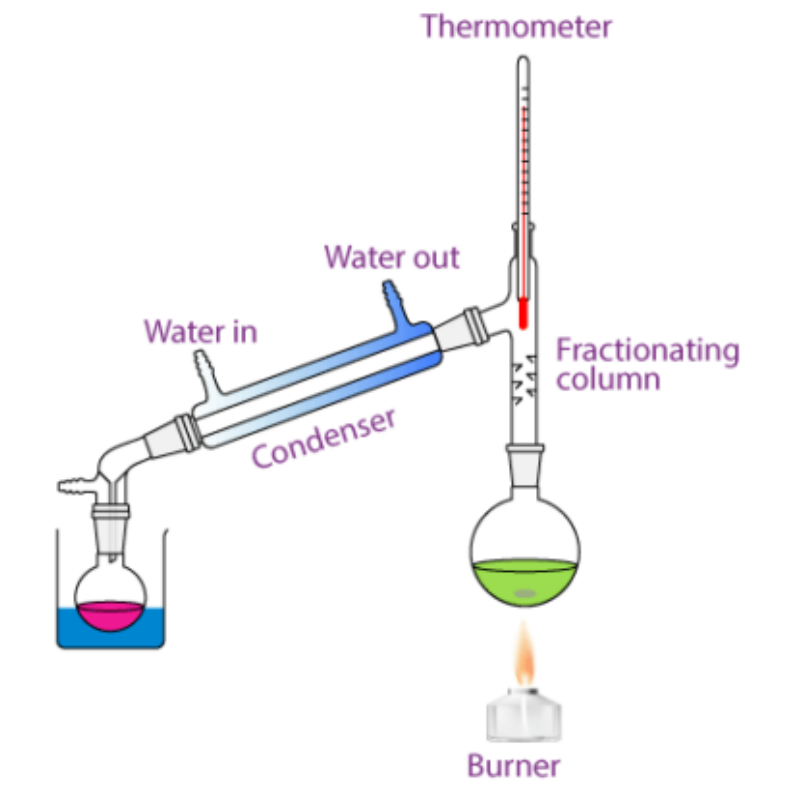
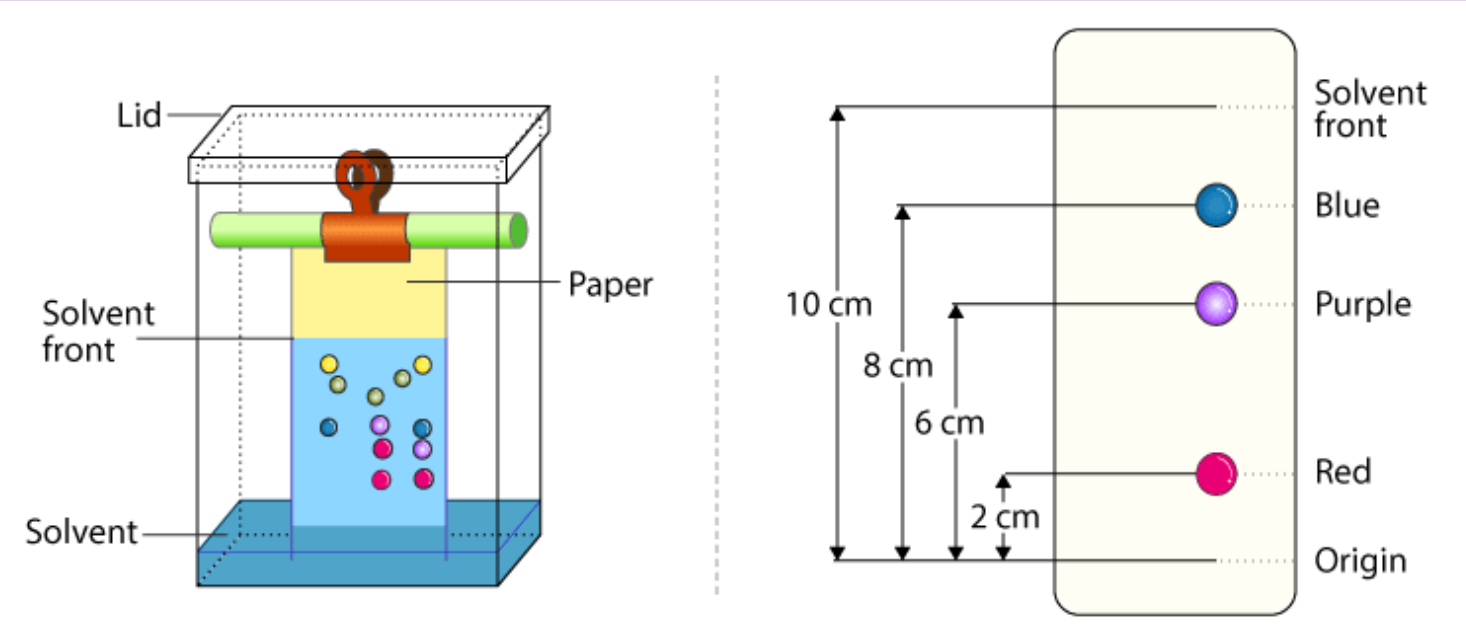
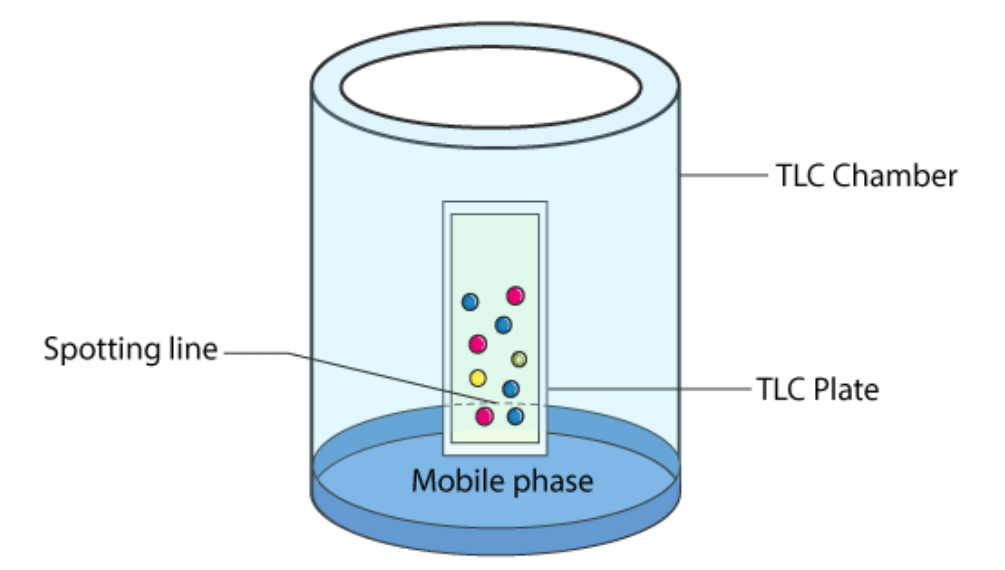
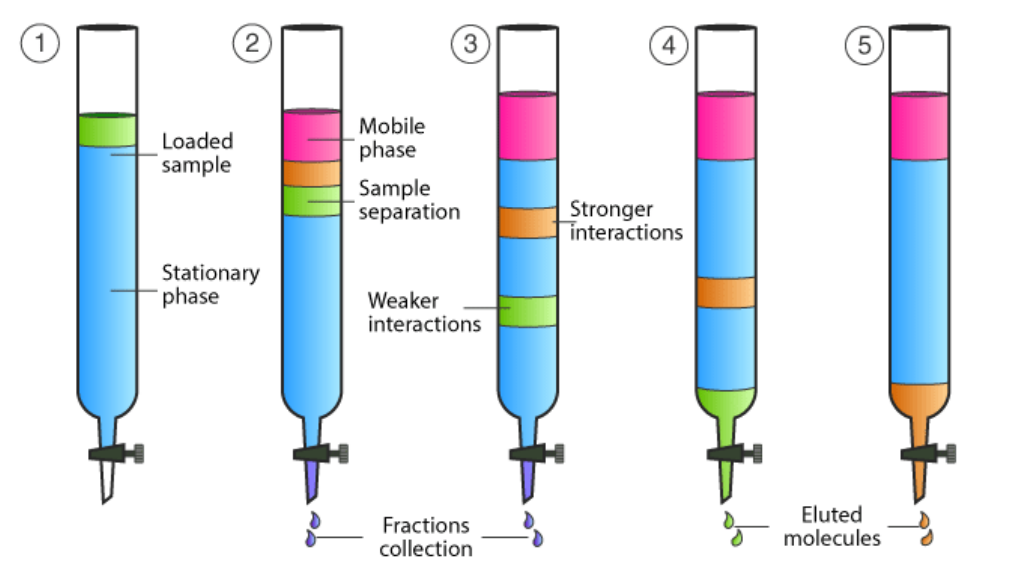
![The absorbance [of light waves] of a chemical is defined by the equation A = εbC (where A is absorbance often within the interval 0 < A < 1, ε is the molar absortivity constant, b is the path length of the curette in cm [if not indicated, it is assumed to be 1cm], and C is the concentration of the chemical in M or m/L). Experimentally, this can be demonstrated by placing a substance in a curette and targeting a specific frequency of light at it to measure its absorbance factor.](https://assets.knowt.com/user-attachments/0259ab28-65f0-42d9-afd8-ad1ef7d5204a.png)