SACE Stage 1 Physics Light & Atoms
1/35
Earn XP
Description and Tags
bruh im screwed; nvm i was fine
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
What is an electromagnetic wave and the wave model for light?
.
.
.
.
SACE Terms:
Notes:
Electromagnetic waves are sinusoidal, transverse waves consisting of electric and magnetic fields which oscillate at right angles to each other and to the direction of wave propagation.
The wave model for light theorises that an oscillating electric field will induce an oscillating magnetic field, and vice versa.
What is plane polarisation of radio and television waves?
.
.
.
.
SACE Terms:
Notes:
Plane-polarisation occurs when vibrations are restricted to a single plane.
Radio and television waves are produce when electrons in the transmitting antenna oscillate. Since the electrons oscillate in one plane, parallel to the antenna (direction of electric field), the emitted electromagnetic waves are therefore plane-polarised in the direction of the vibrating charge
i.e the emitted electromagnetic waves of radio and television waves are plane-polarised in the direction of the vibrating charge (in the antenna).
How are radio-waves transmitted?
.
.
.
.
SACE Terms:
Notes:
When an alternating potential difference is applied, this causes the electrons inside to oscillate along the length of the antenna.
This oscillation thereby induces an electric field, and hence a magnetic field; where each are perpendicular to each other and to the propagation of the wave, and the frequency of the alternating potential difference is the same as the oscillating fields.
The continual reproduction of the fields by each other causes the electromagnetic wave to radiate outwards in the same plane-polarisation as the oscillating electric field, thus transmitting the signal.
How are radio-waves recieved?
.
.
.
.
SACE Terms:
Notes:
When receiving a signal, the antenna must be aligned in the direction of the polarised waves being transmitted.
As the transmitted wave approaches the receiving antenna, the electric field exerts a force (F=Eq), causing the electrons inside to oscillate at the same frequency.
This oscillation produces an alternating potential difference, thus allowing the radio/television wave to be received.
Why does plane-polarisation of the receiving antenna matter?
.
.
.
.
SACE Terms:
Notes:
If the receiving antenna is orientated in the same plane of polarisation as the transmitted wave, a strong signal will be received, since it is the electric field that oscillates the electrons not the magnetic field.
If the receiving antenna is perpendicular to the transmitted wave, the electrons cannot oscillate when the electromagnetic wave approaches, causing a weak signal to be received.
Define monochromatic light
.
.
.
.
SACE Terms:
Notes:
Monochromatic light is light composed of one frequency with waves that radiate outwards away from the source in all directions.
Monochromatic light is light composed of a single frequency with radiating waves in all directions away from the source.
What is a coherent wave source?
.
.
.
.
SACE Terms:
Notes:
Waves that maintain a constant in phase relationship.
What are incandescent light sources and how are they produced?
.
.
.
.
SACE Terms:
Notes:
Incandescent sources of light are neither monochromatic nor coherent, and display a continuous spectrum. They are composed of several colours ranging red to violet, and are produced by heating a material until it glows. The random oscillation of charges result in the emitted light being out of phase.
i.e. not one colour, not one frequency.
In the context of a light globe, the filament is heated with electricity, which causes random vibrations of electrons at a range of different frequencies (non monochromatic; not coherent; out of phase) as they move through the filament.
These oscillations thereby produce electromagnetic radiation/light.
What is the principle of superposition for electromagnetic waves?
.
.
.
SACE Terms:
Notes:
For two waves at a point, the principle of superposition states that the resultant wave is the vector sum of the individual amplitudes.
What is constructive and deconstructive interference?
theres a better card below.
.
.
.
.
SACE Terms:
Notes:
For two waves at a point, the principle of superposition states that the resultant wave is the vector sum of the individual amplitudes.
For two monochromatic light sources in phase, the waves at some distance away will constructively interfere when the path difference from the sources to the point is mλ. Since the amplitude of the resultant wave is equal to the vector sum of the individuals amplitude,
For two monochromatic light sources out of phase, the waves at some distance away will de-constructively interfere when the path difference from the sources to the point is (m + ½)λ.
The path difference between S1 and S2 is ½λ at position X. Explain why a minimum is observed at X.
.
.
.
.
SACE Terms:
Notes:
Waves are out of phase and undergo deconstructive interference when the path difference from the sources to a point is (m + ½)λ.
Hence, if the path difference at X is ½λ, then the waves from the double slits are out of phase and undergo deconstructive interference.
The amplitude of the waves cancel, and the result is light of minimum intensity (dark fringe).
Why is light sometimes shown through a single slit first before passing through Young’s double slit?
.
.
.
.
SACE Terms:
Notes:
If any light is shone through a single slit, it will attain coherency. Hence, by the time light reaches the double slit, it will be monochromatic and coherent.
If light is shone through single slit, it will polarise the waves
polarises waves into same plane as single slit, hence coherent and in phase by the time it reaches double slit
How is double slit interference produced?
.
.
.
.
SACE Terms:
Notes:
Produced in a laboratory using a coherent light source, or using a single slit between the light source and double slit.
The double slit’s interference pattern for light consists of alternating dark and bright fringes of equal width.
What produces the light fringes and dark fringes (interference pattern)?
.
.
.
.
SACE Terms:
Notes:
Waves are in phase and undergo constructive interference when the path difference from the sources to a point is mλ. As per principle of superposition, the amplitudes add together, resulting in light of high/maximum intensity (bright fringe/maxima).
Waves are out of phase and undergo deconstructive interference when the path difference from the sources to a point is (m + ½)λ. As per the principle of superposition, the amplitudes cancel out, resulting in light of low/minimum intensity (dark fringe/minimum)
At particular angles, the propagating waves will meet/overlap inducing either constructive (waves in phase) or destructive interference (waves out of phase).
As per principle of superposition, this produces the dark spots, and bright spots.
What is the intensity distribution graph for double slit interference?
.
.
.
.
SACE Terms:
Notes:
Must be equally spaced, same peak heights.
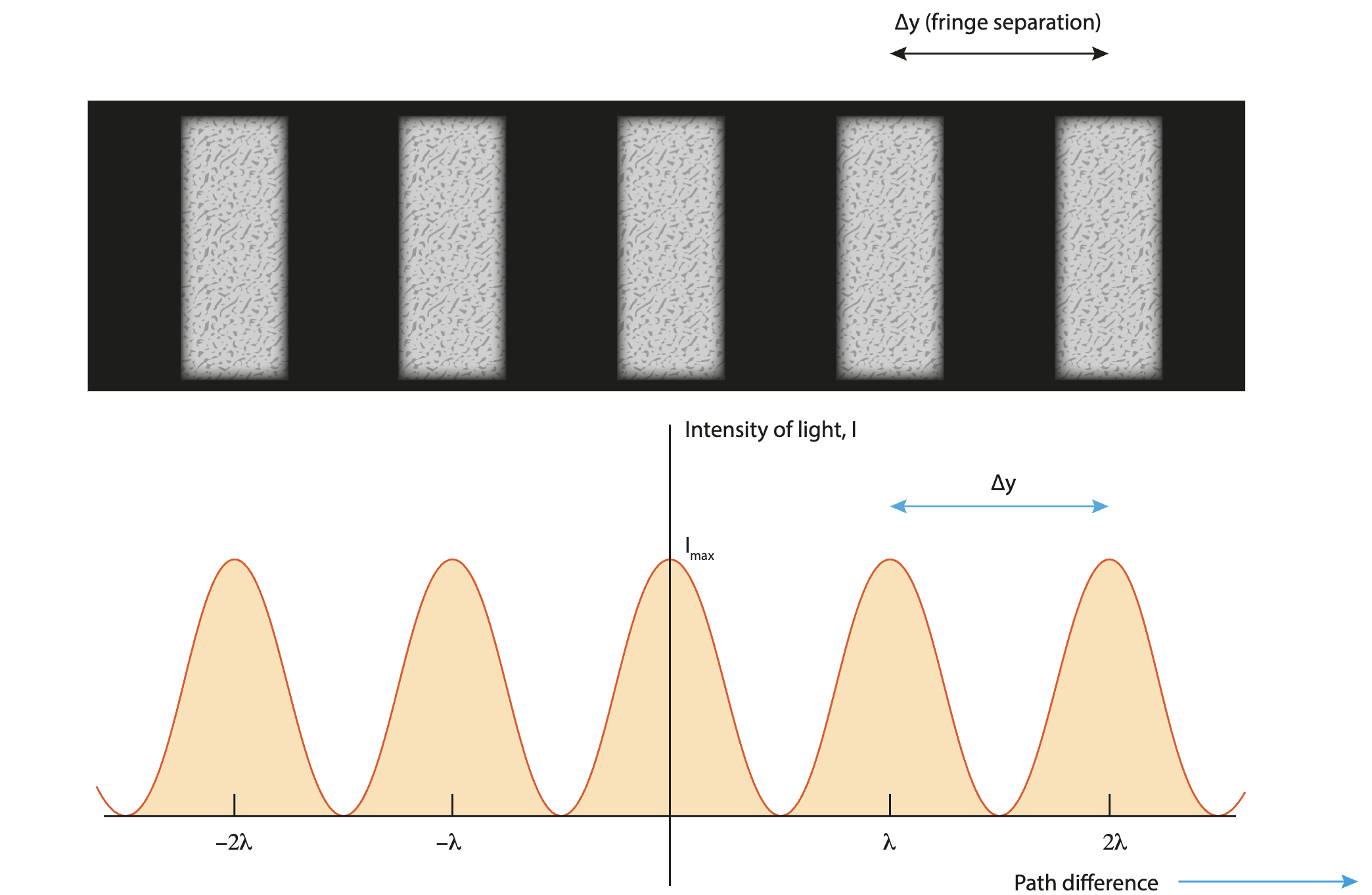
Two narrow parallel slits are illuminated by light of two wavelengths.
λ1=580nm, and λ2 is not known.
On the interference patterns produced on a screen, the fourth bright fringe of λ1 coincides with the sixth dark fringe of λ2. Find λ2.
.
.
.
.
SACE Terms:
Notes:
for λ1 and λ2 to concide, dsinθ for both must be the same.
d1sinθ1=d2sinθ2
∴ m1λ1=m2λ2
(3)(580×10-9)=(5.5)(λ2)
∴ (3)(580×10-9)/(5.5) = 3.16×10-7
=316nm
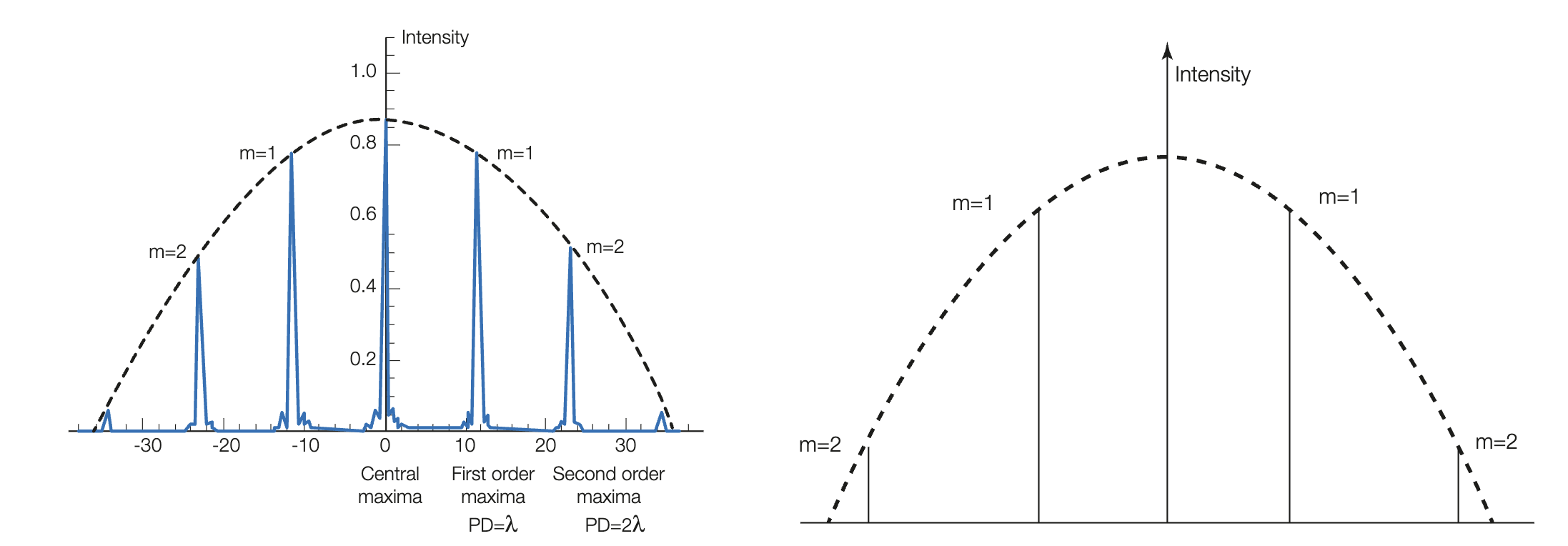
What is the intensity distribution graph for diffraction gratings? (draw it)
.
.
.
.
SACE Terms:
Notes:
The intensity decreases as maximas move away from central maxima
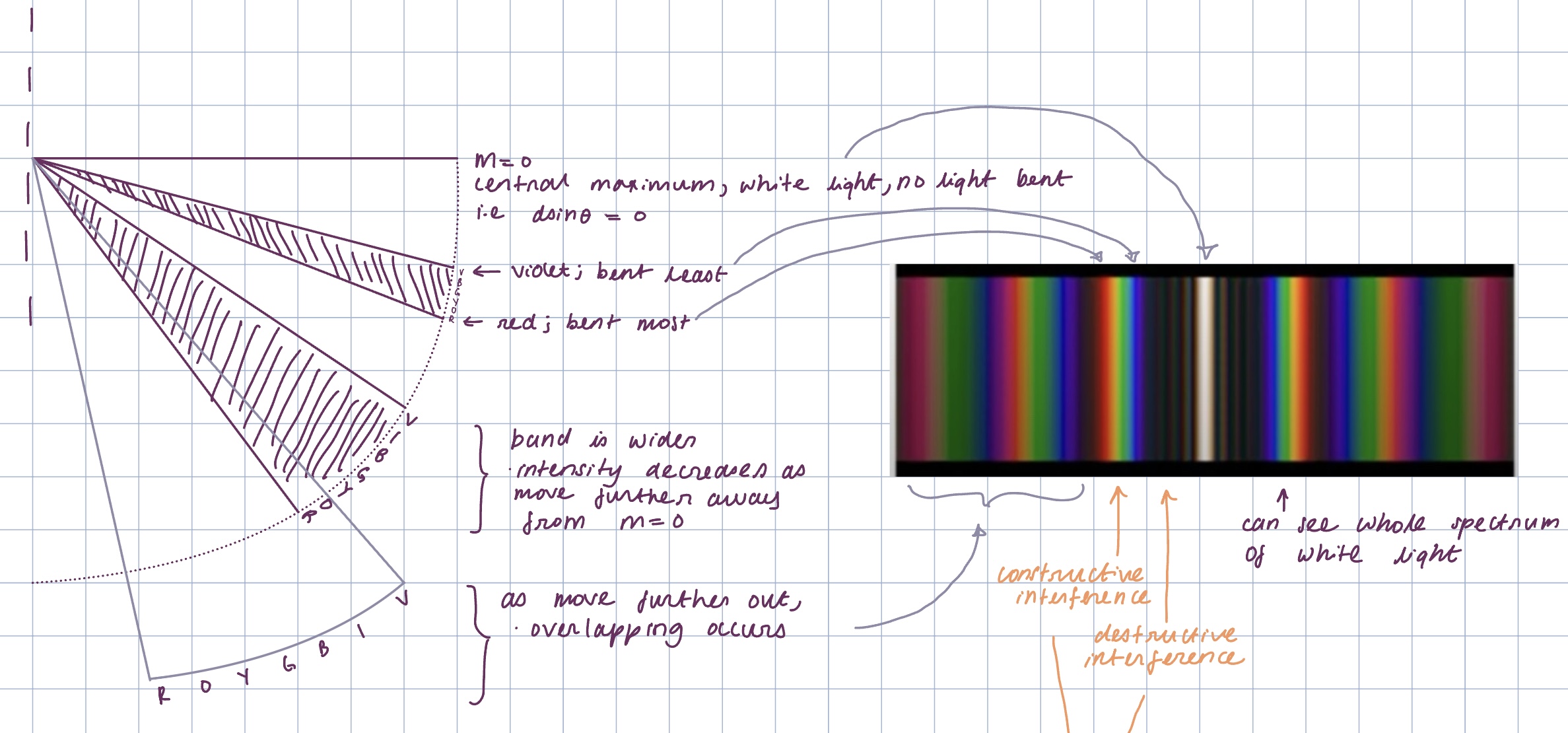
What happens when light white is shone through a diffraction grating?
.
.
.
.
SACE Terms:
Notes:
When light is viewed through a diffraction grating the colours are separated.
Violet has the shortest wavelength (and hence highest frequency), meaning it diffracts at the smallest angle (dsinθ=mλ, sinθ∝λ).
Red light has the longest wavelength (and hence lowest frequency), meaning it diffracts at the largest angle. (With all other colours in between).
However, at the zero order maximum, the path difference and phase for all colours are the same, causing constructive interference, resulting in a sum of white light at m=0.
When does light act as waves, and particles? When do particles act as waves?
.
.
.
.
SACE Terms:
Notes:
Light as Waves:
Double slit/Diffraction grating: The interference pattern produced when light is shown through a a double slit or through a transmission-diffraction grating represents the wave behaviour of light.
Light as particles:
Low Intensity light through double slit: When very low intensity light is incident on double slit, the interference pattern can be observed to build-up over time. This serves as proof that the particle and wave nature of light act together.
Photoelectric effect: When light of sufficient energy and frequency (W and f0) is incident on a metal cathode, it can be absorbed, causing the emission of electrons. Specifically, for light to induce this ejection, it must therefore possess particle-like properties.
Particles as Waves:
Electrons through double slit: When electrons pass through a double slit, the screen that the electrons are incident on glows where electrons strike it. The result is an interference pattern similar to that produced by light. This infers that particles behave in a similar manner as light.
Even if the the intensity of electrons per unit of time decreases, the interference builds up over time (just as low intensity light does).
Davisson-Germer’s crystal lattice: In Davisson-Germer’s experiment, electrons were released from a heated filament and accelerated towards a nickel target with a crystalline surface. Since the crystal lattice possessed a very consistent structure (equal spacing between crystal planes), when particles (like electrons) were reflected through, the lattice replicated the structure of a transmission diffraction grating. Hence, the diffraction pattern produced by these particles was resemblant of that produced by light.
As David-Germer compared the wavelengths
λ=(dsinθ)/m
λ=h/mv
their results supported the de Broglie relationship - such that in order for this interference pattern to be produced, particles must possess some wave-like properties.
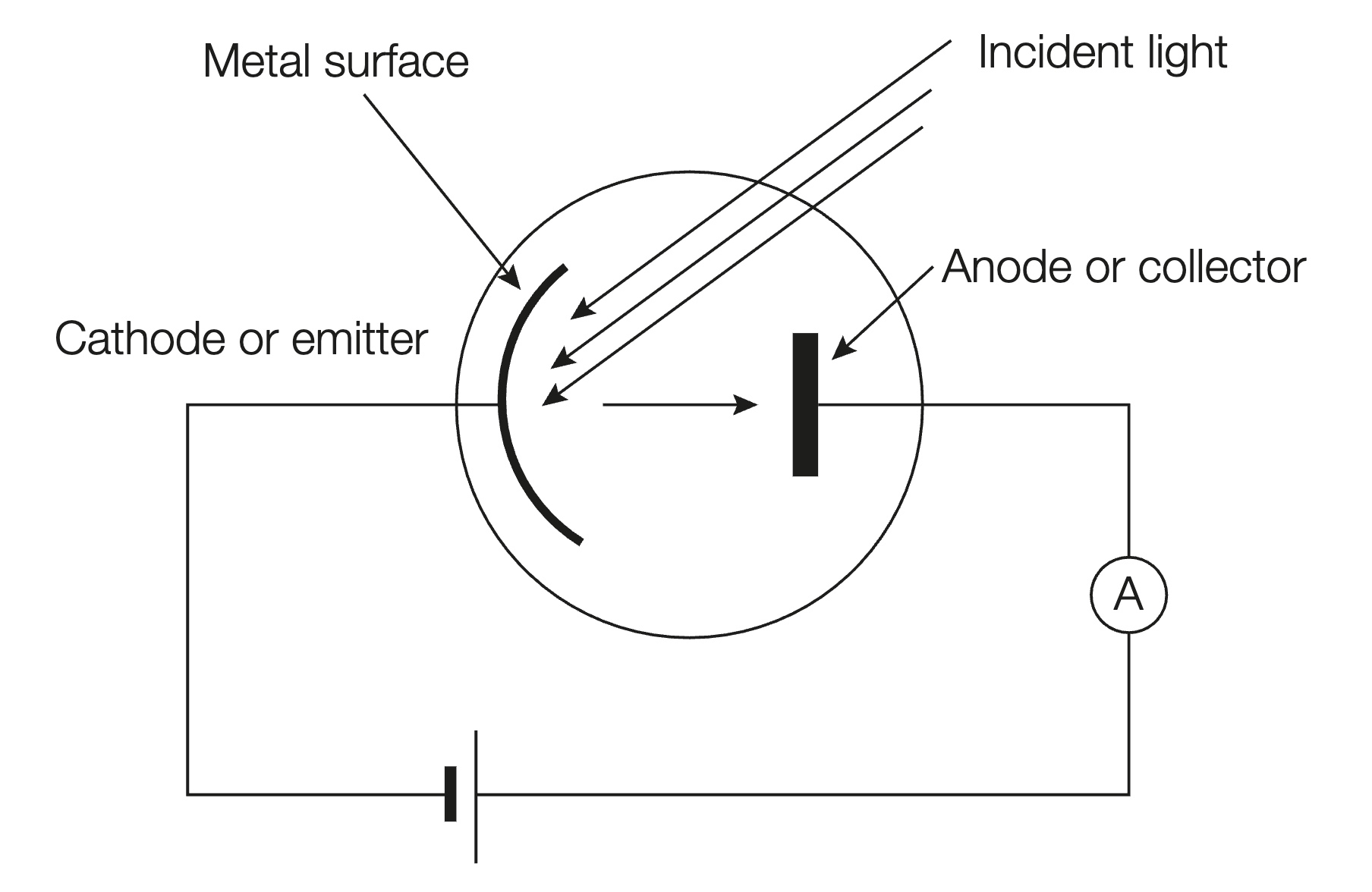
Describe the photoelectric effect
.
.
.
.
SACE Terms:
Notes:
When light of sufficient energy and frequency (E≥W & f≥f0) is incident on metal cathode, it can be absorbed, causing the emission of electrons from the metal.
The evacuated tube allows free movement of electrons towards anode without collision with air particles.
A negative potential difference can be applied to the anode and increased while measuring current. This produces an electric field that repels electrons from reaching anode.
As it is increased, the anode becomes more negative, meaning fewer electrons have the kinetic energy to overcome the repulsive force of it.
(or: as anode becomes more negative, fewer electrons have the kinetic energy to overcome it).
The energy the electrons need in order to overcome the current is given by E=eΔV.
When no more electrons can reach the anode, the current drops to zero, it means stopping voltage has been achieved, meaning the work done by the electric field is equal to the maximum kinetic energy of emitted electrons. (or: meaning the maximum kinetic energy of electrons is equal to the energy required to stop them).
EK(max) = hf-W.
How are X-rays produced in an X-tube?
.
.
.
.
SACE Terms:
Notes:
A current is passed through a filament (cathode) heating it up, causing the emission of electrons.
By applying a large positive potential difference (through high voltage supply), this does work on the electrons (via electric field), and supplies them with kinetic energy, causing them accelerate towards a target metal (usually tungsten) (anode).
Due to the law of conservation of energy, as electrons strike the target metal, their kinetic energy is lost through the emission of an X-ray.
Facts to be aware of/sort of have to know (e.g., why does target metal get so hot?):
Approximately 99% of the kinetic energy of incident electrons are lost as heat, while the rest, X-ray photons. Resultantly, the target metal (anode), subject to this heat, becomes hot.
To prevent the target from metal, water/oil flows through cooling fins, allowing heat to be drawn away from it.
Tungsten is often chosen as target metal due to its durable properties (high melting point, can withstand collisions from electrons).
X-ray tube is evacuated to allow the electrons to move freely inside towards the anode, without risking loss of kinetic energy or collisions with air particles instead.
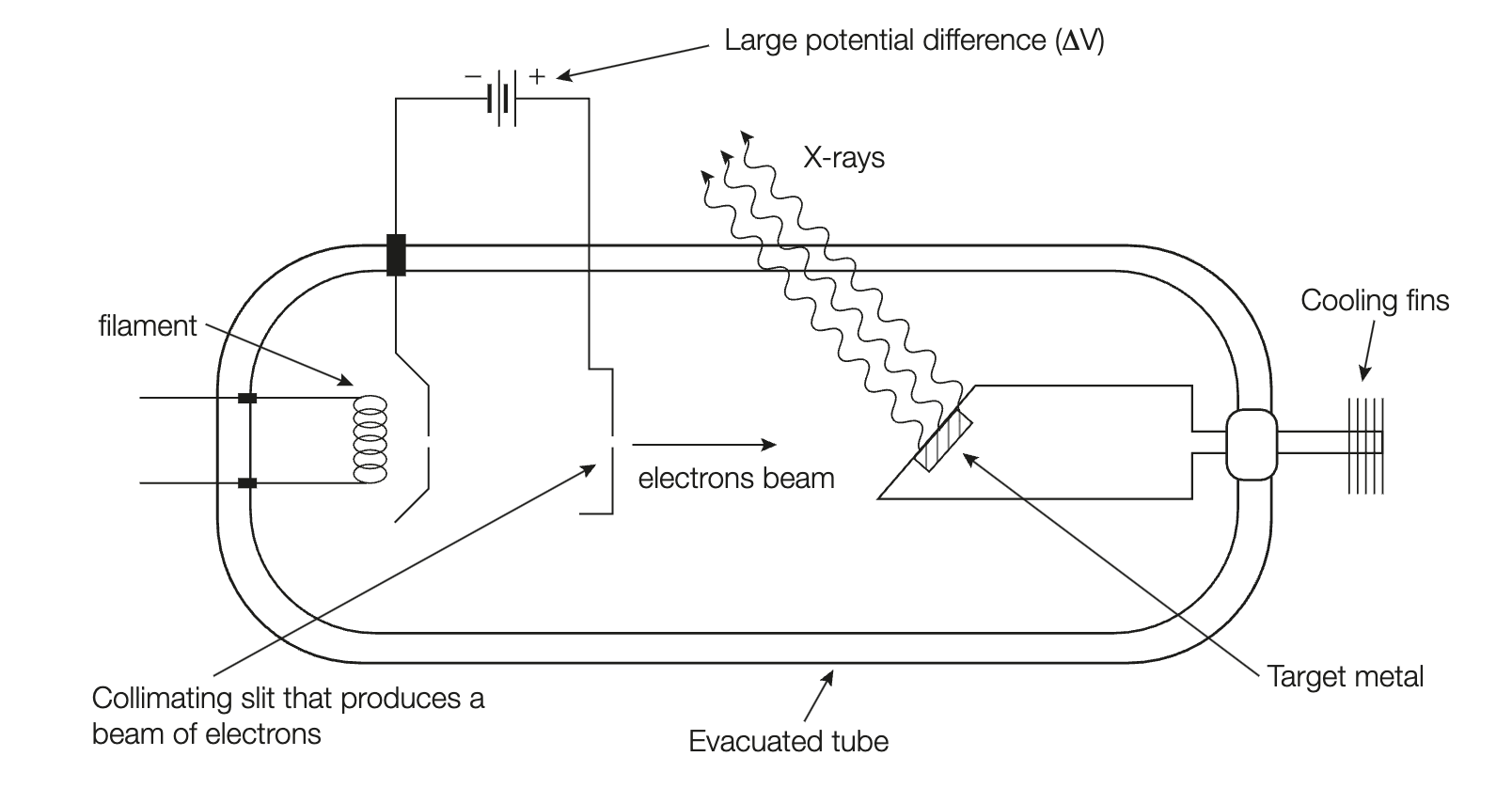
Why does the target metal of an X-ray tube get so hot?
.
.
.
.
SACE Terms:
Notes:
Approximately 99% of incident electrons’ kinetic energy is lost as heat, while the rest, kinetic energy. Resultantly, the target metal is subject to this heat causing it to heat up. Hence, cooling fins with water/oil flowing inside are implemented into the tube and serve to draw heat away from the target metal/anode (prevents target from melting).
Explain why X-rays emitted from an X-ray tube have a continuous range of frequencies up to a maximum?
.
.
.
.
SACE Terms:
Notes:
As electrons collide with the metal target, their path is deviated by the electrostatic force between the electrons and the nuclei of target atoms.
This causes them to slow down, losing kinetic energy.
As per the law of conservation of energy, energy can neither be created nor destroyed, resultantly, the energy lost is emitted as an X-ray photon.
(i.e x-ray has energy equal to the difference before and after collision with target metal)
However, this energy may vary, depending how much the electrons path was deviated, and how closely it collides with the nucleus of target atoms. For example, if the path is deviated slightly, the x-ray is emitted with energy (and hence frequency) much less compared to one which collides very closely.
This results in the continuous range of frequencies of emitted x-rays, and is represented by the bremsstrahlung (curved part of the graph).
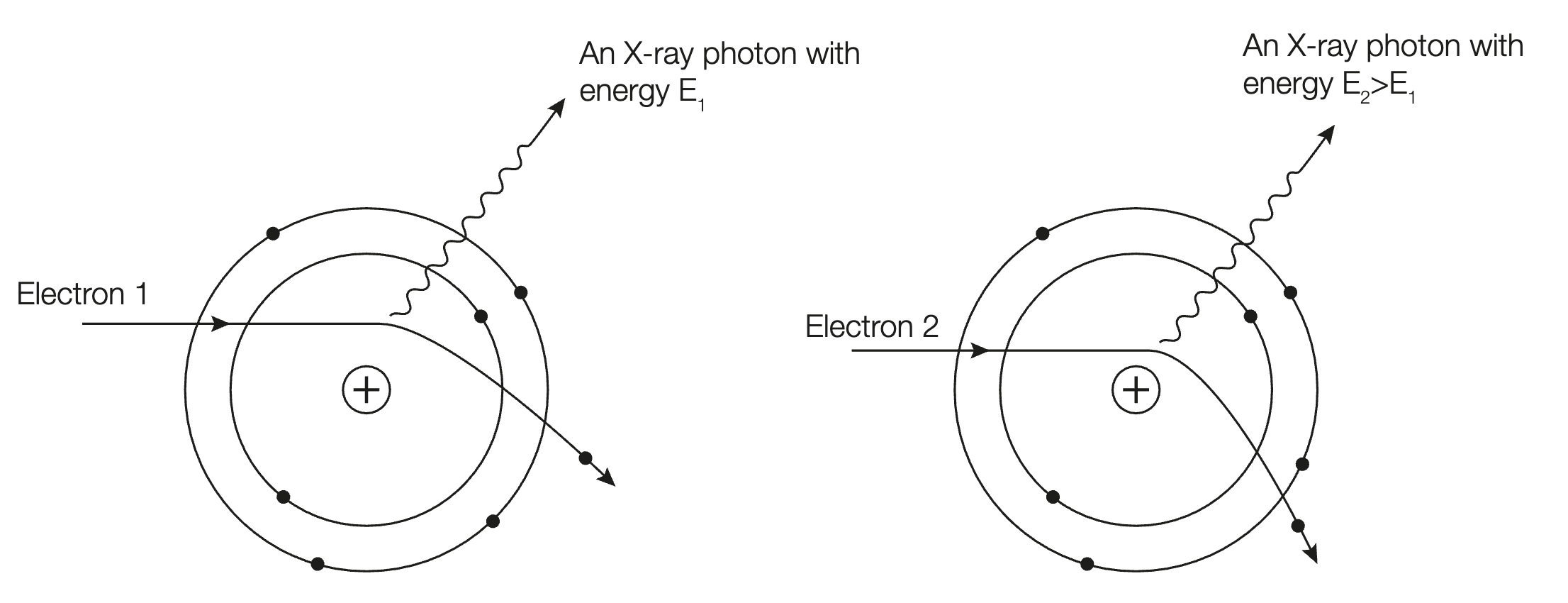
derive f(max)=
.
.
.
.
SACE Terms:
Notes:
E=eΔV, E=hf
When electrons directly collide with the nucleus of the target atom, all of their kinetic energy is transferred and emitted in an X-ray photon, resulting in a maximum frequency.
Hence, EK(max)=EK(max)
hfmax=eΔV
fmax=eΔV/h
what are factors that affect attenuation
.
.
.
.
SACE Terms:
Notes:
density of tissue
more dense=more attentuation
thickness of tissue
more thick=more attenuation
things that make up tissue
more atoms and more molecules=more attenuation
Taking effective photos
.
.
.
.
SACE Terms:
Notes:
To take effective photos, attenuation must be controlled.
low attenuation, must have high penetrating power (hard x-rays)
→ bones
if want high attenuation, must have low penetrating power (soft x-rays)
→ muscles, organs, vessels
how does increased photon intensity affect maximum kinetic energy and stopping voltage?
.
.
.
.
SACE Terms:
Notes:
It does not effect.
→ effects number electrons emitted.
one photon can be aborsebd and emit one electrons.
thus more photons mean more can be absorbed and thus more electrons emitted.
this has no effect on maximum kinetic energy,
→ Vs=ekmax/e therefrore does not effect
increasing filament current
.
.
.
.
SACE Terms:
Notes:
more x-rays released.
Explain davisson-germer experiment.
.
.
.
.
SACE Terms:
Notes:
In Davisson-Germer’s experiment, electrons were released from a heated filament and accelerated towards a nickel target with a crystalline surface. Since the crystal lattice possessed a very consistent structure (equal spacing between crystal planes), when particles (like electrons) were reflected through, the lattice replicated the structure of a transmission diffraction grating. Hence, the diffraction pattern produced by these particles was resemblant of that produced by light.
As David-Germer compared the wavelengths
λ=(dsinθ)/m
λ=h/mv
their results supported the de Broglie relationship - such that in order for this interference pattern to be produced, particles must possess some wave-like properties.
Explain line absorption spectrum, include requirements.
.
.
.
.
SACE Terms:
Notes:
Requirements for line absorption spectrum is incident light with a continuous spectrum, and an analysing device at other side (e.g spectrometer).
When light with a continuous range of frequencies (or of continuous spectrum) are incident on hydrogen gas atoms, electrons in these atoms can absorb the energy of photons which have energy exactly equal to the difference between a lower-energy state and a higher-energy state.
Resultantly, the electrons elevate to a higher-energy state, and light that is not equal to an exact frequency, goes through to other side.
Since the energy differences are discrete, these frequencies appear as dark lines on a continuous spectrum, producing line absorption spectrum.
(or: If incoming light has energy equal to the exact frequency/energy difference between two states, electrons absorb the light, and jump from ground state to energy level with the same frequency as the light).
continuous spectrum is incident on the cold gas of an element, discrete frequencies are absorbed.
Explain line emission spectrum
.
.
.
.
SACE Terms:
Notes:
If the vapour of a pure element is heated up, and it is viewed through a diffraction grating or a spectrometer, the emission of discrete frequencies of light will be observed which is unique to the gas.
Through line emission spectrums, the atoms which comprise a gas can be observed by comparing it with that of a known gas.
The presence of these discrete frequencies in the spectra serve as evidence for the different states in atoms.
Explain how line emission/absorption spectrums serve as evidence for the different states in atoms.
.
.
.
.
SACE Terms:
Notes:
Compare spontaneous emission and stimulated emission
Explain whether baryon number of each baryon is consistent with quark composition
.
.
.
.
SACE Terms:
Notes:
The baryon number for every baryon is +1, and the baryon number for every quark is +1/3.
Since every baryon comprises three quarks, it thereby follows that baryon number is consistent with quark composition when combined.
1/3 + 1/3 + 1/3 = 3/3 = 1
max energy happen when electron hits target and then all energy turns into kinetic energy therefore max energy of photon equals max energy of the photons