Chemistry A level electronic configurations and bonding and shapes of molecules
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No study sessions yet.
29 Terms
Definition of an orbital
A region around the nucleus that can hold up to 2 electrons with opposite spins
What is the difference between shells, subshells and orbitals
Orbitals are a pair of electrons
Subshells are sections of a shall
Shells are the energy level
Covalent bond definition
A strong electrostatic force of attraction between a shared pair of electrons and the nuclei of bonded atoms
How do ionic bonds dissolve in water
Water has a positive and negative end, so ions attract and separate from the lattice structure and water surrounds the ions
Lone pair of electrons
Electrons not involved in bonding
Difference between a sigma bond and a pi bond
A sigma bond’s orbitals overlap end to end, and a pi bond has a side overlap
The 3 sigma bond diagrams
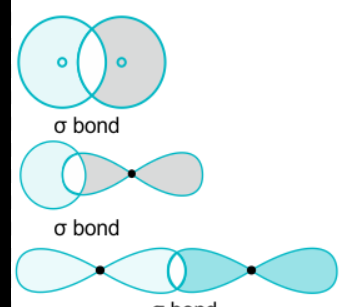
How many covalent bonds can phosphorus form
5
Can boron form covalent bonds
Yes (e.g. BF3)
Names of all the subshells
S P D F
How to calculate number of electrons in a given energy level
2n²
What is the aufbau principle?
Electrons fill the lowest energy subshells singly
Shape of an s orbital
Sphere
Shape of P orbital
Dumbell
Electron density
How likely you are to find an electron in a given position at a time
What is the heisenberg uncertainty principle
You cant know both the position of the electron and its momentum
Exceptions to the order of filling electrons
Copper and chromium, they both only have 1 electron is 4s
Angle of a tetrahedronal structure
109.5
Angle of linear structure
180
Angle of a pyramidal structure
107
Angle of non linear structre
104.5
Angle of a trigonal planar
120
Angle of tetrahedronal structure
109.5
Angles of an octohedronal shape
90
How much will a lone pair decrease the bond angle by
2.5
Symbol for molecule going into of page
dashed wedge

Symbol for molecule in line with page
line
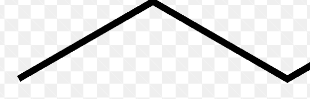
Symbol for bond coming out of page
Solid wedge

Relative atomic mass defintion
The weighted mean mass of an atom compared to 1/12 of a C-12 atom