module 5: electrochemical gradients
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
A measure of disorder (thermodynamics); diffusion increases entropy.
Movement of ions against concentration gradient using ATP
pump proteins
eg sodium-potassium pump
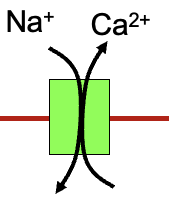
Uses energy from one ion gradient to move another ion
carrier proteins
eg sodium-calcium exchanger
Proteins that allow ions to cross membranes via facilitated diffusion.
eg potassium channels
Why do ions spread out?
Molecules in liquids are in constant motion due to thermal agitation
average center-to-center distance between water molecules?
2.8 angstroms (Å) - short distances between each other so doesn’t take long for water molecules before they start bumping into each other (psec)
what did Adolf Fick show
demonstrated that the number of molecules moving across an interface is proportional to the area of the interface and the concentration gradient
ie:
more molecules will move across a surface (like a membrane) if:
1. The surface area is larger – More space for molecules to pass through.
2. The concentration difference is bigger – More molecules on one side means a stronger push to the other side.
key idea behind Fick’s Law
Diffusion depends on the interface area and the concentration gradient
what Einstein show regarding diffusion?
Diffusion happens due to the random movement of molecules
How does diffusion depend on the number of dimensions?
In one dimension, movement is slower. In three dimensions, molecules diffuse further because they have more space to move => less chance of colliding
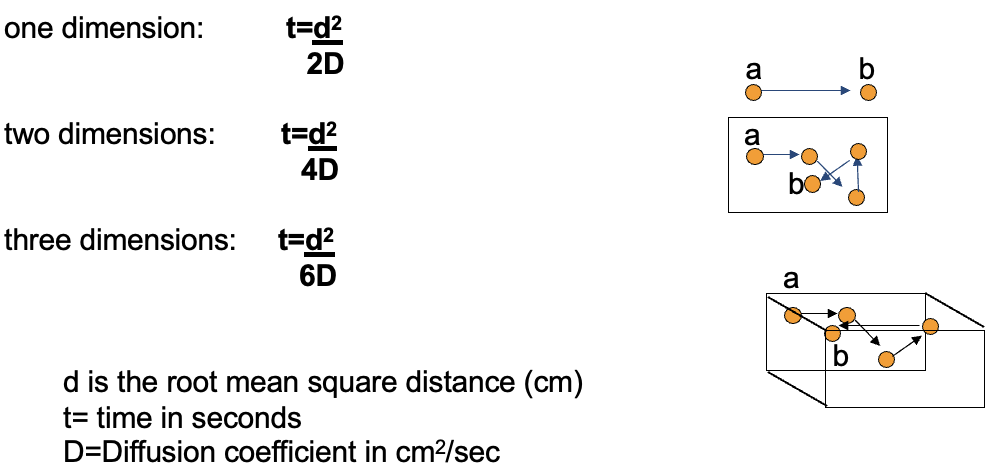
What is the biological significance of diffusion dimensions?
One-dimensional diffusion happens along DNA.
Two-dimensional diffusion happens in membranes.
Three-dimensional diffusion happens in cytosol or extracellular fluid.
How does dimensions relate to catalysts?
Catalysts provide a two-dimensional surface that increases the chances of molecules interacting
Why do signaling molecules in membranes have higher chances of interacting?
They are confined to a two-dimensional plane, which increases the probability of collisions.
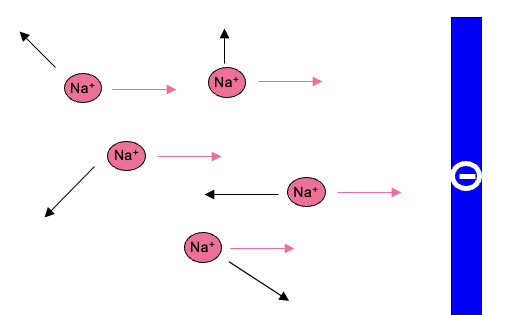
Movement of charged particles in an electric field
electrophoretic movement either adds to or subtractis from diffusion
Electrochemical Gradient (+ equation)
Total gradient combining concentration and electrical forces
determines the direction in which ions move in
(total) electrochemical gradient = gradient caused by diffusion - gradient caused by electrochemical movement
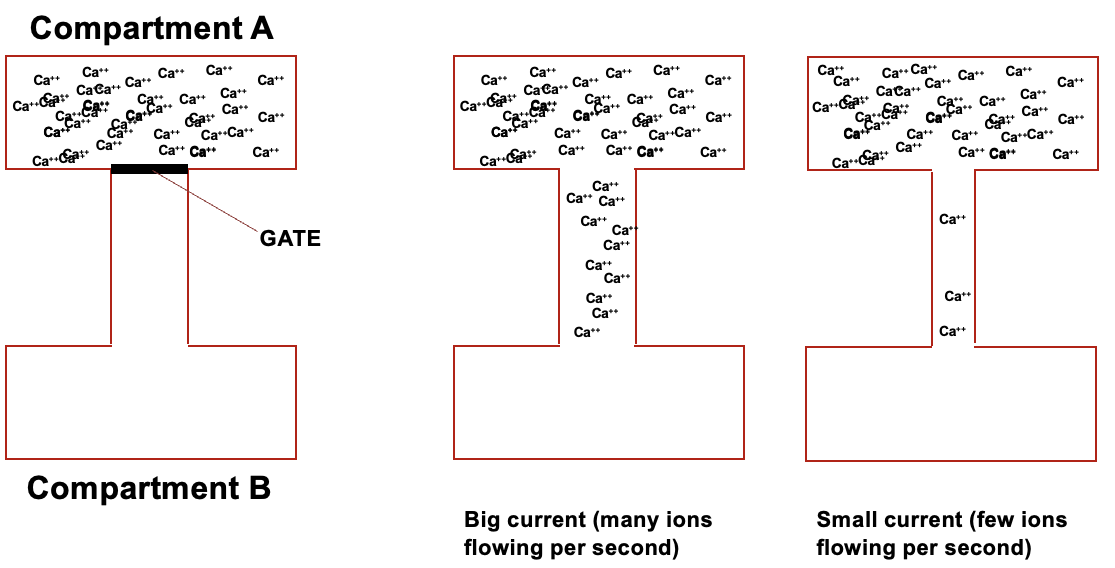
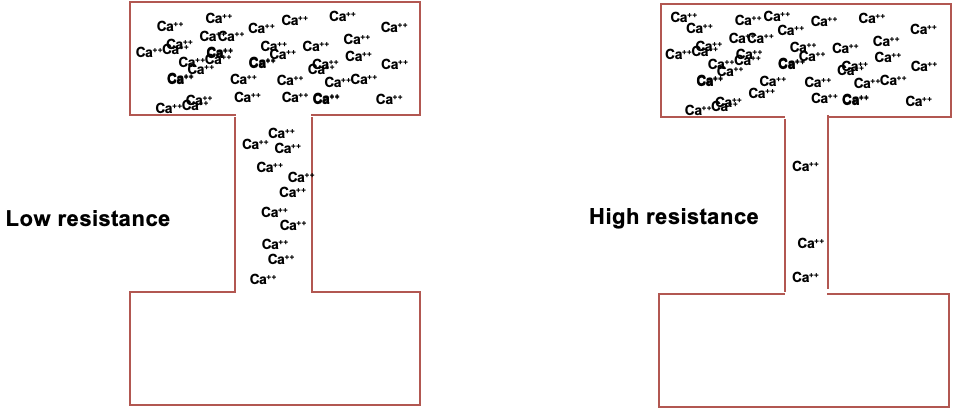
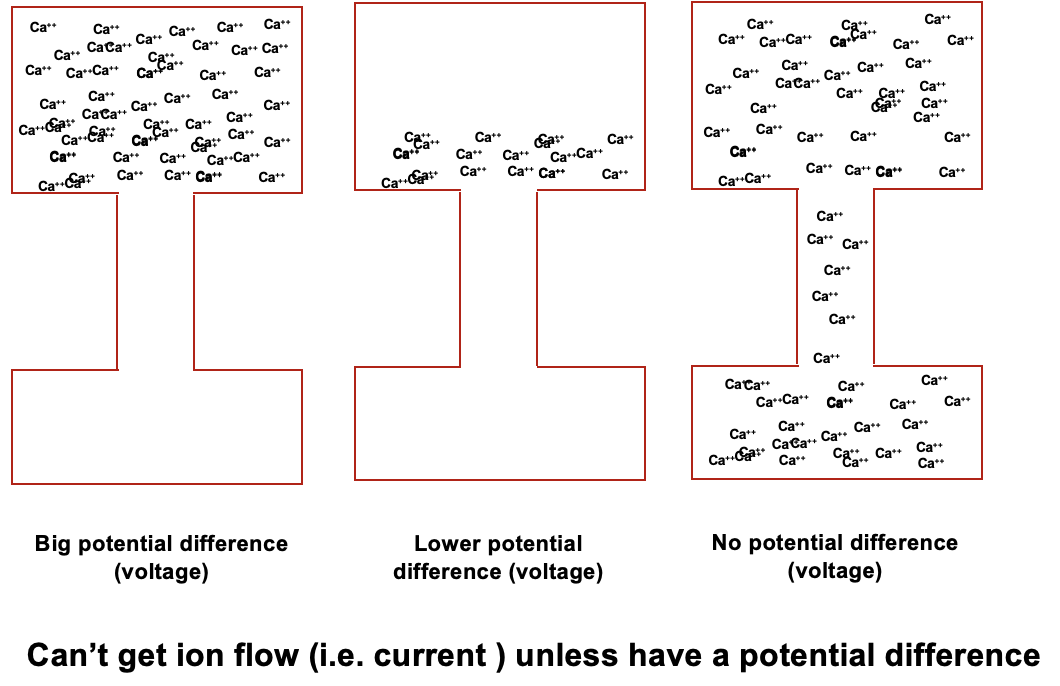
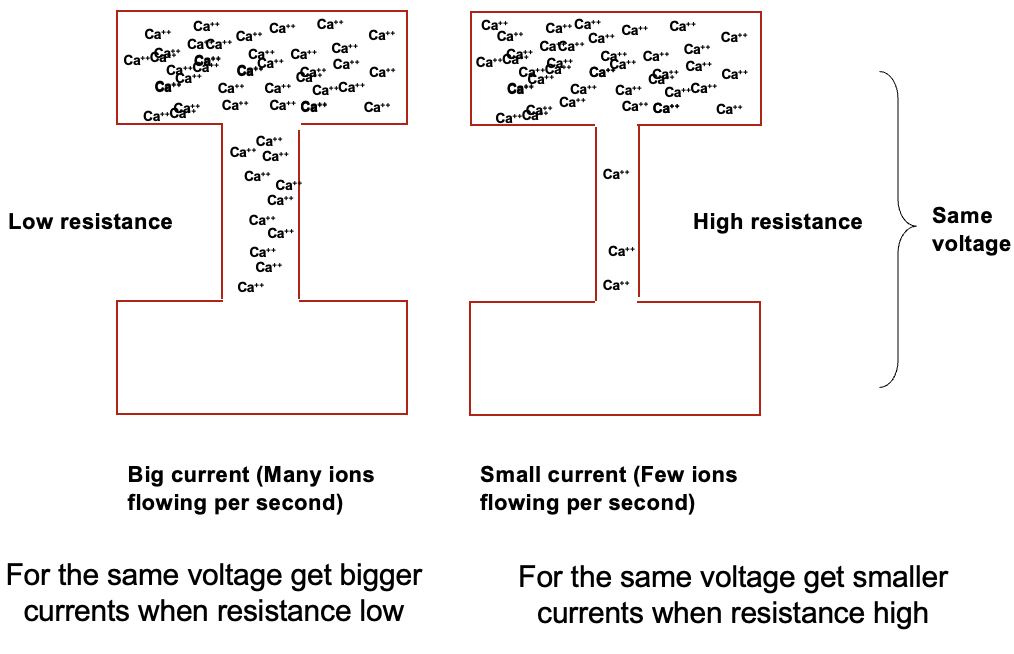
relationship between current and resistance
if you have 2 boxes of same voltage, the current can be different as it’s determined by resistance
factors which determine rate at which ions move across the membrane
size of electrochemical gradient
strongly negative on one side of membrane => ions diffuse into the cell (high electrochemical gradient)
weakly negative on one side of the membrane => low concentration gradient so less ion movement (low electrochemical gradient)
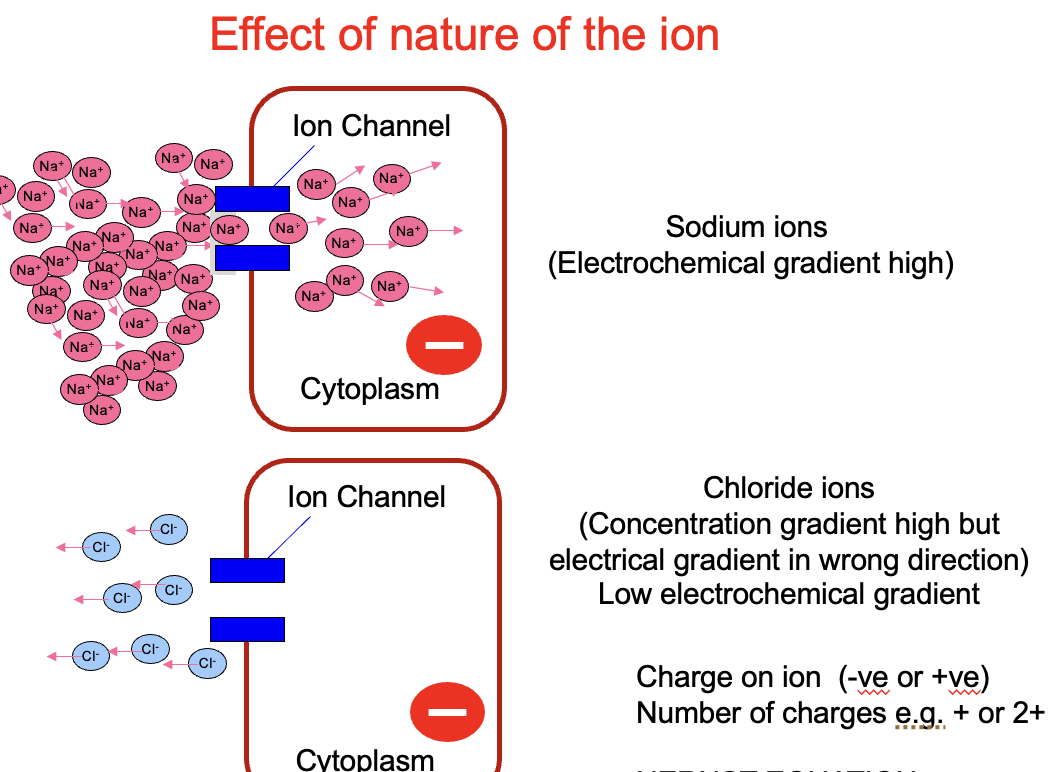
nature of the ion
sodium ion channels have higher electrochemical gradient that chloride ion channels (see image)
number of open ion channels
many open channels leads to quicker ion movement into the cell compared to less open channels
properties of the ion channel
selectivity of plasma membrane
permeability of plasma membrane