IB Exam (Unit 2)
1/31
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
U1: What does Molecular Biology teach us?
explains living processes in terms of the chemical substances involved
U2: What about Carbon makes it so useful?
can form four covalent bonds
allows a diversity of stable compounds to exist
U3: State what life is based on. List examples of it.
Life is based on carbon
Examples:
carbohydrates
lipids
proteins
nucleic acids
U4: Outline Metabolism in relation to Molecular Biology.
the web of all the enzyme-catalyzed reactions in a cell or organism
U5+6: Define Catabolism and Anabolism
Catabolism: the breakdown of complex molecules → simpler molecules
the hydrolysis of macromolecules into monomers
Anabolism: the synthesis of simpler molecules → complex molecules
formation of macromolecules from monomers by condensation reactions
A1: Outline how urea is an example of a compound that is produced by living organisms but can also be artificially synthesized
Theory of Vitalism believed vital forces gave living things life
urea is found in urine
believed to only be created through vital forces
Wöhler creates it artificially using silver isocyanate and ammonium chloride
begins disproving vitalism
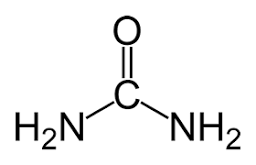
A1:
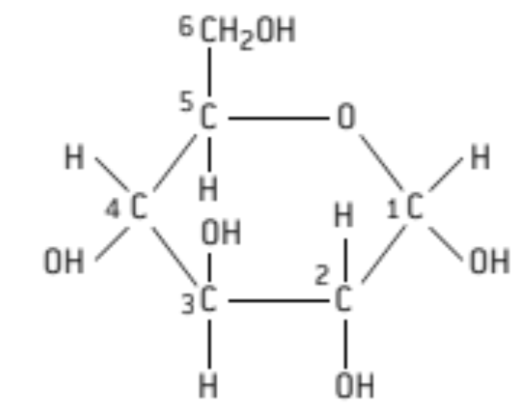
S: Draw a molecular diagram of glucose
hexagon
5Cs and 1O
C #5 has a CH2OH
C #3 has a OH on top
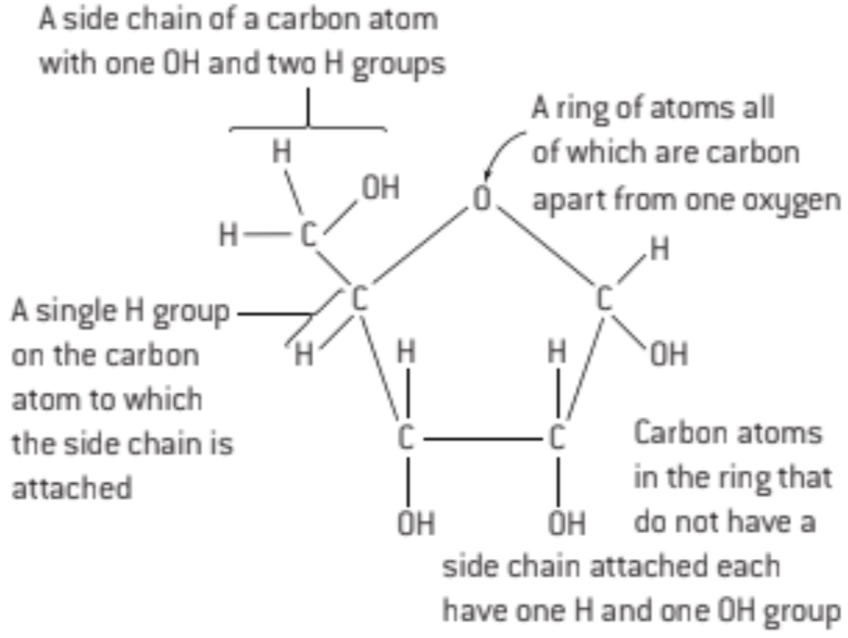
S: Draw a molecular diagram of ribose
pentagon (4C and 1O)
side chain with one -OH and 2 -H
side chan with lone H
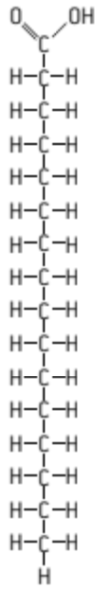
S: Draw a molecular diagram of a Saturated Fat
can also be written as:
H-(CH₂)n-COOH
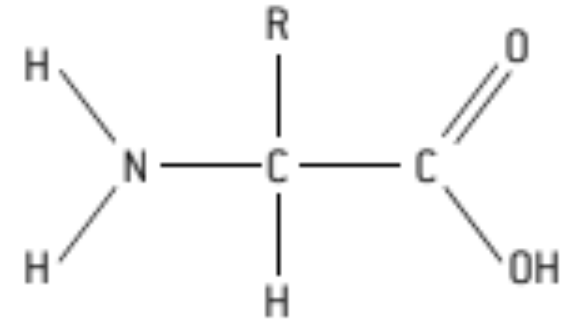
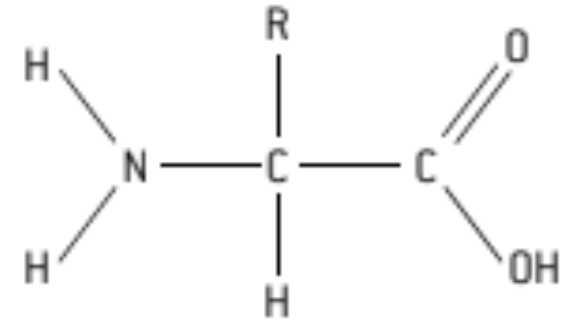
S: Draw a molecular diagram of an Amino Acid
H = hydrogen atom
NH or NH₂ amine group
(-COOH) = carboxyl group
R = r group or radical
U: Identify the qualities of a Sugar
U: Identify the qualities of a lipid
have two or three hydrocarbon chains
the quadruple ring structure of steroids
Hydrocarbon chains often shown as a zigzag line
rings of carbon atoms shown as hexagons or pentagon
U: Identify the qualities of a protein
H = hydrogen atom
NH or NH₂ = amine group
(-COOH) = carboxyl group
R = r group or radical
Look for bond between N-H (peptide bond)