Chapter 7 - Chemical Equilibrium
0.0(0)
Studied by 2 peopleCard Sorting
1/35
Earn XP
Description and Tags
Last updated 7:23 PM on 1/11/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
1
New cards
Reaction Gibbs energy
The slope of the graph of the Gibbs energy plotted against the extent of the reaction
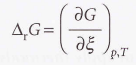
2
New cards
Exergonic
Since the process is spontaneous, it can be used to drive another process, such as another reaction, or used to do non-expansion work
3
New cards
Endergonic
The reaction can be made to occur only by doing work on it, such as electrolyzing water to reverse its spontaneous formation reaction
4
New cards
Standard reaction Gibbs energy
The difference in the standard molar Gibbs energies of the reactants and products
5
New cards
General case of a reaction
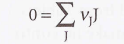
6
New cards
Thermodynamic equilibrium constant
An equilibrium constant K expressed in terms of activities
7
New cards
Le Chatelier's principle
A system at equilibrium, when subjected to a disturbance, responds in a way that tends to minimize the effect of the disturbance
8
New cards
Exothermic reactions
Increased temperature favors the reactants
9
New cards
Endothermic reactions
Increased temperature favors the products
10
New cards
Van 't Hoff equation
An expression for the slope of a plot of the equilibrium constant as a function of temperature.
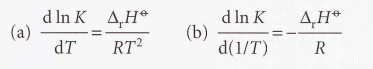
11
New cards
Electrodes
Metallic conductors
12
New cards
Electrolyte
An ionic conductor
13
New cards
Salt bridge
A tube containing a concentrated electrolyte solution that completes the electrical circuit and enables the cell to function
14
New cards
Galvanic cell
An electrochemical cell that produces electricity as a result of the spontaneous reaction occurring inside it
15
New cards
Electrolytic cell
An electrochemical cell in which a non-spontaneous reaction is driven by an external source of current
16
New cards
Oxidation
The removal of electrons from a species
17
New cards
Reduction
The addition of electrons to a species
18
New cards
Redox reaction
Reaction in which there is a transfer of electrons from one species to another
19
New cards
Reducing agent (Reductant)
The electron donor
20
New cards
Oxidizing agent (Oxidant)
The electron acceptor
21
New cards
Redox couple
The reduced and oxidized species in a half-reaction
22
New cards
Anode
The electrode at which oxidation occurs
23
New cards
Cathode
The electrode at which reduction occurs
24
New cards
Electrolyte concentration cell
The electrode compartments are identical except for the concentrations of the electrolytes
25
New cards
Electrode concentration cell
When the electrodes themselves have different concentrations, either because they are gas electrodes operating at different pressures or because they are amalgams with different concentrations
26
New cards
Liquid junction potential
An additional source of potential difference across the interface of the two electrolytes
27
New cards
Cell reaction
The reaction in the cell written on the assumption that the right-hand electrode is the cathode
28
New cards
Cell potential
This potential difference
29
New cards
Electromotive force
The resulting potential difference
30
New cards
Nernst equation
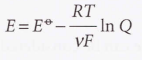
31
New cards
Standard hydrogen electrode
The specially selected electrode
32
New cards
Standard potential
The right-hand electrode and the standard hydrogen electrode is the left-hand electrode
33
New cards
Electrochemical series
The metallic elements arranged in the order of their reducing power as measured by their standard potentials in an aqueous solution
34
New cards
Ion-selective electrode
An electrode that generates a potential in response to the presence of a solution of specific ions
35
New cards
Gas-sensing electrode
Consists of a glass electrode contained in an outer sleeve filled with an aqueous solution and separated from the test solution by a membrane that is permeable to gas
36
New cards
Asymmetry potential
Observed even when the activity of the test species is the same on both sides of the membrane