Keto Enol Tautomerism
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
How do we get a keto enol tautiomer from a ketone?
Rewrite the ketone as an alcohol and add a double bond on either side

For compounds where two carbonyl groups are present where do we place the double bond?
We focus on the right carbonyl and place the double bond towards the nearest carbonyl as it is the most stable
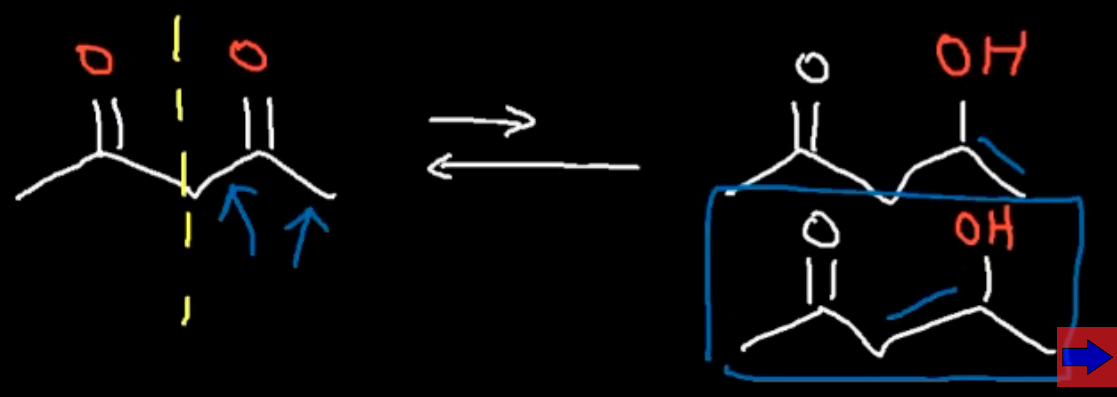
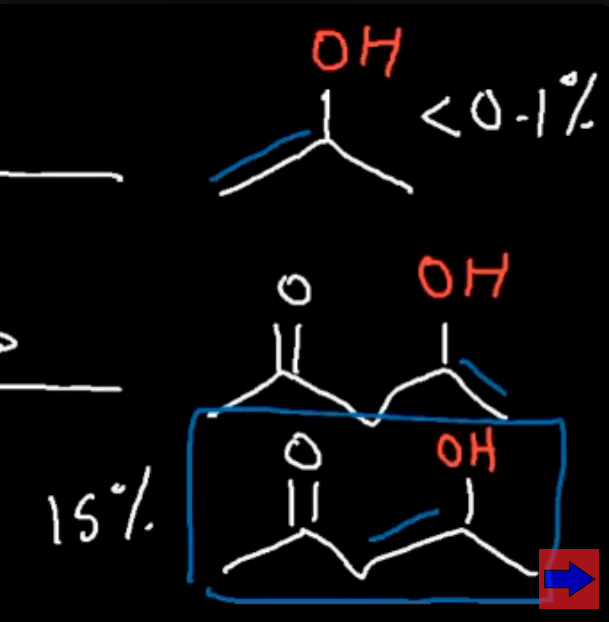
Which enol is more stable the one in our first reaction or the one in the second?
Our second enol is far more stable 15% vs 0.1%
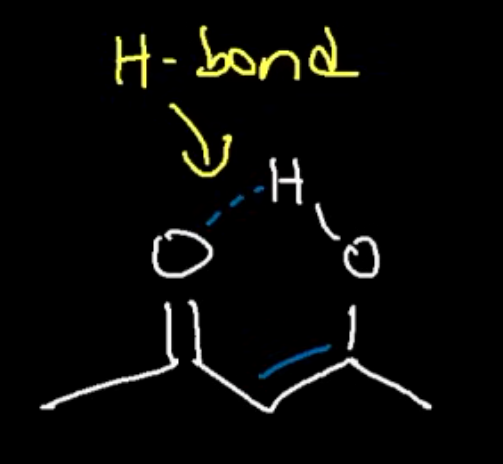
Why is our second enol more stable than the first
Due to conjugation and being stabilised by an intramolecular hydrogen bond
Are aromatic enols more stable than it’s ketone form?
Yes so the arrow would be longer for the product (equilibrium favouring the enol tautiomer rather than the ketone tautiomer)
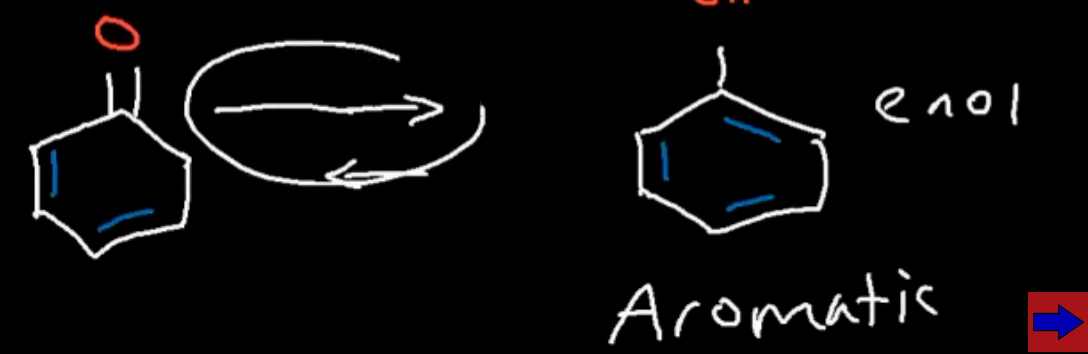
What’s special about the hydrogen which is a carbon away from the carbonyl group?
It is relatively acidic so pKa roughly around 20
Reaction of base catalysis of enol ketone tautomerism (when a base reacts with a ketone e.g OH)
Basic conditions so deprotnation occurs first
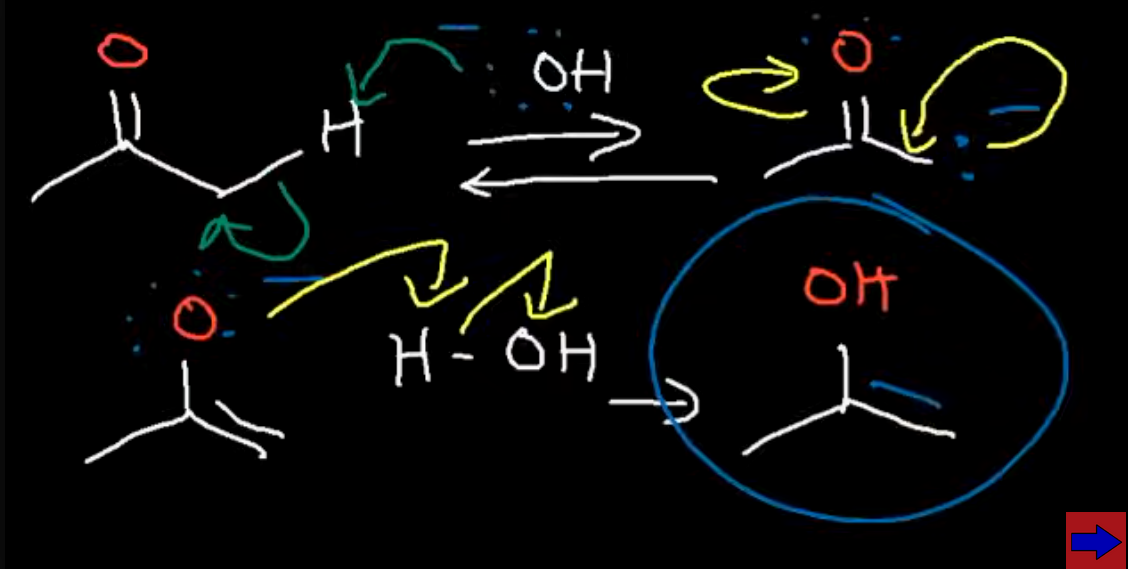
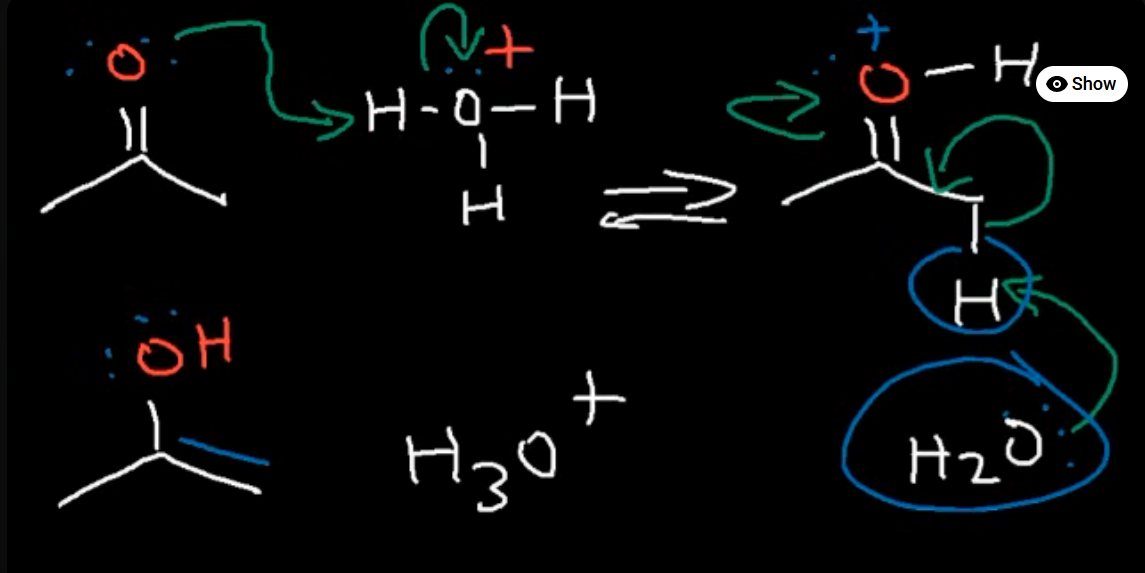
Reaction of a ketone with an acid catalyst (H3O+)
Acidic conditions so protanation occurs first and our catalyst is regenerated at the end