Resource Constraints Water
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
dehydration
its not that heat that kills, its this → leads to death
humans/mammals can only tolerate 12-14% ___
water and life
essential to organisms, most comprised of 50-90%
humans - 60-70% and jellyfish up to 95%
need to maintain salt concentrations
water is a limiting resource
water budgets
all organisms must regulate internal water concentration based on inputs and outputs, these differ between organisms
aquatic organisms water budget
Wi (internal) = Wd (drinking) - Ws (secretion) ± Wo (osmosis)
salinity in aquatic environments
different organisms are adapted to various amounts of salt (ppt)
freshwater has about 5 ppt, estuaries have about 10-25 ppt (where marine meets fresh), and oceans generally have about 35 ppt
brackish environments and salinity
lakes display range of salinity (1-400 ppt)
are also different ion concentrated, ex sodium or sulfate
diffusion
movement of particles from areas of high to low concentration due to random movement of particles
salt concentrations are equalized in solution
osmosis
a process similar to diffusion but involves the movement of water down a concentration gradient through a semipermeable membrane
ex phospholipid bilayer, permeable only to some molecules such as water
osmolarity
refers to total concentration of solute particles in a solution, describes the solute concentration relative to the volume of the solution
can also refer to an organism in relation to its environment
hypo, hyper or iso
hypoosmotic
low solute and high water (than an environment)
ex marine fish
h20 = high water
hyperosmotic
high solute and low water (than an environment)
ex freshwater fish
Isoosmotic
same solute and water (than an environment)
ex marine invertebrates
salt-water balance marine organisms
mostly isosmotic, or equal to environment
W(internal) = Wd (drinking) - Ws (secretion) ± Wo (osmosis)
many fish can be hypoosmotic and risk water loss
W(internal) = Wd (drinking) - Ws (secretion) ± Wo (osmosis)
how do marine fish prevent water loss
since many are hypoosmotic …
drink constantly to counteract dehydration, always have their mouths open
low urination rates and volumes, get rid of excess salt through specialized chloride cells in gills
salt-water balance freshwater organisms
mostly hyperosmotic (risk salt loss)
W(internal) = Wd (drinking) - Ws (secretion) ± Wo (osmosis)
how to freshwater organisms prevent salt loss
do not drink, mouth stays closed
excrete excess internal water in dilute urine
replace salts by absorbing sodium chloride in gills and by ingesting food
fish moving between salt and fresh
acclimate to their new environment
or anadromous
ex salmon, smelt, shad, striped bass, sturgeon
anadromous
born in freshwater but spend most of their life in the sea and then return to freshwater to spawn
they are able to cope with changes in salinity/water concentration through shifting secretion cells (take in salt in freshwater and excrete salt in ocean)
ex salmon
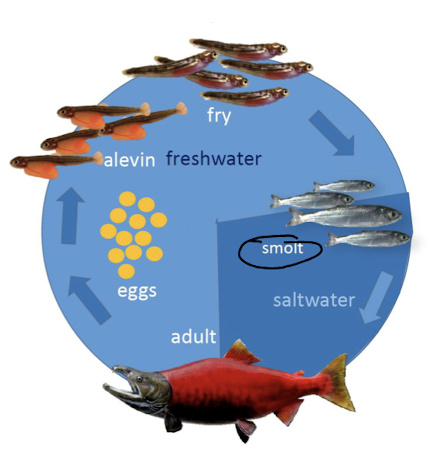
catadromous
born in the ocean, spend most of their life in freshwater and return to the ocean to spawn
ex most eels
water budgets for terrestrial systems
Wi = Wd + Wf (food) + Wa (air) - We (evaporation) - Ws (secretion)
plant water budget
Wi = Wr (roots) + Wa (air) - Wt (transpiration) - Ws (secretion)
secretion can be seeds, fruits or nectar
water potential
potential energy or capacity to do work
usually negative, so 0 (high) is pure water and -100 (low) is hot air
water moves from high → low
includes pressure, differs from osmosis
water potential equation
ψ = ψ0 (reference, pure water) + ψg (gravity) + ψs (osmotic pressure) + ψh (water vapour pressure) + ψm (matric pressure, adhesion) + ψp (sum of pressures, ET)
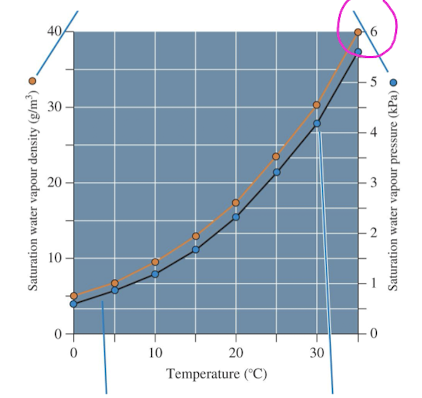
water vapour density
quantity of water vapour that air actually holds
saturation is the maximum quantity at a given temp
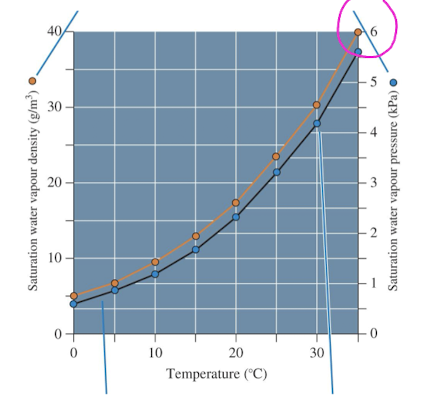
air temp and water vapour
warm air can hold more water vapour than cold air and therefore has higher pressure (closer to saturation)
but cold air can hold less water vapour than warm air and so has low pressure (low saturation)
plant water potential
tree canopy will have the lowest, due to ET
the trunk has low to moderate, with matric and osmotic pressures being low
roots have medium to high
and soil has the highest, with highest ψs
(this allows water to move up the tree/plant)
terrestrial plant water acquisition
deep roots are a strategy in plants to acquire deeper water
in drier climates roots tend to be deeper so they can access more groundwater
water acquisition terrestrial animals
Wi = Wd + Wf + Wa - We - Ws
mainly gain water via food and drink, metabolically
or via aerobic respiration
Wf is very efficient
terrestrial water conservation
thicky waxy cuticle/layer can aid in avoiding water loss by covering the epidermis
the drier the climate the more/thicker cuticle (phenotypic plasticity)
ex tree needles, beetles and snail shells
animals storing water
cacti store water in trunks and arms → low surface area to volume ratio to reduce water loss
camels store extra water in their blood stream, they also minimize water loss in excretion (concentrated urine or feces)
behavioural water conservation
kangaroo rats and scorpions use burrows where the microclimate is cooler and lower transpiration
wilting
another water conservation strategy as surface area is reduced (during water stress)
and there is a drop in turgor pressure, transpiration rate also drops as does photosynthesis