Pharmacology of Antimicrobials (Antibiotics 2)
1/66
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
67 Terms
Define pharmacokinetics
What the animal does to the drug.
Define pharmacodynamics
What the drug does to the animal/microbe.
What makes up clinical efficacy?
In vitro sensitivity data + pharmacokinetic & pharmacodynamic data
What are antibiotics?
Products produced naturally e.g. penicillin & gentamicin
Or semi-synthetic products e.g. amoxicillin
What is the mechanism of action of antibiotics?
To prevent growth and/or survival of invading organisms while minimising damage to host
Selective toxicity
Tend to target receptors or pathways unique to prokaryote cells i.e. not in eukaryotic cells
Give an example of an antibiotic that disrupts the cell wall production or function
B-lactams —> Penicillins, Cephalosporins
Give an example of an antibiotic that inhibits DNA function
Sulphonamides, Fluoroquinolones, aminocoumarins
Give an example of an antibiotic that inhibits protein synthesis
Aminoglycosides, Tetracyclines, Macrolides, Florphenicol
Give an example of an antibiotic that disrupts cell membrane function
Ionophores
What makes up the chemotherapeutic triangle?
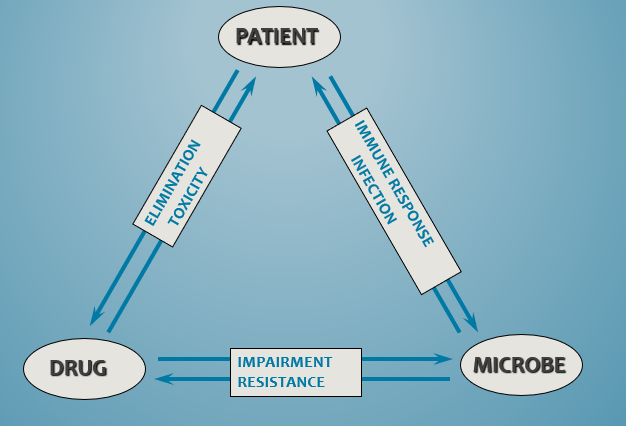
How are antibiotics classified?
How they affect bacteria
The range of bacteria they affect
Post-antibiotic effect
Whether they are concentration or time dependent in how they kill bacteria
Chemical structure and properties e.g. molecular size, lipid solubility etc….
Bacteriostatic antibiotic definition
Antibiotics that prevent replication but do not kill susceptible bacteria
Give an example of a bacteriostatic antibiotic
Tetracyclines, macrolides
Bacteriocidal antibiotic definition
Antibiotics that kill susceptible bacteria
Give an example of a bactericidal antibiotic
Fluoroquinolones, B-lactams, trimethroprim
Why can you not give bacteriostatic drugs with bactericidal drugs?
Bactericidal drugs require actively growing bacteria cells in order to function and with bacteriostatic drugs stopping them growing, the result would be antagonistic.
How can an antibiotic be bactericidal and bacteriostatic?
Dose-dependant (usually bactericidal at higher doses).
What is required for bacteriostatic antimicrobials to work?
The patients immune system must be strong enough to clear infection.
Give an example of a narrow spectrum antibiotic
Penicillin, macrolides
Give an example of a broad spectrum antibiotic
Fluoroquinolones, 2nd/3rd gen cephalosporins
What is empiric therapy?
Treatment of an infection before the organism has been identified.
What type of antibiotic does empiric treatment rely on?
Broad spectrum.
What is definitive therapy?
Treatment of an infection where the organism has been identified and a specific therapy has been chose (narrow spectrum).
What is prophylactic therapy?
Giving antibiotics to prevent an initial infection or its recurrence after infection.
What is routine prophylactic use?
Repeated, habitual use —> treating every batch of animals without attempts to reduce ongoing need to use antibiotics —> determines whether antibiotic use necessary
What is exceptional prophylactic use?
Specific non-routine situations where there is evidence/risk-based assessment showing the risk of an infection is very high and consequences are likely to be severe
What is curative treatment?
Treatment of a sick animal or group of animals following the diagnosis of infection and/or clinical disease.
What is control (metaphylaxis) treatment?
Treatment of a group of animals after the diagnosis of infection and/or clinical disease in part of the group —> aim of preventing the spread of infectious disease to animals in close contact and at considerable risk and which may already be (sub-clinically) infected.
What are categories A-D of antibiotics? Give examples of each
A = “avoid” —> rifampicin, clofazimine
B = “Restrict” —> cephalosporins, quinolones
C = “Caution” —> aminoglycosides, macrolides
D = “Prudence” —> penicilins, tetracyclines
How long does the antibiotic need to be there for (what does tis depend on?)
Mode of action —> time over minimum inhibitory conc.
Conc. dependent
Area under curve dependent
Ratio of plasma to tissue distribution may not matter as long as enough gets to where it is needed —> judged by minimum inhibitory conc. of organism
What is the post-antibiotic effect (PAE)?
The ability of a drug to suppress or kill bacteria after the drug concentration has dropped below the minimum inhibitory conc. (MIC)
(highly dependent on the antimicrobial & pathogen)
What does a good PAE mean for an antibiotic?
Should require less frequent administration.
What does it mean if an antibiotic is concentration-dependant?
The rate and extent of killing increases as drug concentration increases (until max).
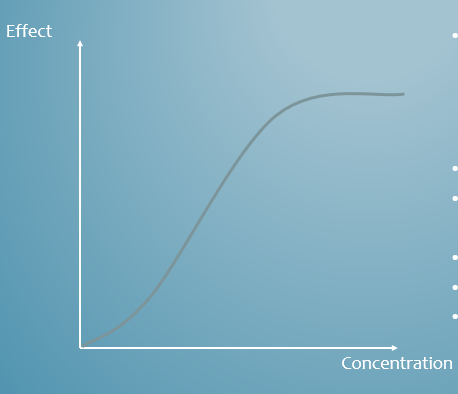
Give an example of a concentration-dependant antibiotic
Aminoglycosides, Fluoroquinolones and Metronidazole
What is the PAE for concentration-dependant antibiotics? What are dosing intervals for concentration-dependent antibiotics?
Longer PAEs
Long dosing intervals
What does it mean if an antibiotic is time-dependant?
Overall effect is strongly correlated to time above the MIC.
Bacteriocidal activity does not increase with increasing conc. once MIC been reached
Give an example of a time-dependant antibiotic
Penicillins, cephalosporins, macrolides.
What is the main aim of the pharmacokinetics of antimicrobials?
Obtain effective concentrations at the site of the infection & for an adequate duration
What things must we consider when examining the pharmacokinetics of antimicrobials
Dosage (and form)
Route of administration
Dosing rate
Access to site of infection (physicochemical properties of the drug)
Give at least 2 methods of administering antibiotics
Injectables
Topicals
Intramammary or intrauterine infusions
Tablets
Capsules
Oral suspensions
Oral pastes
Give at least 2 advantages of administering an antibiotic by injection
Can be used for drugs poorly absorbed, inactive or ineffective if given orally —> action of GI tract (gastric acid, peptidase etc.)
Poor bloody supply, immunosuppressed, dehydrated, GI infection/disease
IV route —> immediate onset of action & may get higher initial concentrations depending on tissue
IM and SC routes may be slow or delayed onset of action
Client training and compliance to give injection
Give at least 2 disadvantages of administering an antibiotic via injection
Can be painful
Cost —> product + syringe + needle
Aseptic technique required
Side effects —> higher initial concentration in heart and other tissues
MUST be soluble preparation
Needle stick injury, sharps disposal safety
What is imperative when administering an antibiotic intravenously? Why?
Must be given slowly or via infusion as the initial concentration will be very high and hence it needs time to pass through the animals body.
What needs to be considered when administering drugs IM or SC?
Formulation of dug
Conc. of drug
Vascularity of injection site
Physiochemical properties
Absorption time
Where is the injection site preferred to be in large animals (used for their meat)?
Usually lateral neck preferred for large animals (consider scarring & meat quality)
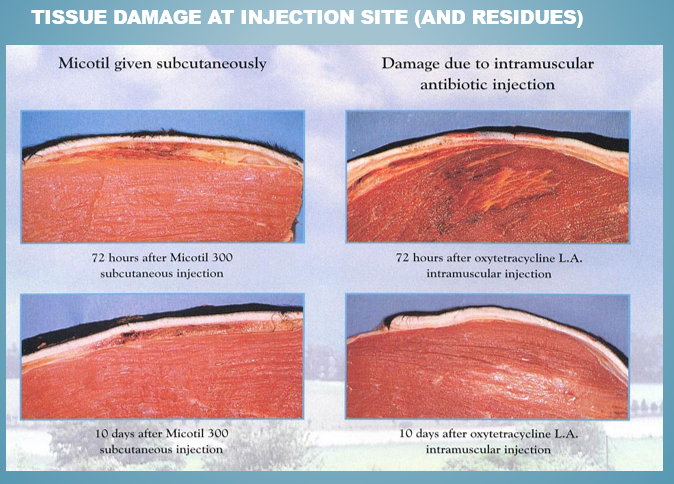
When giving drugs parenterally, what is the main consideration you should have?
What is the withdrawal period —> varies with formulation of drug & between species due to metabolism
What is a risk of giving drugs parenterally?
IM drugs can cause local tissue damage which may lead to persistance of drug residues and also hide and carcass/hide damage.
What must you consider when administering drugs orally?
Systemic availability
Metabolism of the drug before it reaches its target
What factors affect the systemic availability of an antibiotic given orally?
Stability in gastric contents (if feed is presence)
Susceptibility to inactivation (e.g. by bacteria)
Physiochemical properties
What determines the drug distrubition systemically?
Blood flow to target & metabolic rate
First pass effect (pre-systemic metabolism)
Ability to penetrate cellular barriers
Binding to plasma proteins
Presence of food or binding to food
What factors affect rate of drug distribution?
Perfusion of the tissues
Diffusion of the drug
Define volume of distribution
Measure of the apparent space in the body available to contain the drug at a steady state (not actual volume)
(Reflection of the amount left in the blood stream after the drug has been absorbed)
What does a small volume of distribution indicate?
Drug is held in the blood stream rather than diffusing into tissues.
What does a large volume of distribution indicate?
Very little drug remains in the blood stream as it has diffused into tissues.
What factors can binding of plasma proteins to antibiotics affect?
Tissue penetration
Volume of distribution
Half-life
Elimination rate
Is plasma protein binding a bad thing?
Not necessarily, for drugs like B-lactams, it allows them to be long-acting.
The larger the volume of distribution the ___ it takes to clear the drug at ___ rate of clearance
longer
constant
What is half life of antimicrobials and why is it important?
Time required for plasma concentration to half after reaching pseudo-equilibrium distribution.
Allows us to determine dosing intervals.
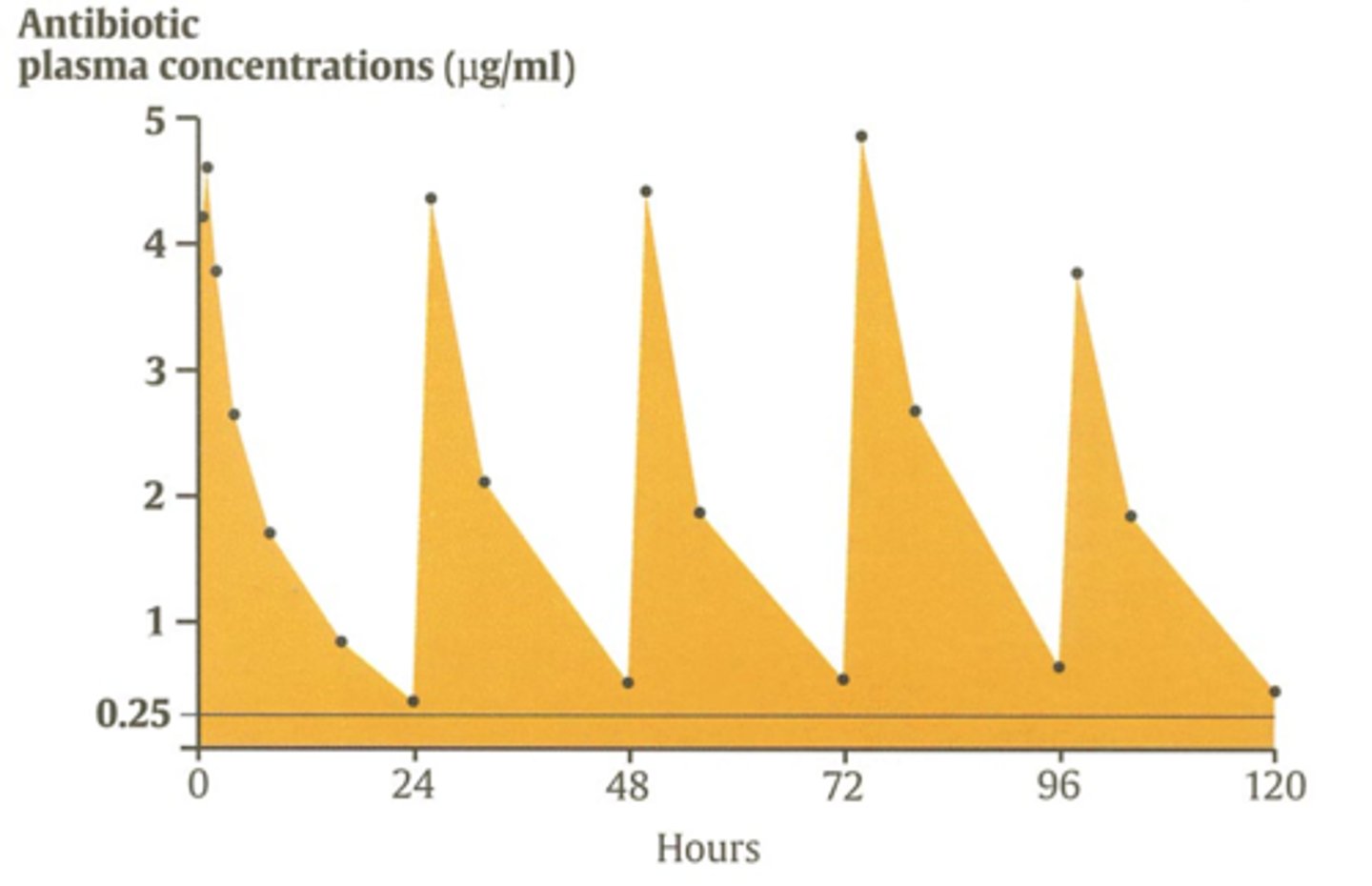
How should we determine the dose intervals of most antimicrobials?
Must use the half life to ensure that plasma concentrations of antibiotics stay above the MIC.
Compare half life with clearance
Clearance = ability to eliminate the drug from the body
Half life = overall elimination during the terminal phase which depends on both clearance and distribution.
What is synergism of drugs?
Potentiation of one drug action by another —> may be due to preventing drug metabolism or blocking bacterial metabolism in different ways
Conc. dependent
What are the main organs which eliminate drugs?
Liver (metabolism), kidney (excretion i.e. unchanged).
What are the lesser methods of eliminating drugs (list at least 2)
bile
sweat
milk
faeces (for oral antibiotics with poor absorption)
What does the unchanged fraction measure?
The amount of drug excreted by the kidney.
What does 1-fu measure?
The amount of drug metabolised by the liver.
What factors affect the amount of drug elimination that can occur in the liver?
Blood flow to the liver
Activity of the enzymes in the liver (yield/functional capacity)
What factors may reduce elimination in the liver?
Elderly have poorer blood flow
Neonates have a low liver enzyme activity
Some drugs reduce liver enzyme activity
Extensive liver damage (cirrhosis, liver fluke)