Atomic Structures Study Guide
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
32 Terms
What are the three subatomic particles?
Protons, Neutrons, & Electroms
What is the symbol for a proton?
p+
What is the relative mass of a proton?
1 AMU
Where is the proton located?
Nucleus
What is the charge on protons?
Positive
What determines the identity of an element?
The number of protons in an atom.
What is the symbol for a neutron?
N
What is the relative mass for a neutron?
1 AMU
Where are neutrons located?
Nucleus
What is the charge of a neutron?
No charge; neutral.
What do neutrons determine?
Isotope
What is the symbol for electrons?
e-
Where are electrons located?
Electron Cloud
What is the charge of electrons?
Negative
What do electrons determine?
Chemical properties & reactivity of an atom.
What does the atomic number tell you?
Number of protons in an atom.
What determines mass of an element?
The mass number
What determines chemical properties?
Electrons
What is an isotope?
An atom of the same element with a different number of neutrons.
Write the three different isotope notations:
Hyphen notation: Carbon-14, Nuclear notation: /^{14}_{6}C, Isotope with mass number: C-14.
What is the mass number?
Sum of protons & neutrons in an atom.
What is atomic mass?
Average mass of all isotopes in an element.
What is a cation?
A positively charged ion formed when an atom loses one or more electrons.
What is an anion?
A negatively charged ion formed when an atom gains one or more electrons.
Draw a Bohr Model.
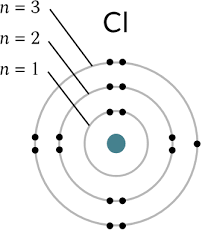
Which element does the Bohr Model work for?
The Bohr Model primarily works for hydrogen and other single-electron systems.
Which model is better for the atom?
The Quantum Mechanical Model
What are valence electrons?
Electrons located in the outermost shell.
How do you identify how many valence electrons an atom has?
Look at the group number.
Draw a Lewis Dot Structure.
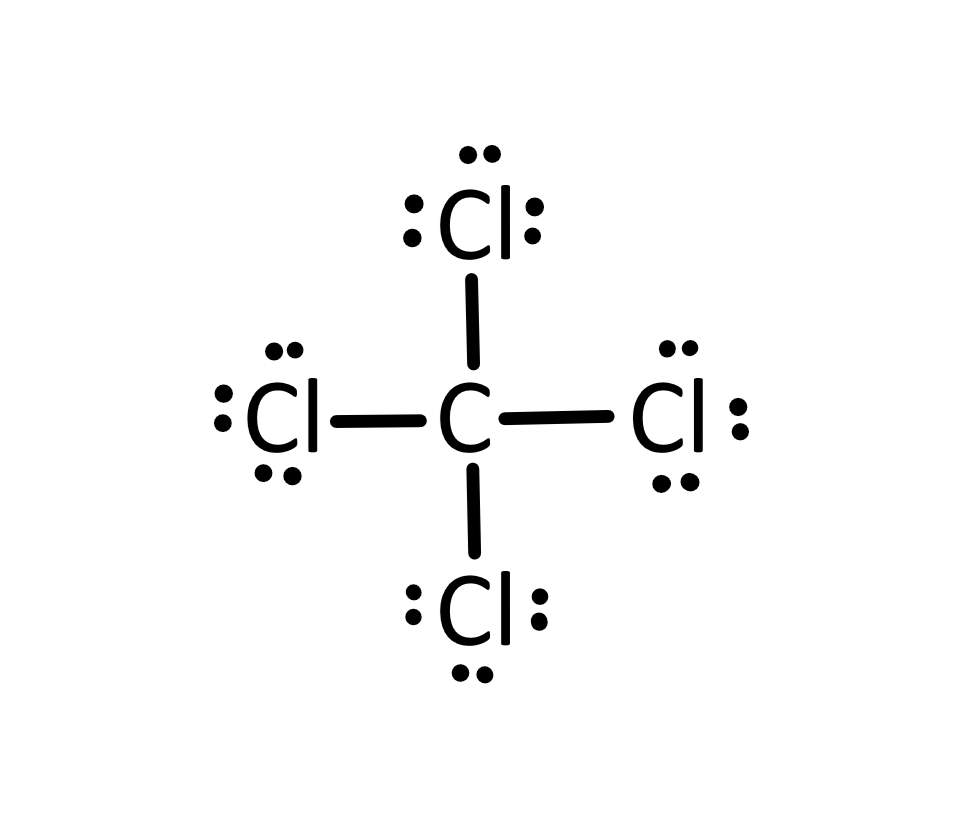
What is the octet rule?
Atoms tend to gain, lose, or share electrons to have eight electrons in their outer shell.
Be able to use the periodic table!