Enthalpy Changes
0.0(0)
Card Sorting
1/14
There's no tags or description
Looks like no tags are added yet.
Study Analytics
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
15 Terms
1
New cards
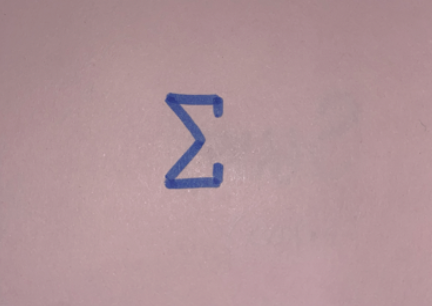
Sum
(sigma)
2
New cards
Conductivity
requires free moving charged particles
-delocalised e-
-ions
MUST BE WRITTEN EXACTLY
3
New cards
Endothermic phase changes
-melting
-boiling/evaporation
-sublimation
(solid > gas)
4
New cards
Exothermic Defn
heat is released to the surroundings
5
New cards
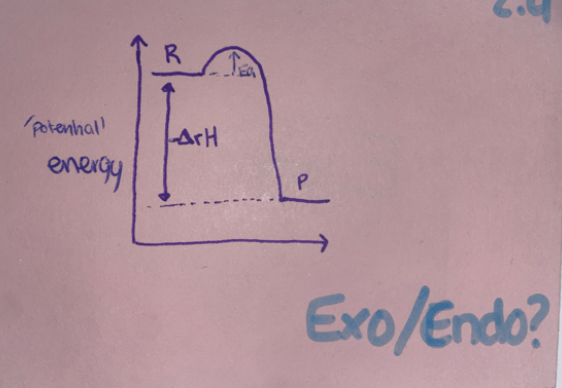
Exo
6
New cards
Exothermic phase changes
-Freezing
-Condensation
-Reverse Sublimation
(gas>solid)
7
New cards
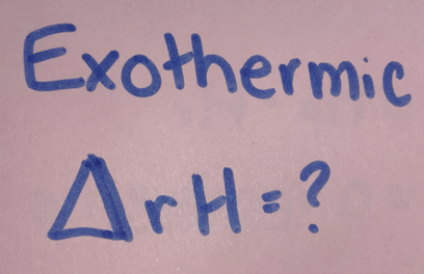
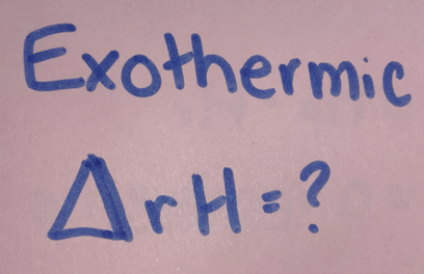
= Hp-Hr
= a negative no
8
New cards
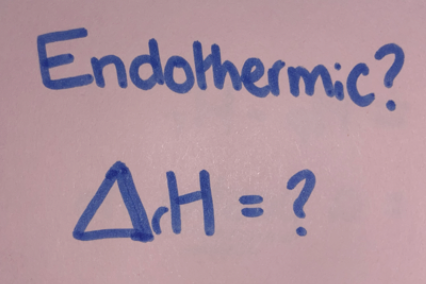
=Hp-Hr
= a positive no
9
New cards
Bond forming?
Endo/Exo?
Exo
10
New cards
Bond breaking?
Endo/Exo?
Endo
11
New cards
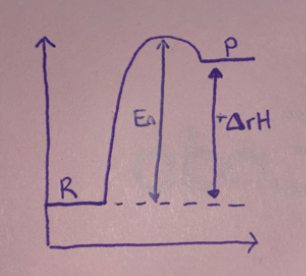

Endo/Exo?
Endo
12
New cards
Endothermic defn
Heat is absorbed from its surroundings
13
New cards
Test tube feels cold
Endo/Exo?
Endo
14
New cards
Test tube feels hot
Endo/Exo?
Exo
15
New cards
Enthalpy
The total energy (of a substance including all forms)