Unit 1 AP Biology Exam Review
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
34 Terms
TOPIC 1.1 Structure of Water and Hydrogen Bonding
ENDURING UNDERSTANDING: Living systems are organized by predator prey relationships and all of the organisms they interact with.
LEARNING OBJECTIVE
SYI-1.A Explain how the properties of water that result from its polarity and hydrogen bonding affect its biological function.
ESSENTIAL KNOWLEDGE
SYI-1.A.1 The subcomponents of biological molecules and their sequence determine the properties of that molecule.
SYI-1.A.2 Living systems depend on properties of water that result from its polarity and hydrogen bonding.
SYI-1.A.3 The hydrogen bonds between water molecules result in cohesion, adhesion, and surface tension.
Why is water polar?
Since oxygen has a higher electronegativity than hydrogen, the electrons of the molecule tend to group closer to the oxygen than to the hydrogen atoms causing an uneven distribution of electron density.
Draw a water molecule and label the charges.
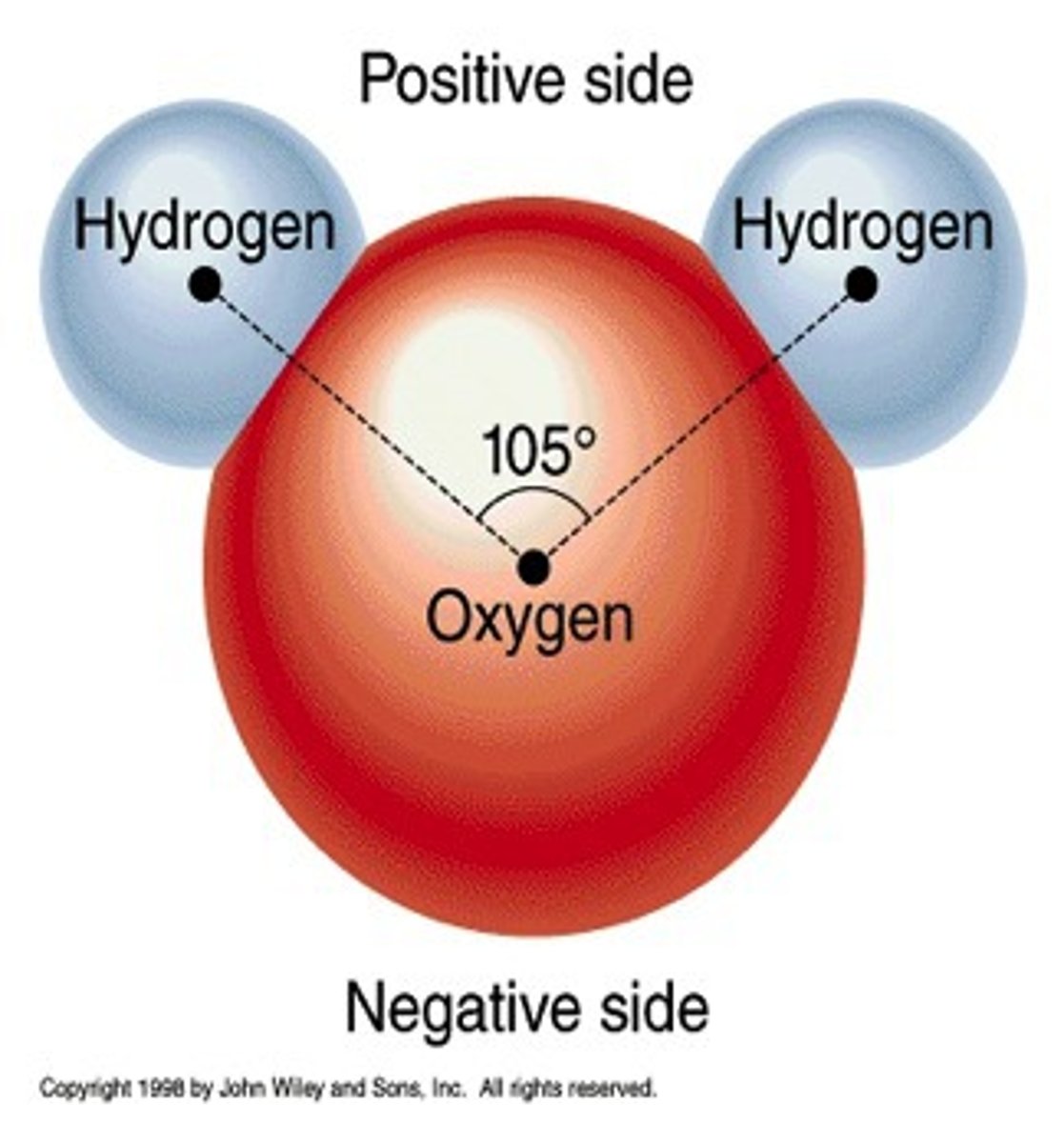
Because it is polar, water can form ______________ bonds.
hydrogen bonds/ polar bonds
Briefly explain: how do plants take advantage of adhesion and cohesion to transport water?
The adhesion helps the transportation of sticking in the stems, while cohesion pulls the water down into the roots. This is called capillary action and this process helps plants collect the necessary nutrients from water as it transports down the stem.
Draw a representation of adhesion in water molecules
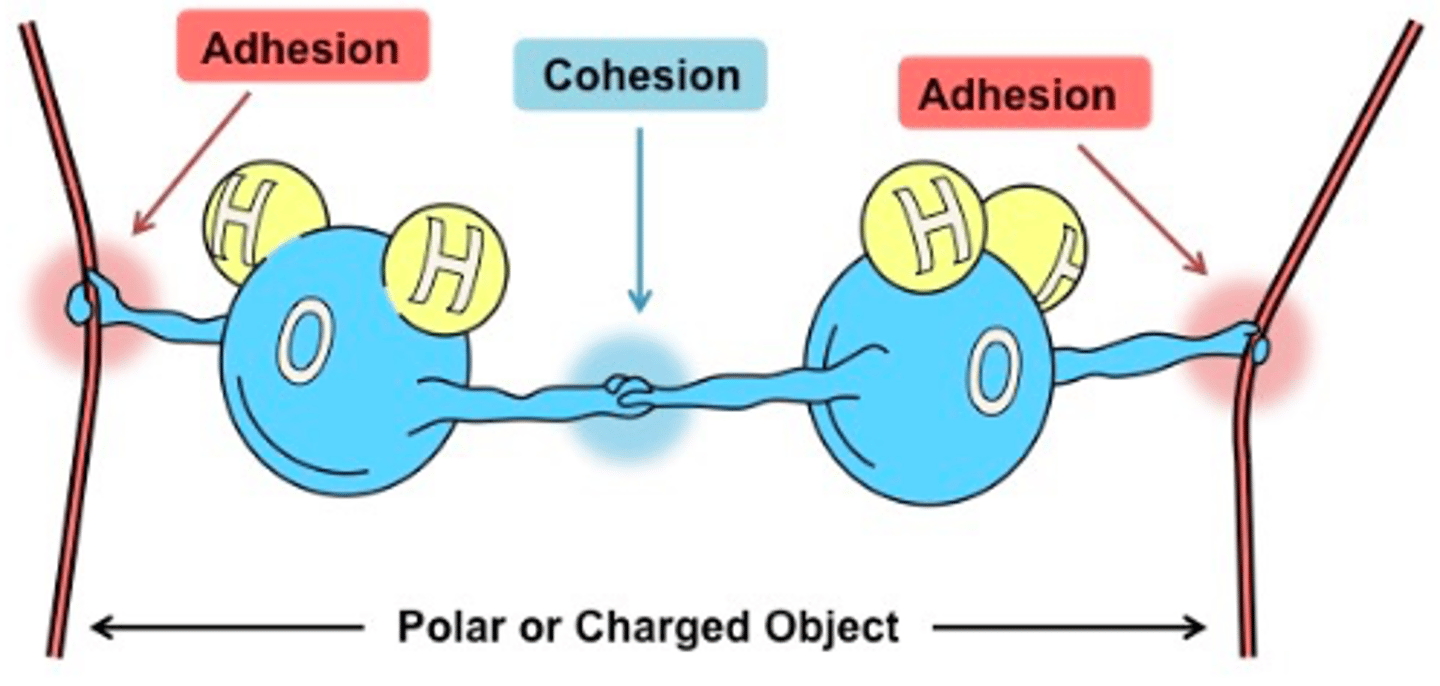
Draw a representation of cohesion in water molecules
Water is a 'universal solvent' - what does that mean?
s capable of dissolving more substances than any other liquid. ... This allows the water molecule to become attracted to many other different types of molecules.
How does the polarity of water give it this 'solvent' property?
Water molecules have a polar arrangement of the oxygen and hydrogen atoms—one side (hydrogen) has a positive electrical charge and the other side (oxygen) had a negative charge. This allows the water molecule to become attracted to many other different types of molecules.
This property of water makes it so important to life. Provide two specific biological examples of water being used as a solvent to support life.
Water helps cells transport and use substances like oxygen or nutrients
Water has a high boiling temperature which allows animals to regulate their temperature.
TOPIC 1.2 Elements of Life
ENDURING UNDERSTANDING: Ecosystems require lots of maintenance and energy with the exchange of food and energy.
LEARNING OBJECTIVE
ENE-1.A Describe the composition of macromolecules required by living organisms.
ESSENTIAL KNOWLEDGE
ENE-1.A.1 Organisms must exchange matter with the environment to grow, reproduce, and maintain organization.
ENE-1.A.2 Atoms and molecules from the environment are necessary to build new molecules—
a. Carbon is used to build biological molecules such as carbohydrates, proteins, lipids, and nucleic acids. Carbon is used in storage compounds and cell formation in all organisms.
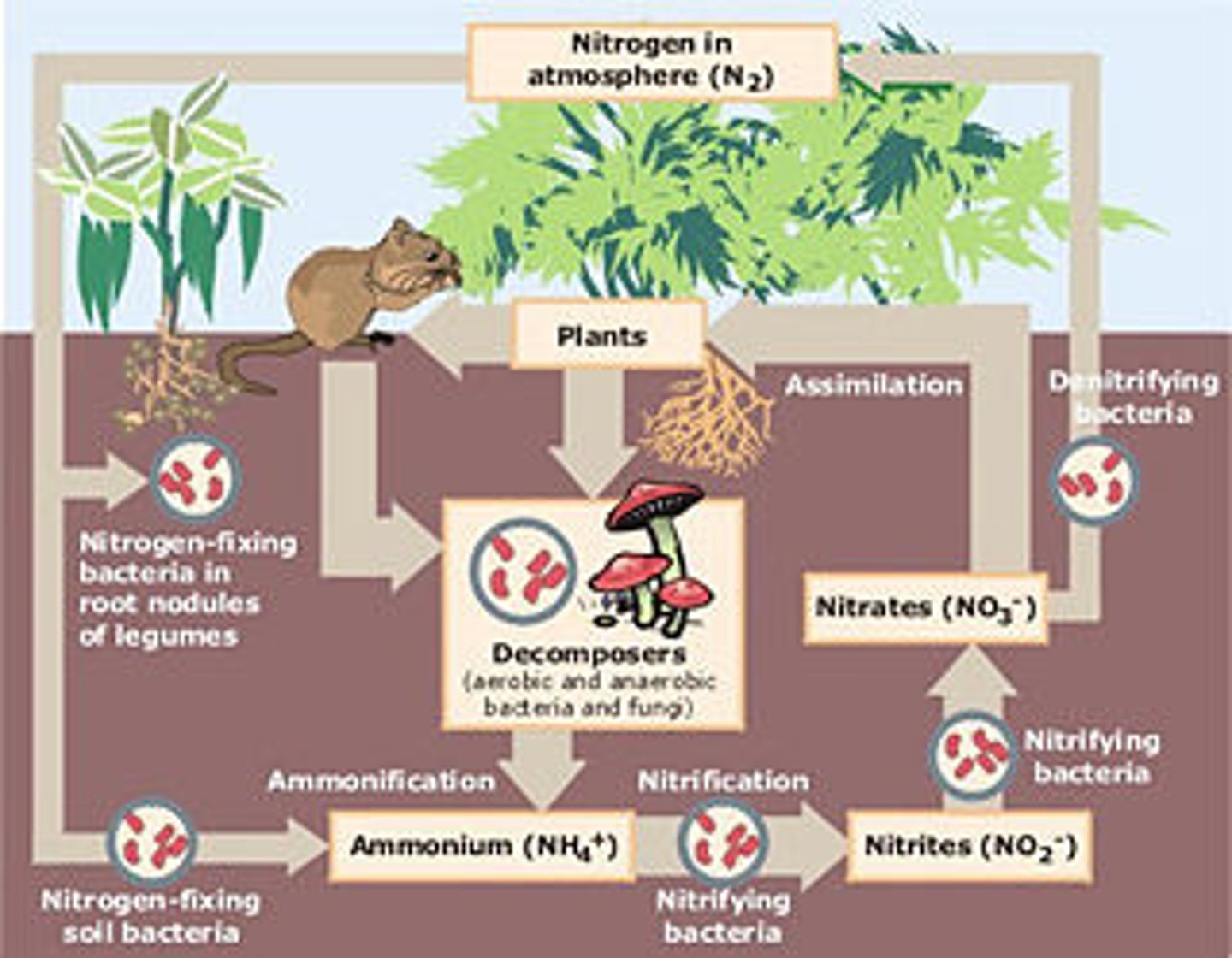
b. Nitrogen is used to build proteins and nucleic acids. Phosphorus is used to build nucleic acids and certain lipids.
List the elements that make up each macromolecule:
Carbohydrates: CHO (1:2:1 ratio)
Lipids: CHO
Proteins: CHON S (possibly sulfur)
Nucleic acids: CHONP
What element is the basis for all organic molecules?
carbon
Draw a heterotroph beside an autotroph. Add to the drawing showing how each one obtains Carbon, and how the two organisms are linked by the Carbon cycle.
Draw a Plant beside an Animal. Add to the drawing showing how each obtains Nitrogen, and how they are linked (include a Decomposer where needed).
TOPIC 1.3 Introduction to Biological Macromolecules
ENDURING UNDERSTANDING: Living systems keep each other in check and balance themselves in an ecosystem.
LEARNING OBJECTIVE
SYI-1.B Describe the properties of the monomers and the type of bonds that connect the monomers in biological macromolecules.
ESSENTIAL KNOWLEDGE
SYI-1.B.1 Hydrolysis and dehydration synthesis are used to cleave and form covalent bonds between monomers.
X The molecular structure of specific nucleotides, carbohydrate polymers, and amino acids is beyond the scope of the AP Exam
What is the name of the chemical reaction going from left to right? ________________
What is the name of the chemical reaction going from right to left? ________________
dehydration synthesis
hydrolysis
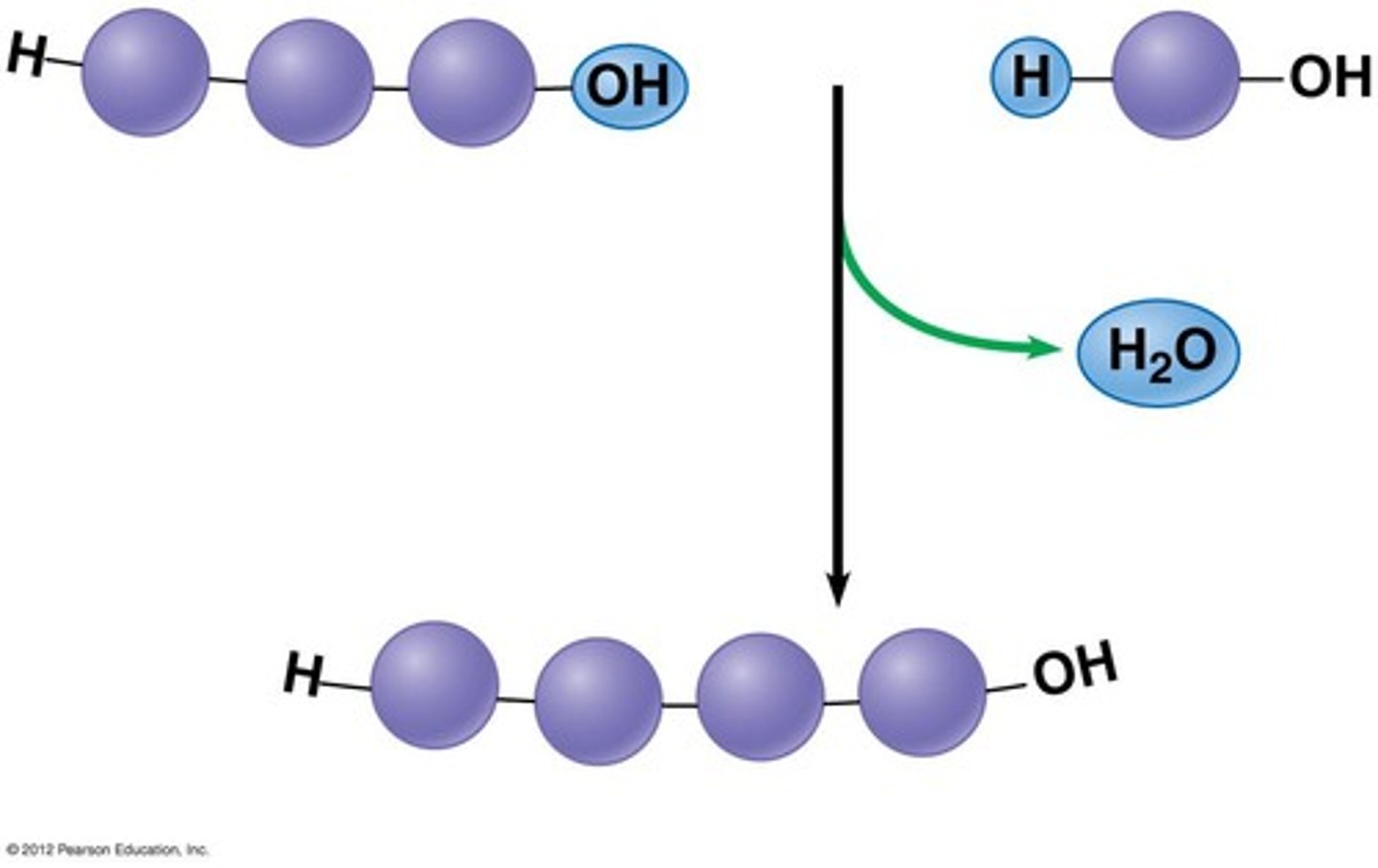
What does 'dehydration' literally mean (its word parts)? ___________________
Why is this a good word to describe the process of dehydration synthesis?
de = reduce
hydro = water
because water is taken out during this reaction and 2 compounds are merged
What does 'hydrolysis' literally mean (its word parts)? _____________________
Why is this a good word to describe the process of hydrolysis?
hydro = water
lysis = dissolution
because it is the decomposition of compounds or biological polymers into smaller molecules by a chemical reaction with water.
TOPIC 1.4 Properties of Biological Macromolecules
ENDURING UNDERSTANDING: Living systems keep each other in check and balance themselves in an ecosystem.
LEARNING OBJECTIVE
SYI-1.B Describe the properties of the monomers and the type of bonds that connect the monomers in biological macromolecules.
ESSENTIAL KNOWLEDGE
SYI-1.B.2 Structure and function of polymers are derived from the way their monomers are assembled
a. In nucleic acids, biological information is encoded in sequences of nucleotide monomers. Each nucleotide has structural components: a five-carbon sugar (deoxyribose or ribose), a phosphate, and a nitrogen base (adenine, thymine, guanine, cytosine, or uracil). DNA and RNA differ in structure and function.
b. In proteins, the specific order of amino acids in a polypeptide (primary structure) determines the overall shape of the protein. Amino acids have directionality, with an amino (NH2) terminus and a carboxyl (COOH) terminus. The R group of an amino acid can be categorized by chemical properties (hydrophobic, hydrophilic, or ionic), and the interactions of these R groups determine structure and function of that region of the protein.
c. Complex carbohydrates are comprised of sugar monomers whose structures determine the properties and functions of the molecules.
d. Lipids are nonpolar macromolecules—
Differences in saturation determine the structure and function of lipids.
Phospholipids contain polar regions that interact with other polar molecules, such as water, and with nonpolar regions that are often hydrophobic.
Nucleic Acids:
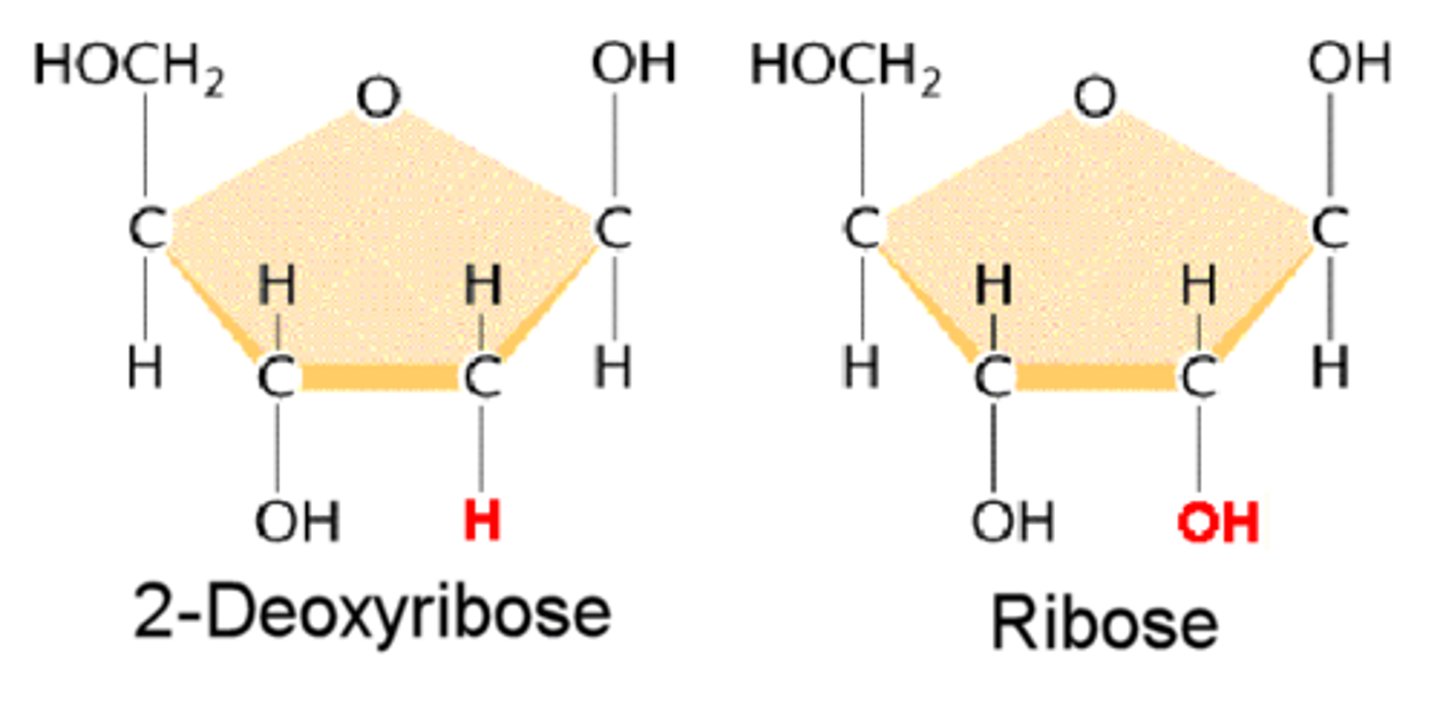
Consider the name 'de-oxy-ribose' and study the structure of each nucleotide below. Which image is the ribose nucleotide? Which is the deoxyribose nucleotide? Highlight the single difference between them.
difference = OH OH vs OH H
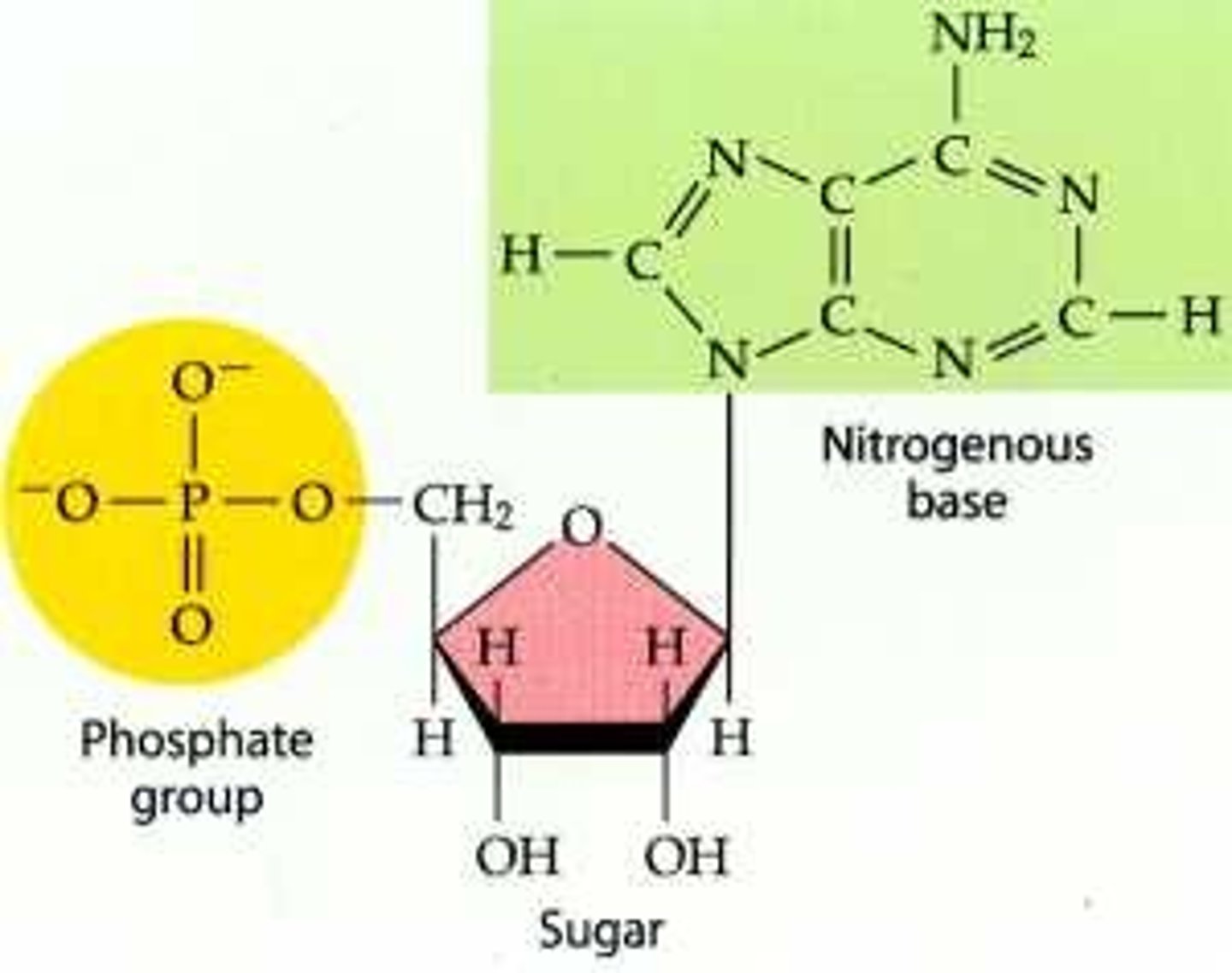
For each nucleotide monomer, circle and label the phosphate, sugar, and nitrogen base. Which part actually carries the coded biological information?
nitrogen base
Carbohydrates
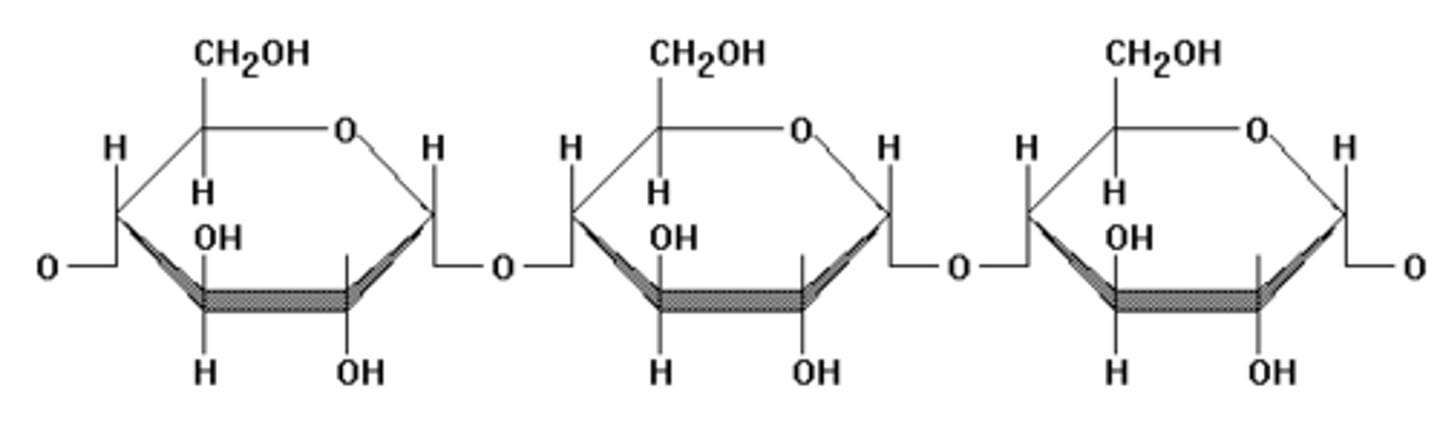
Circle a single sugar monomer.
each hexagon is a sugar monomer, so there are 3 monomers in my picture.
Proteins
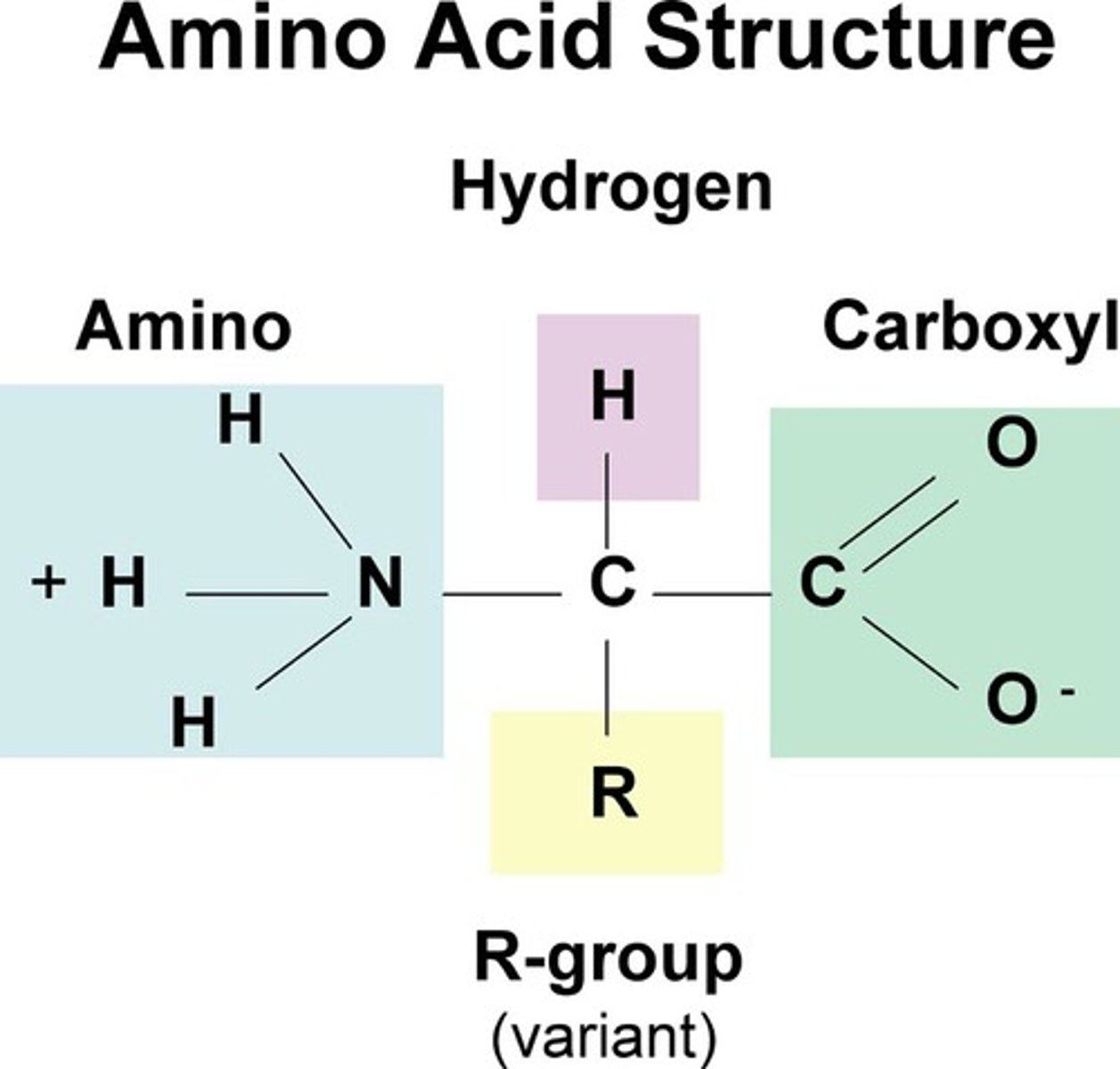
In one amino acid, circle and label the Amino terminus and Carboxyl terminus.
For each amino acid, draw a square around the R group.
Draw a heart <3 between the two amino acids with R groups that would be attracted to each other, and would cause the amino acid chain to fold so that they would be together.
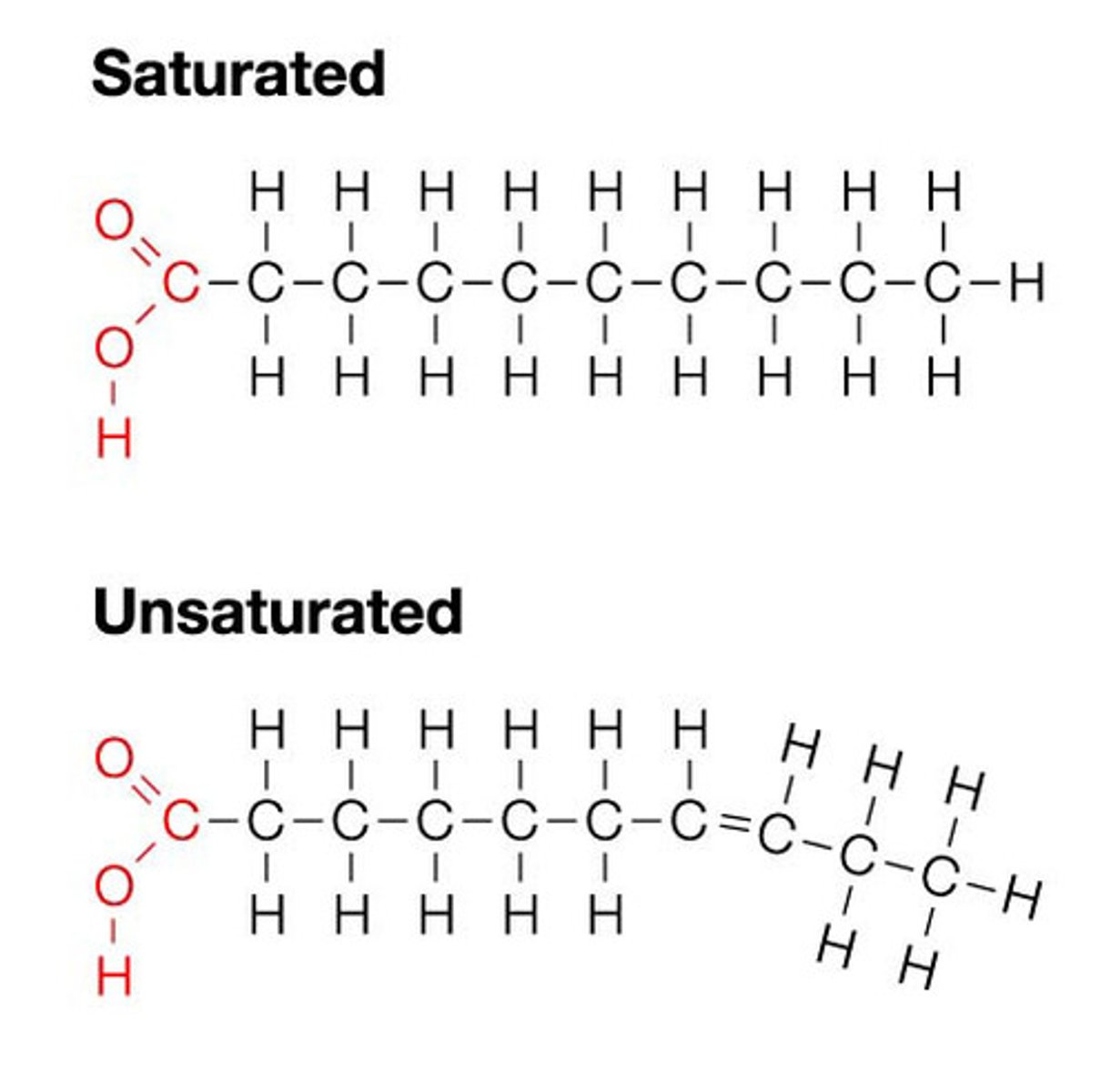
Consider what the word 'saturated' means. Which of these fatty acid chains is fully saturated with Hydrogen (a saturated fat)? Which is the Unsaturated fat?
Saturated means that it has formed a double bond and cannot be packed tightly, this is mainly used for phospholipid tails in the cell membrane.
The bottom/right one is unsaturated because it breaks off, while the top one is saturated and stays compact.
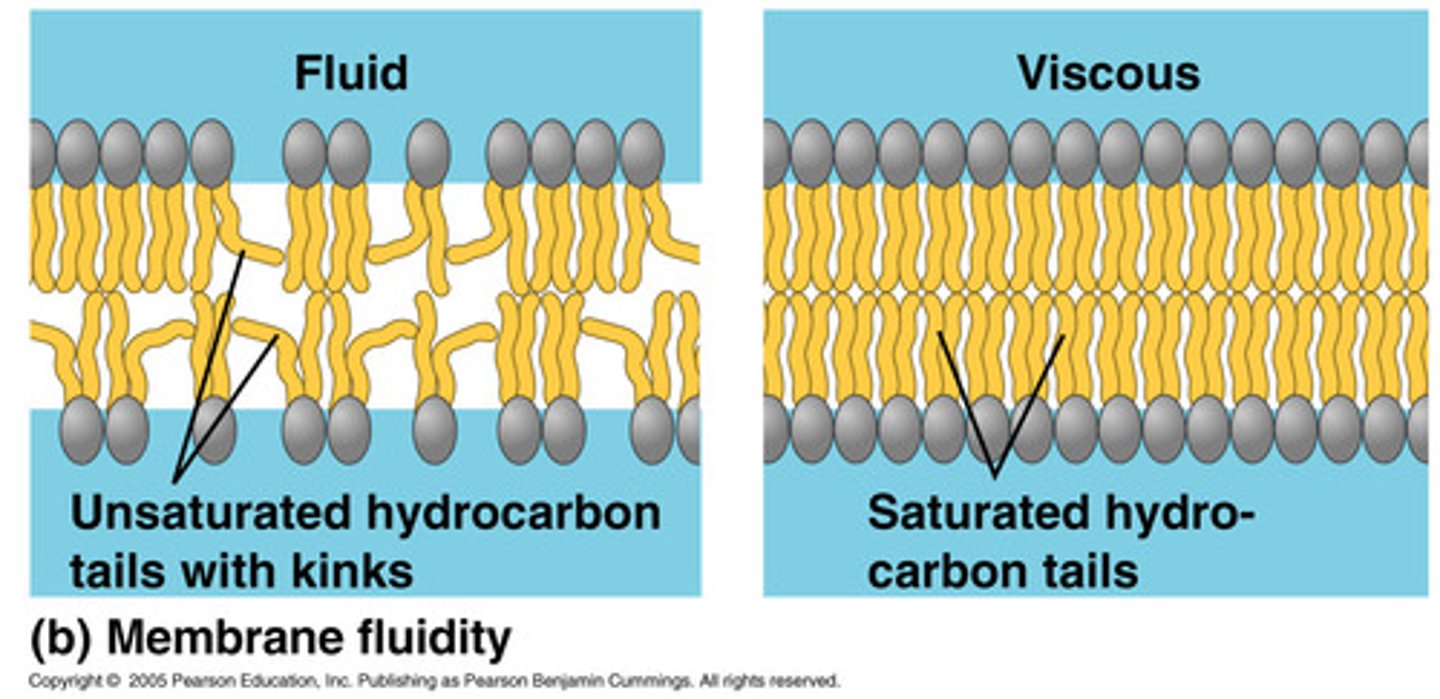
In cold temperatures, phospholipid bilayers with saturated fatty acid tails are more rigid, while phospholipid bilayers with unsaturated fatty acid tails maintain the fluidity and flexibility that cells require.
Based on the shape of each fatty acid tail, explain why this is the case.
the saturated one is more tightly packed and cannot move as easily as the unsaturated with its bent fatty acid tails
TOPIC 1.5 Structure and Function of Biological Macromolecules
ENDURING UNDERSTANDING: Living systems are organized in a hierarchy of structural levels that interact
LEARNING OBJECTIVE
SYI-1.C Explain how a change in the subunits of a polymer may lead to changes in structure or function of the macromolecule.
ESSENTIAL KNOWLEDGE
SYI-1.C.1 Directionality of the subcomponents influences structure and function of the polymer—
a. Nucleic acids have a linear sequence of nucleotides that have ends, defined by the 3' hydroxyl and 5' phosphates of the sugar in the nucleotide. During DNA and RNA synthesis, nucleotides are added to the 3' end of the growing strand, resulting in the formation of a covalent bond between nucleotides.
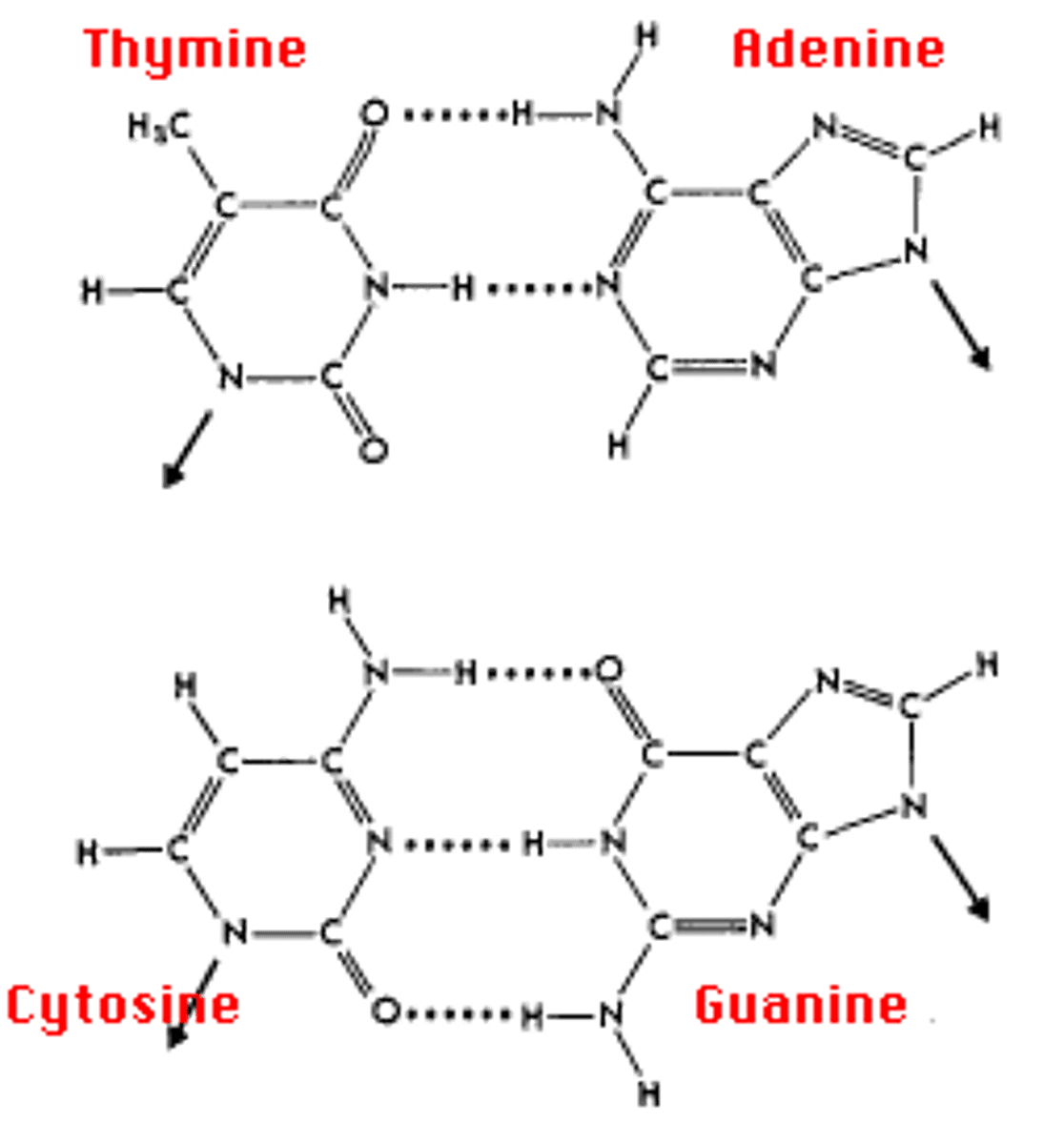
b. DNA is structured as an antiparallel double helix, with each strand running in opposite 5' to 3' orientation. Adenine nucleotides pair with thymine nucleotides via two hydrogen bonds. Cytosine nucleotides pair with guanine nucleotides by three hydrogen bonds.
c. Proteins comprise linear chains of amino acids, connected by the formation of covalent bonds at the carboxyl terminus of the growing peptide chain.
d. Proteins have primary structure determined by the sequence order of their constituent amino acids, secondary structure that arises through local folding of the amino acid chain into elements such as alpha-helices and beta-sheets, tertiary structure that is the overall three-dimensional shape of the protein and often minimizes free energy, and quaternary structure that arises from interactions between multiple polypeptide units. The four elements of protein structure determine the function of a protein.
e. Carbohydrates comprise linear chains of sugar monomers connected by covalent bonds. Carbohydrate polymers may be linear or branched.
Nucleic Acids
Choose a color for the phosphate groups and a different color for the sugars - color each of them. This is the __________ - ___________ backbone.
5' -3'
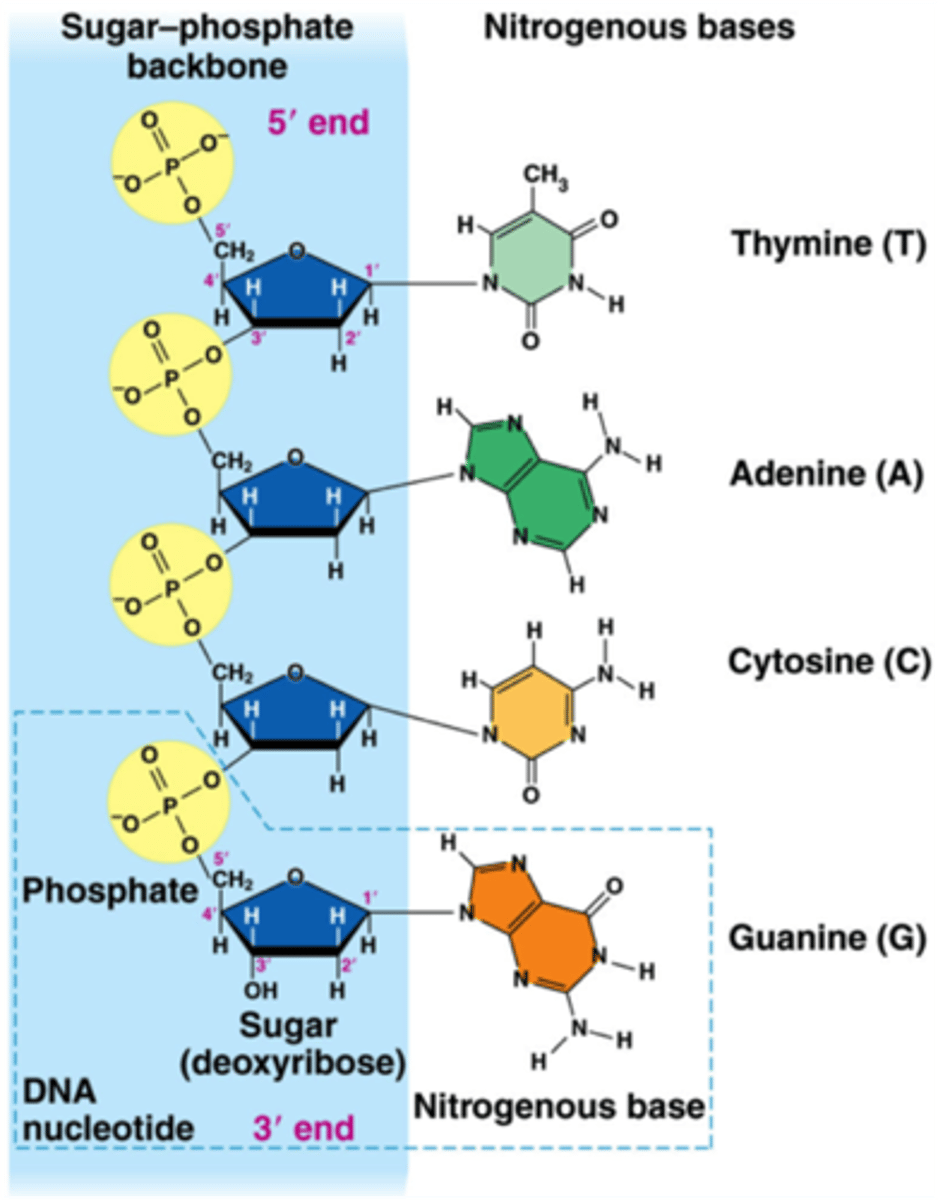
Label the 5' and 3' end of each strand (read the essential knowledge if you need help!)
Add a 'nucleotide' to each strand by drawing a rectangle to represent it - be sure you add them to the appropriate end (5' or 3')!
What bond did you just draw to connect your new nucleotides? ______________________
phosphodiester bond
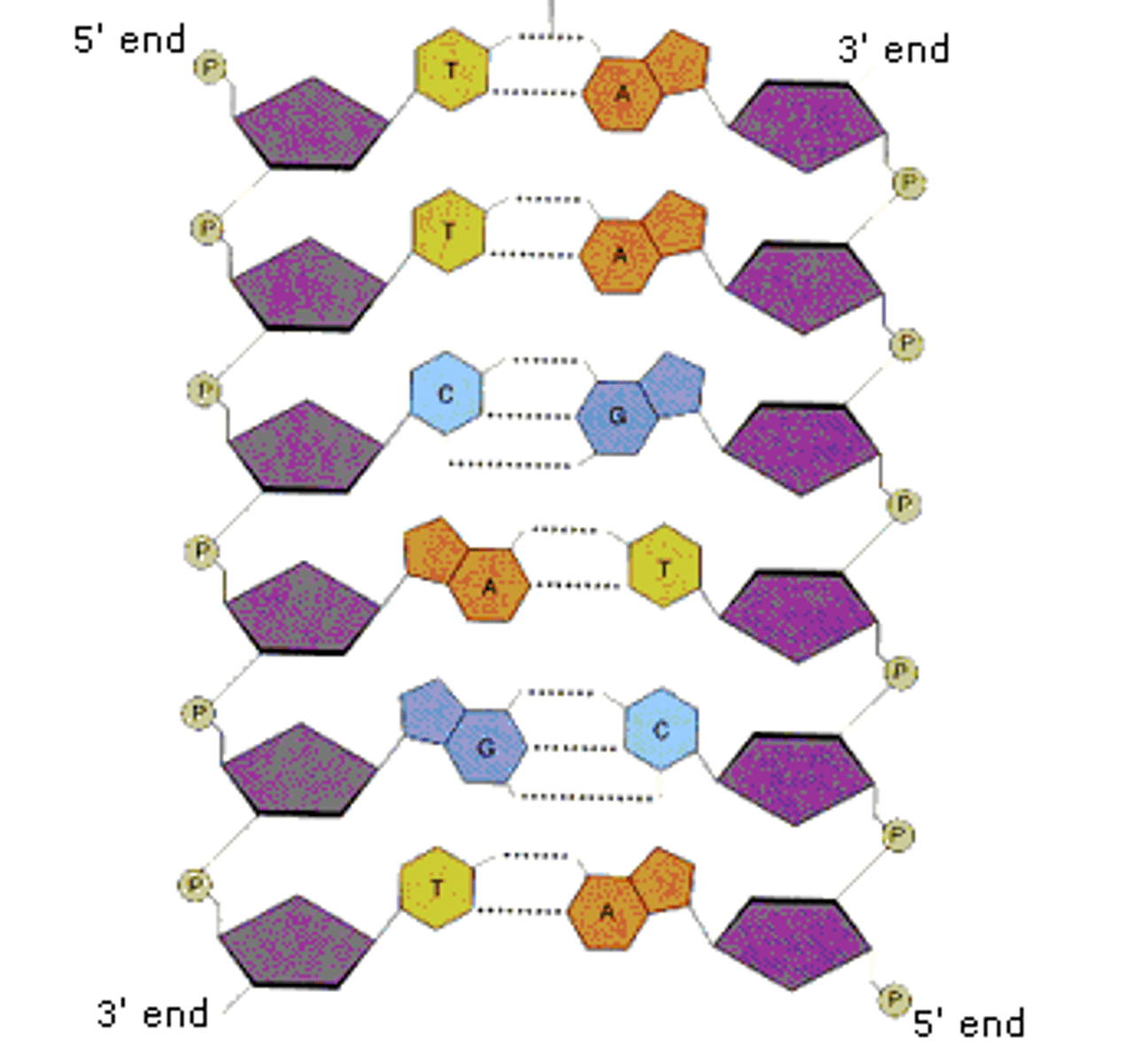
Choose a color for hydrogen bonds. Highlight the two bonds between the A - T and the three bonds between the G - C.
Which strand of DNA would be more stable (less likely to separate into two strands)? Explain why.
TCTTAGATTAT
AGAATCTAATA
GGATCGCCGAT
CCTAGCGGCTA
The G-C bond because there is more bonds which produces a stronger connection.
Proteins:
Draw a very simple diagram that represents each level of structure to protein folding.
- Primary
- Secondary
- Tertiary
- Quaternary
Primary structure:
- Amino acid sequence
Secondary structure:
- A helix
- beta sheet
Tertiary structure:
- Polypeptide chain
- R-groups:
- Decide structure/ function
- Hydrophobic: folds inward
- Hydrophilic: folds outward
Quaternary structure:
- Assemble subunits
- Multiple different 3D proteins working together
Carbohydrate
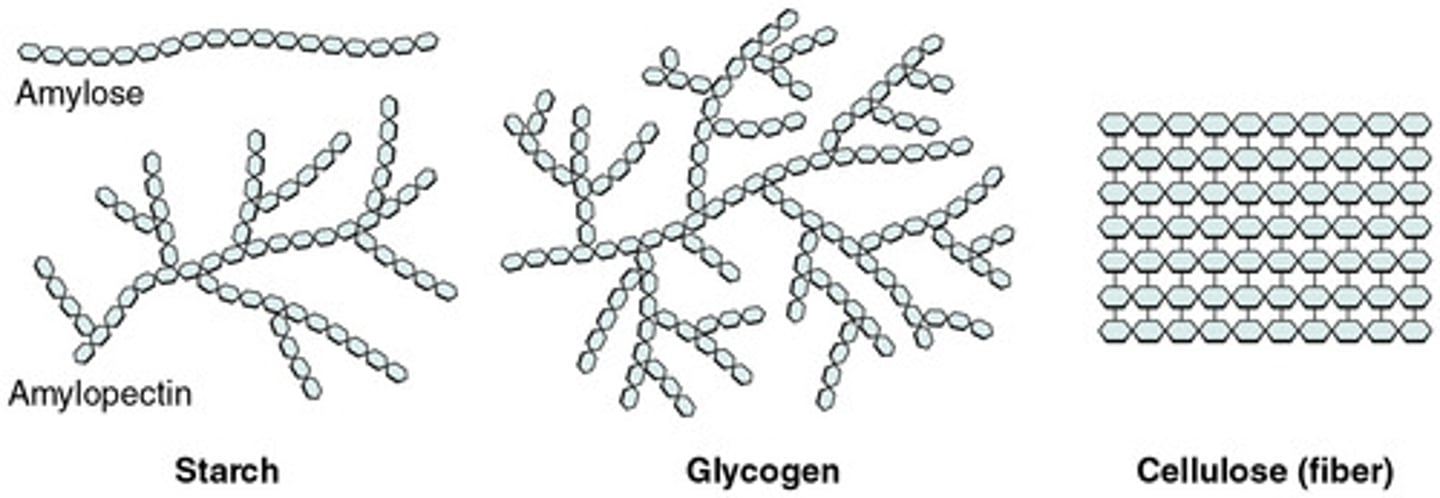
In the diagram, circle one sugar monomer.
Use your textbook or some other resource to identify and label the starch, glycogen, and cellulose molecules. Also indicate the function of each.
starch: Energy for long term, and provides carbon atoms for the synthesis of other substances in the body.
glycogen: important energy reserve, used in cellular respiration and photosynthesis
cellulose: used in plants and creates cell wall
TOPIC 1.6 Nucleic Acids
ENDURING UNDERSTANDING: DNA is the key component for all life
LEARNING OBJECTIVE
IST-1.A Describe the structural similarities and differences between DNA and RNA.
ESSENTIAL KNOWLEDGE
IST-1.A.1 DNA and RNA molecules have structural similarities and differences related to their function—
a. Both DNA and RNA have three components—sugar, a phosphate group, and a nitrogenous base—that form nucleotide units that are connected by covalent bonds to form a linear molecule with 5' and 3' ends, with the nitrogenous bases perpendicular to the sugar-phosphate backbone.
b. The basic structural differences between DNA and RNA include the following:
DNA contains deoxyribose and RNA contains ribose.
RNA contains uracil and DNA contains thymine.
DNA is usually double stranded; RNA is usually single stranded.
The two DNA strands in double-stranded DNA are antiparallel in directionality.
Use the information in the standards on the left to compare DNA and RNA. Then come up with at least three additional similarities or differences between them (consider location, size, function)
DNA: double-stranded, deoxyribose, thymine, stay inside nucleus
Same: cytosine, guanine, adenine, phosphate, nucleic acids, makes proteins, holds genetic information
RNA: single-stranded, ribose, single nucleotide base, uracil, travel outside the nucleus, mRNA, tRNA, rRNA