Organic Chem: Carboxylic acids
0.0(0)
Card Sorting
1/3
There's no tags or description
Looks like no tags are added yet.
Last updated 2:39 PM on 3/24/25
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
4 Terms
1
New cards
Properties
Has a carbonyl group: -COOH (carbonyl + hydroxyl)
Weak acids (because of presence of H+ ions)
Very polar —→ HIGHEST boiling points
2
New cards
How are carboxylics made
Formed thru controlled oxidation of aldehydes with oxidizing agents
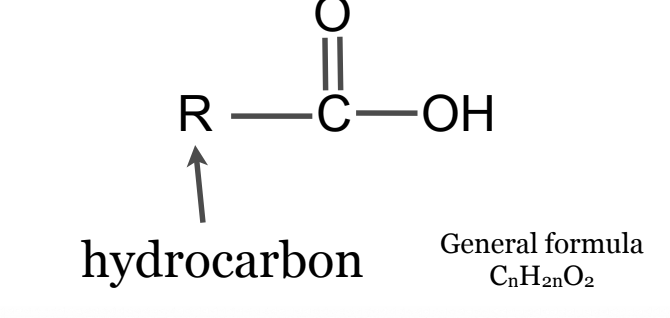
General formula: CnH2nO2
3
New cards
How to name carboxylic acids
First part: The alkane of the longest chain (INCLUDE the carbon from the carboxyl as well)
second part; Drop the -e and change to -oic acid
No need for locant because carboxyl is always at terminal carbons
4
New cards
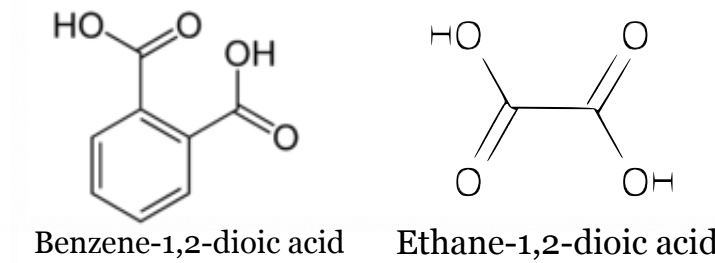
Naming Dicarboxylic acids
Full name , position of carbonyl group, -dioic acid.