Biology - OCR Enzymes
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No study sessions yet.
42 Terms
Enzymes
biological catalysts. Increase the rate of metabolic reactions.
Anabolic
A process in which large molecules are built from small molecules . - EG. for growth
Catabolic
A process in which large molecules are broken down
Metabolism
All of the chemical reactions that occur within an organism
Specificity
Each enzyme catalyses one biochemical reaction, or which there are thousands in any given cell.
Activation energy
energy supplied to start a chemical reaction. Enzymes lower this, increasing the rate of reaction
Active site
An area within the tertiary structure of the enzyme, its shape is complementary to the substrate molecule.
R-groups within this interact with the substrate forming temporary bonds.
Substrate
A specific reactant acted upon by an enzyme
Lock and key
Model of enzyme activity that explains how a particular enzyme will only fit with one particular type of substrate.
Holds substrate in such a way that atom-groups are close enough to react
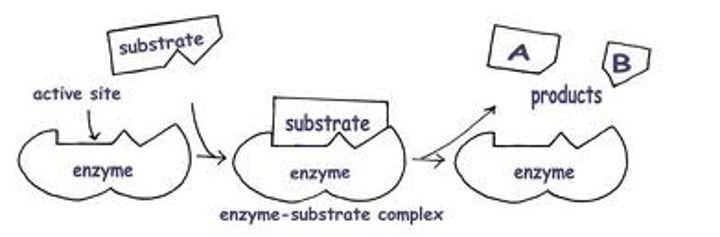
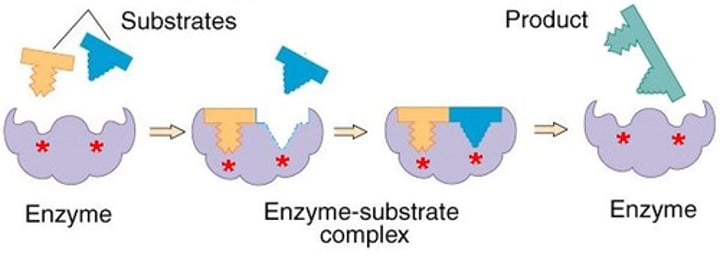
Enzyme-substrate complex
A temporary complex formed when an enzyme binds to its substrate molecule(s).
Enzyme-product complex
complex formed when a substrate is converted to a product or products while bound to the active site of an enzyme.
Induced fit
The change in shape of the active site of an enzyme so that it binds more snugly to the substrate, induced by entry of the substrate.
Initial interaction weak, but enzyme's tertiary structure changes to strengthen binding and weaken substrate - Lower AE.
Intracellular
Located inside a cell.
- Synthesis of polymers from monomers.
- Catalase breaks down H2O2 to H2O and O2
Extracellular
Located outside a cell.
- Nutrients that are polymers are too large to enter cell, so are broken down. Enzymes released to digest these.
- Single celled organisms release enzymes into immediate environment, break down molecules and then absorb.
Starch digestion
-Amylase (produced by salivary glands and pancreas) break down starch to maltose in the mouth.
- Maltose broken down into glucose by the enzyme maltase (small intestine).
Amylase and maltase
Enzymes that convert starch to glucose
Trypsin
A type of protease, (secreted from the pancreas into the small intestine). Breaks proteins into peptides.
These can be broken down further into amino acids by other proteases.
Increase temperature
= increase the kinetic energy of particles = increase the number of successful collisions = increase rate
Q10
Temperature coefficient. By how much the rate of a reaction increases with a 10C rise in temp.
- Usually taken as 2, so rate doubles with 10C increase.

Denature
At high temperatures, bonds holding the protein together will vibrate. Higher temps means bonds will strain and break. This changes the tertiary structure.
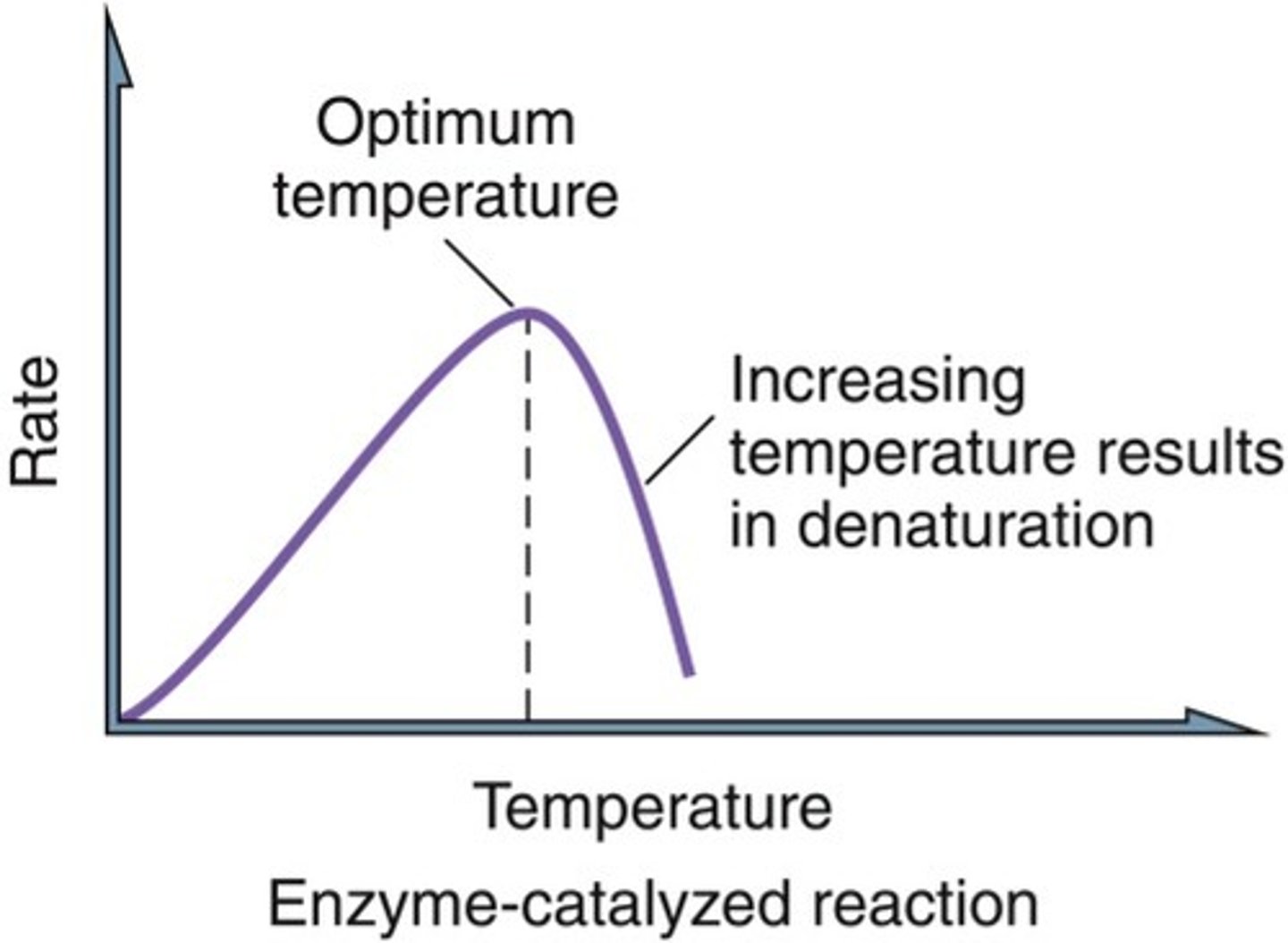
optimum temperature
The temperature at which enzyme has highest rate of activity.
- can vary, most around 40C
-Rapid decrease of rate above optimum as enzyme denatures.
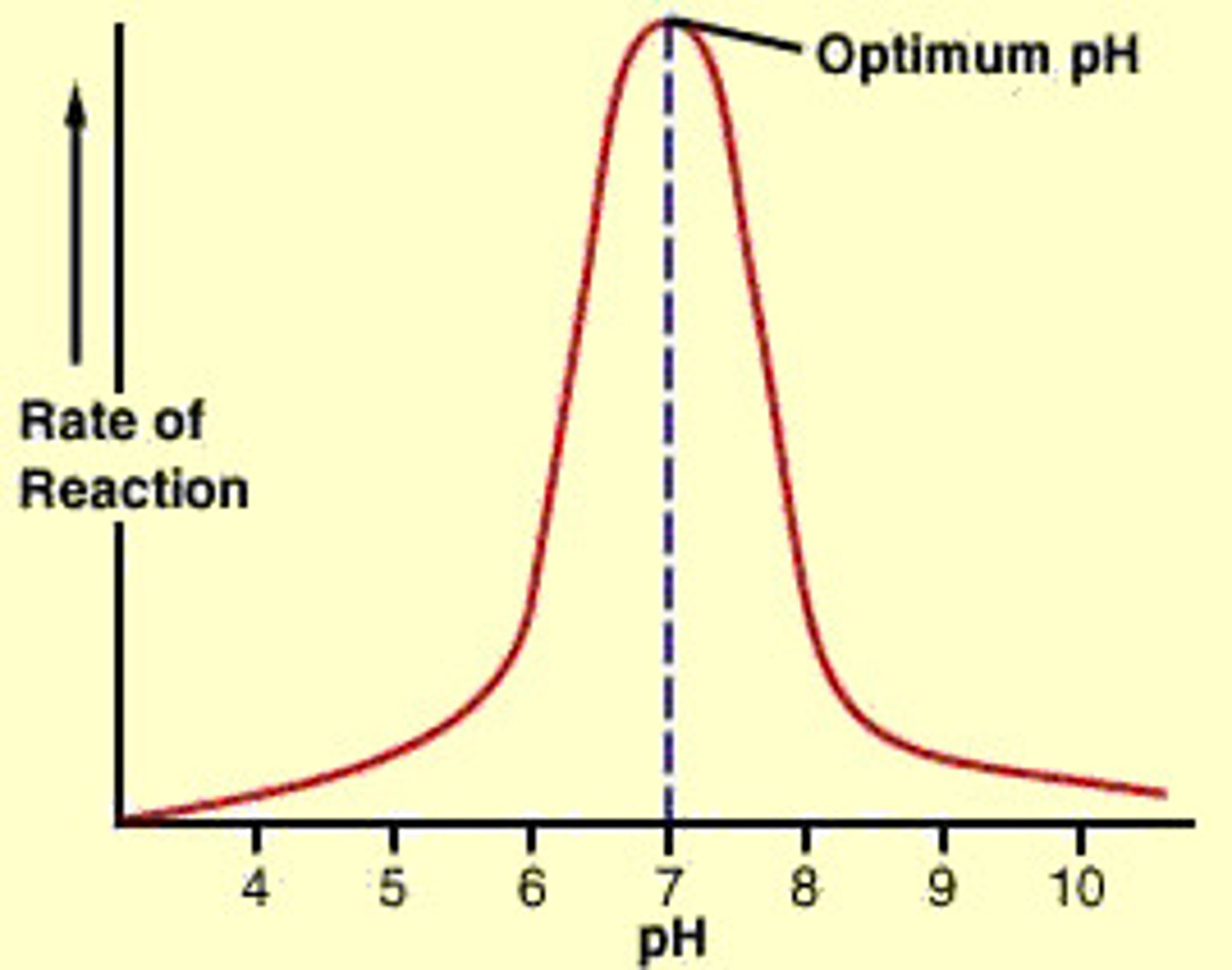
optimum pH
the pH at which an enzyme achieves maximum activity, it is the correct shape
Low pH
acid, More H+
High pH
base, fewer H+
pH
H+ ions interact with the R-groups holding the enzyme in its precise shape. This can change the degree of R-group interaction.
Renaturtion
If pH returns to the optimum, protein can resume its normal shape.
Substrate and enzyme concentration
Increased substrate= more colisions and more complexes.
Increased enzyme = more active sites.
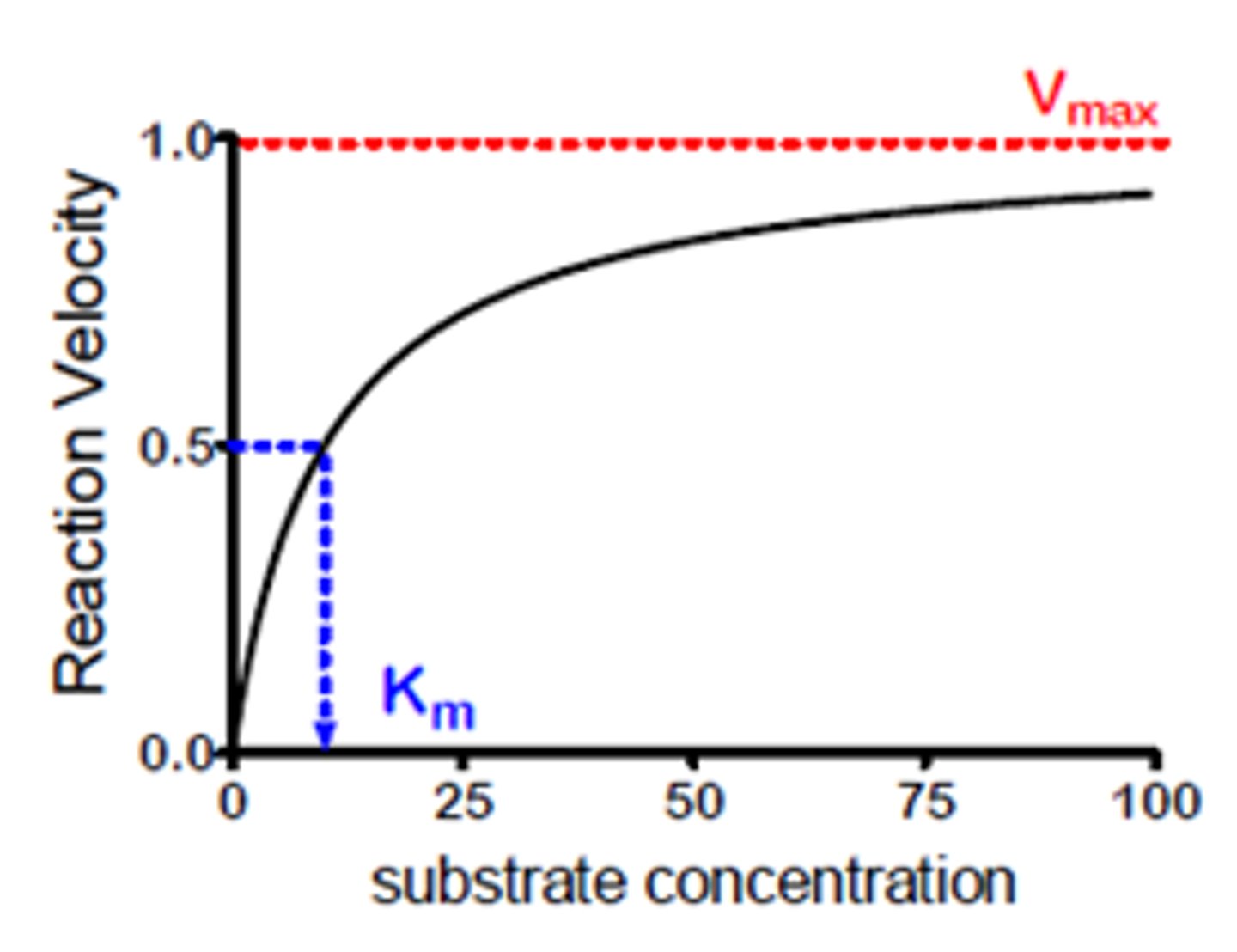
-Both increase the rate up to the Vmax, when all active sites are occupied. The other becomes the limiting factor.
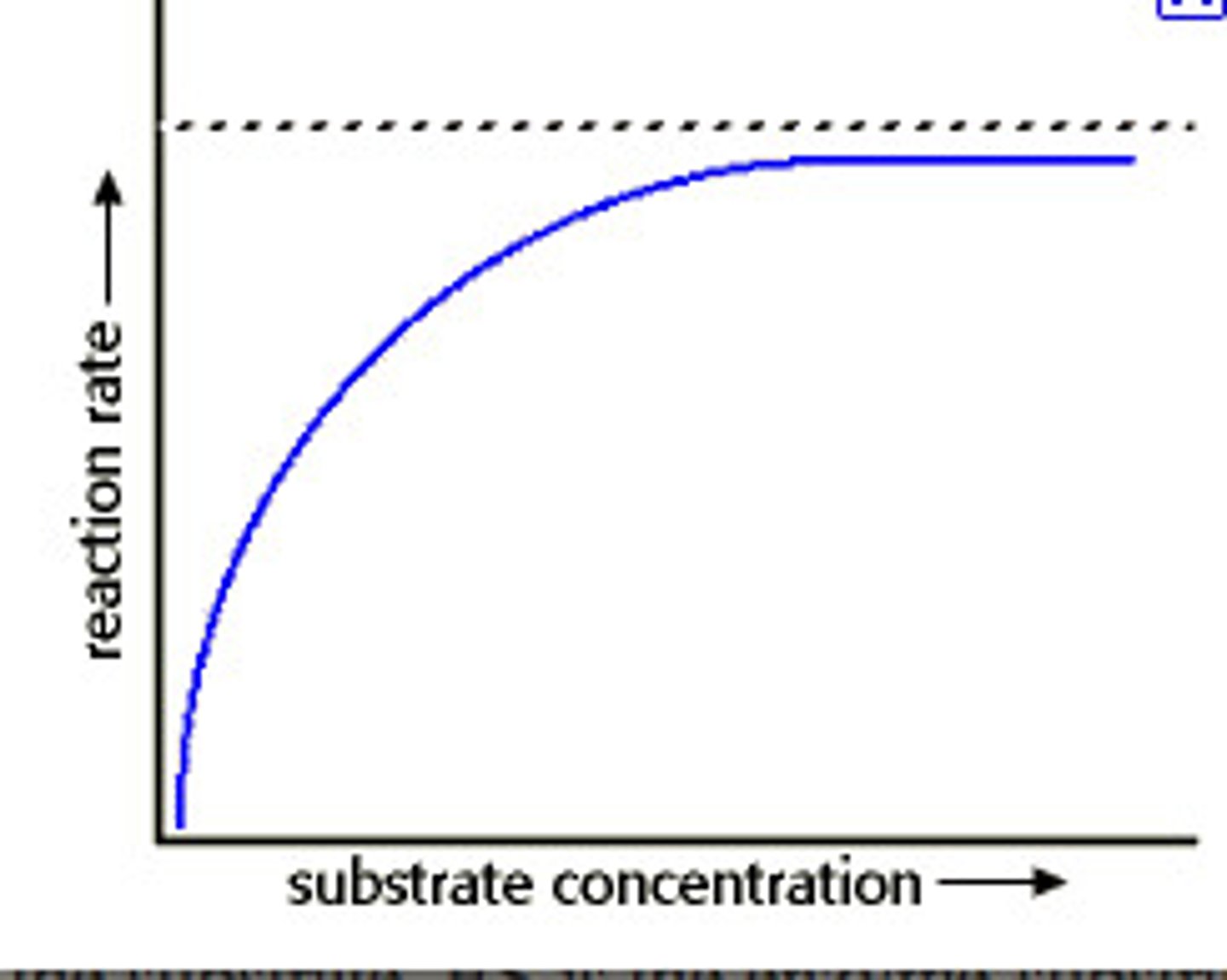
Vmax
The maximum rate possible with a given amount of enzyme.
Prosthetic group
A permanent, tightly bound cofactor.
Cofactor
Inorganic molecule.
A nonprotein molecule or ion that is required for the proper functioning of an enzyme. Obtained from diet
Coenzyme
organic molecule serving as a cofactor. Derived from vitamins
Precursor activation
Inactive precursor enzymes need to change their tertiary shape to be activated. Achieved by addition of a cofactor.
Apoenzyme
Before the cofactor was added, precursor protein.
Holoenzyme
When cofactor is added and precursor enzyme is activated.
Proenzymes (zymogens)
enzyme made in inactive form. Activated by change in conditions.
end product inhibition
When the final product in a metabolic pathway inhibits an enzyme that acts earlier on in the pathway.
-Negative feedback
- Non-competitive reversible inhibition.
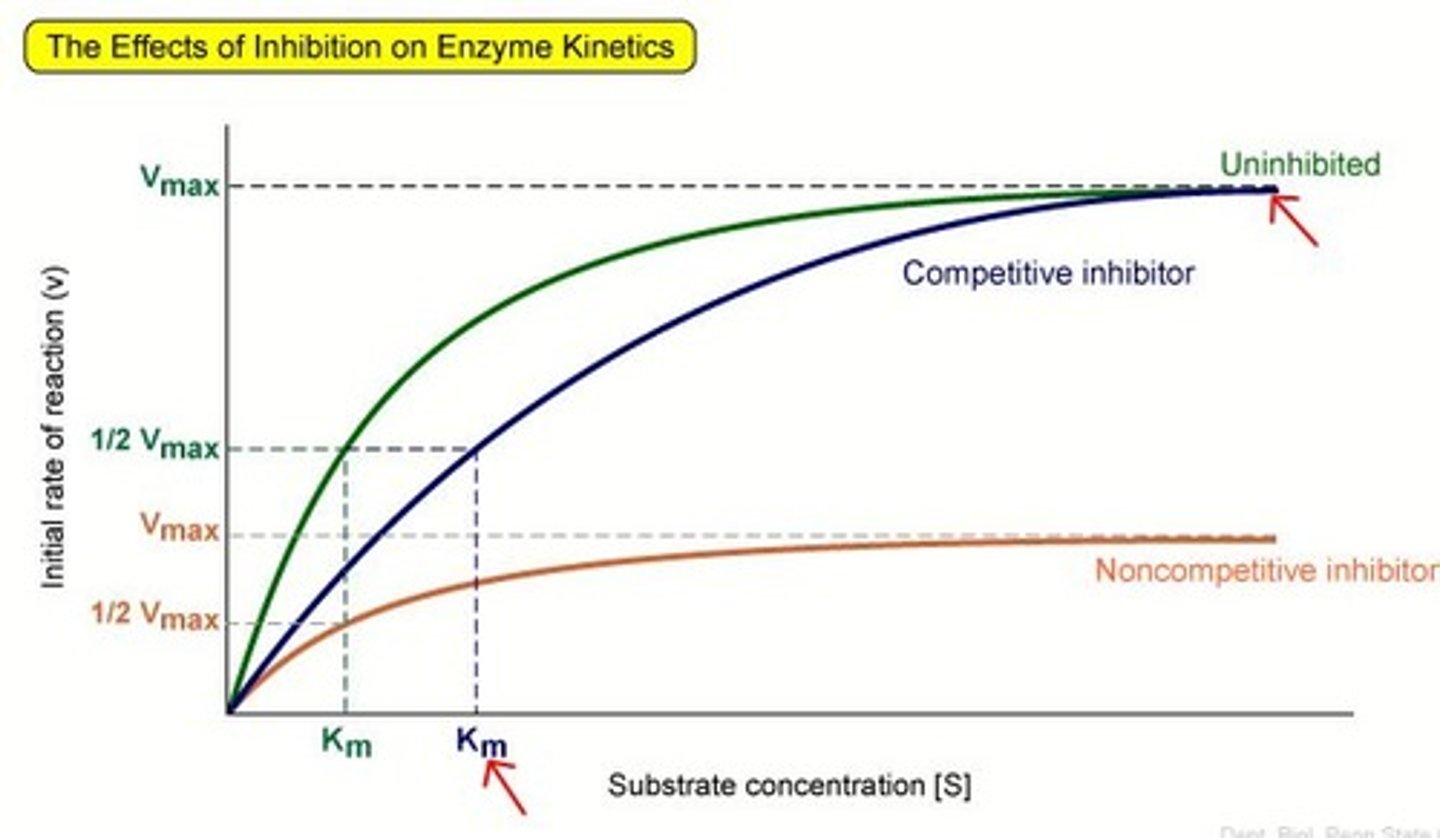
Competitive inhibition
A molecule that has a similar shape to the substrate of an enzyme fits into the active site. Blocks substrate. Prevents enzyme catalysing reaction.
- Most only bing temporarily and are reversible (Apart from Aspirin).
Statins
competitive inhibitors of enzymes used to synthesis cholesterol.
Aspirin
Irreversibly inhibits active site of COX enzymes, preventing synthesis of prostaglandin and thromboxane.
non-competitive inhibitor
Binds to the enzyme at the allosteric site. Changes tertiary structure and active site. No longer complementary.
- increasing conc of enzyme or substrate will not overcome impact of inhibitor.
Organophosphates
Irreversible non-competitive inhibitors.
Proton Pump Inhibitors
PPI, Irreversibly non-competitively inhibit enzyme responsible for secreting h+ into stomach.