BIO 101 Exam 1
1/149
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
150 Terms
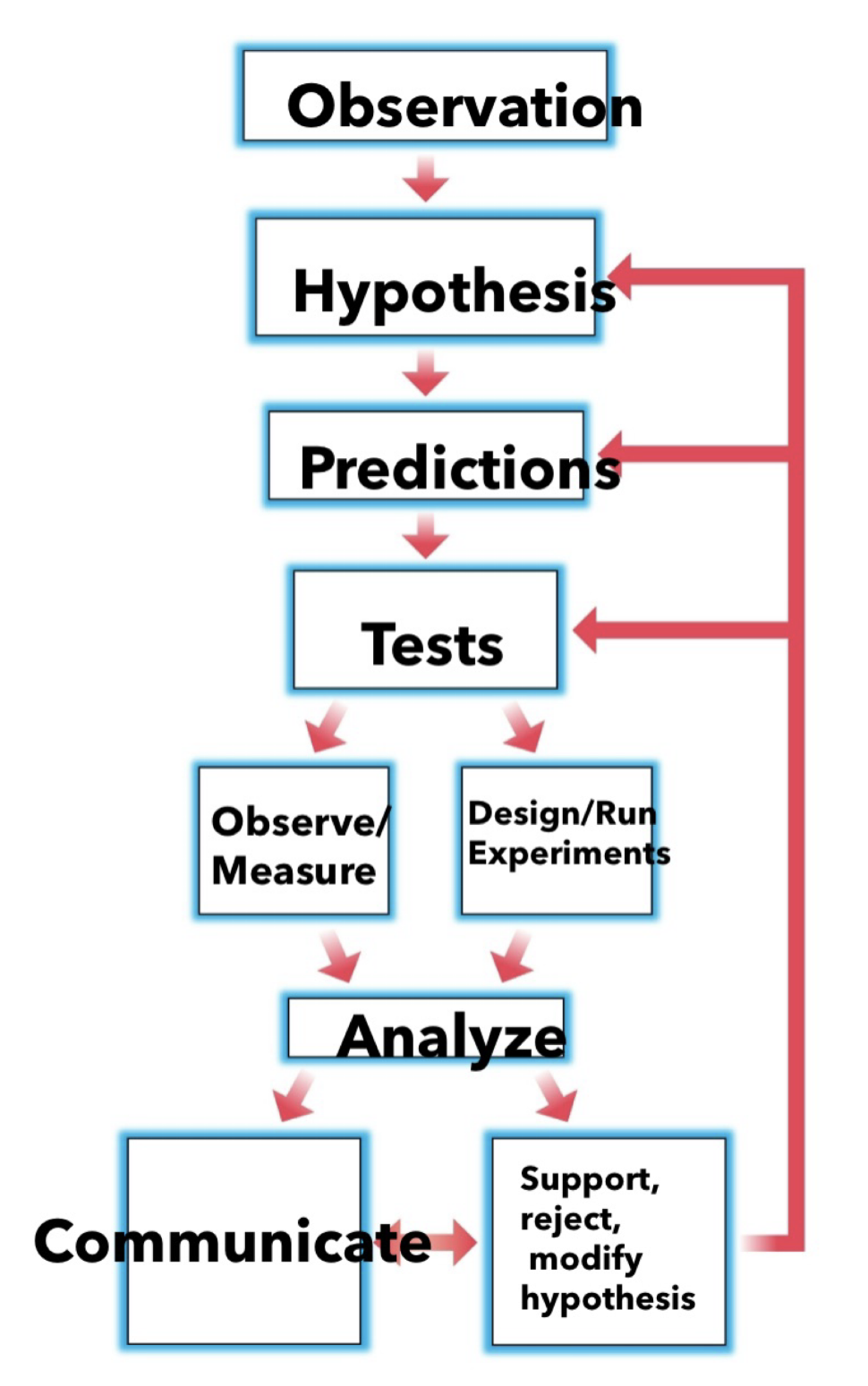
Steps of the Scientific Method
Bond type that connects amino acids
Peptide bonds (covalent)
Partial negative charge of one molecule is attracted to partial positive charge on another molecule
hydrogen bonds
organic molecules and compounds contain _______
carbon
two atoms share electrons in this bond
covalent
Bonds held together by difference in charges
ionic
Hydrogen bonds are weak electrical attractions; why do hydrogen bonds take so much energy to break?
hydrogen bonds have extreme quantity
Unique properties of water caused by hydrogen bonds
Attraction between the partially negative oxygen of one water molecule and the partially positive hydrogen of an adjacent water molecule.
Why is water a polar molecule?
Electrons are not shared equally, electrons are concentrated around oxygen atom
Why is water the “universal solvent”
Polar property makes it good at dissolving other polar molecules
Structure of proteins
chain of amino acids connected by peptide bonds
Function of proteins
regulate where cell reactions occur, support cell structure, aid in movement/mobility, transport molecules across cell membranes
Levels of protein structure
Peptide Bonds: sequence of amino acids held together by strong covalent peptide bonds
Secondary Level: hydrogen bonds between amino acids, form alpha helixes & beta sheets
Tertiary Level: R group interacts with bond types → forms 3D structures
Quaternary Level: interactions between tertiary structures
Structures of Nucleic Acids
DNA- Adenine+Thymine, Guanine+Cytosine
RNA- Adenine+Uracil, Guanine+Cytosine
Monomer of protein
amino acid
Monomer of nucleic acids
nucleotides
What are nucleotides made of?
Phosphate group, pentose sugar, nitrogenous base
Function of DNA
stores genetic information (blueprint)
Function of RNA
RNA carries out functions + directs protein synthesis
Structure of carbohydrate
long chains of monosaccharides create polysaccharides
Monomer of carbohydrates
monosaccharides: glucose, ribose, frutose
Function of carbohydrates
Energy storage (glycogen), energy source, building biomolecules
Building blocks of lipids
hydrocarbons, fatty acids, glycerol
Structure of phospholipids
made of triglycerides, glycerol, and phosphates, hydrophillic head, hydrophobic tail
Function of lipids
long term energy storage, hormone regulation, nuclear envelope
Characteristics of living things
-able to survive & reproduce autonomously
-composed of one or more cells
-sense & respond to environment
-obtain energy from the environment
-maintain homeostasis
-can evolve as a group
Structure of unsaturated vs saturated fats
saturated fats have a carbon-carbon single bond (harder to break down), unsaturated fats have a carbon-carbon double bond (easier to break down)
Structure of viruses
DNA in a membrane sheath, can’t reproduce without a host
Structures of cell membrane
-phospholipid bilayer: hydrophillic heads on the outside, hydrophobic tails on the inside
-transport proteins
-receptor proteins
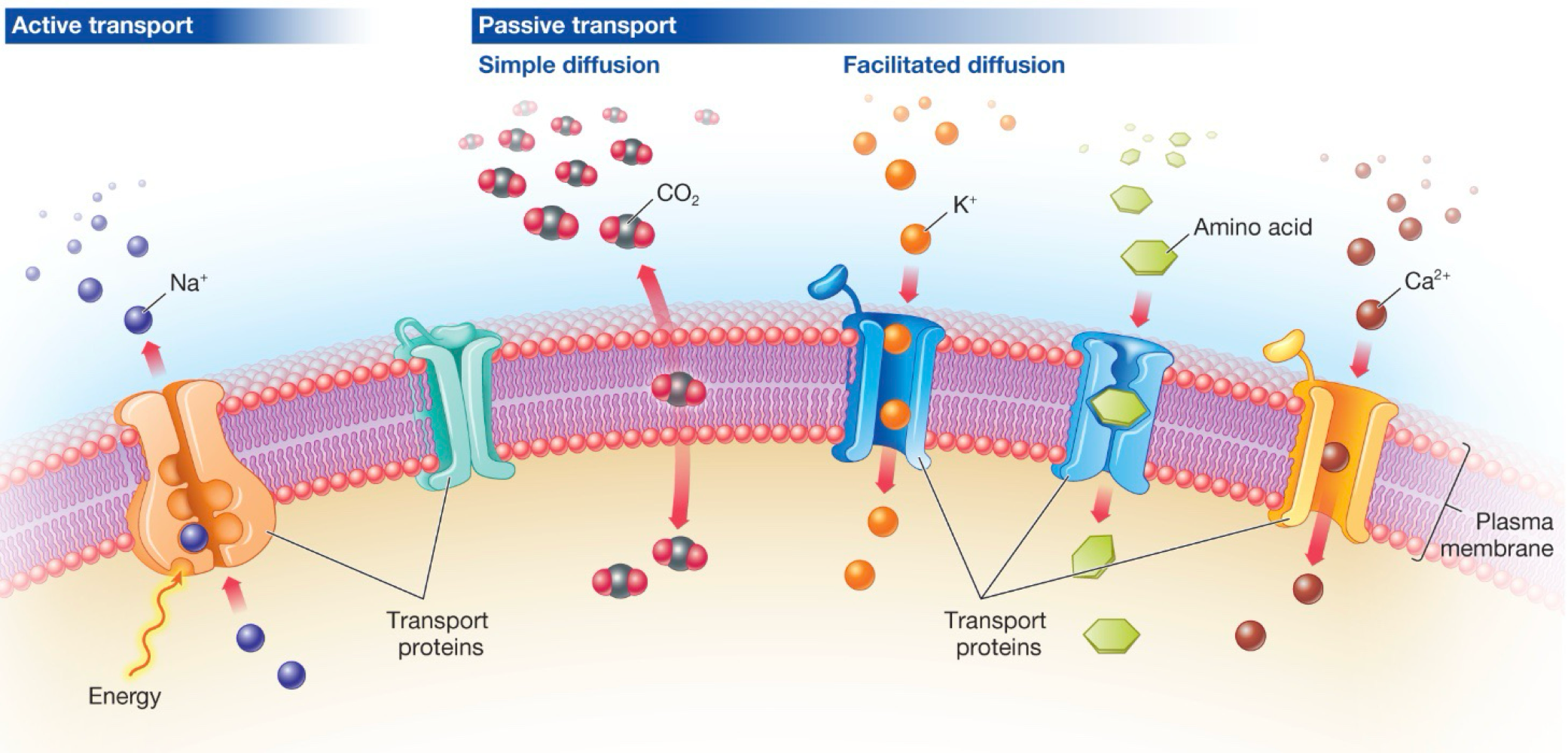
What does the selectively permeable membrane filter for?
-charge exclusion (non-polar molecules can pass through hydrophobic core)
-size exclusion
-receptor proteins
-transport proteins
Receptor protein
sites where a molecule from another cell can bind to
Function of receptor proteins
recieve molecular signals, communicate with other proteins, respond to surroundings
Transport proteins
gates, channels, and pumps that allow molecules to move in/out of the cell
Passive transport
spontaneous movement of a substance without addition of energy (high to low concentration)
Active transport
movement of a substance requiring input of energy (low to high concentration)
Simple diffusion
small, unchagred molecules ot water, oxygen, CO2 can slip through large molecules in the phosophlipid bilayer
Osmosis
simple diffusion of water
Isotonic
equal concentration of water in cell and outside solution
Hypertonic
Cell has lower concentration of solute than environment
Hypotonic
Cell has higher concentration of solute than environment
Facilitated diffusion
passive transport of hydrophilic substances
-high → low concetration
-transport proteins or vesicles
Vesicles
cell “bulge” that moves across the membrane to transport molecules in/out
Endocytosis
process by which substances are transported into cells by vesicles
Receptor mediated cytosis
receptor proteins recongnize surface characteristics of substances to be incorporated into the cell
Phagocytosis
“cellular eaating”
-where molecules are ingested via selective endocytosis
-used to ingest and destroy viral particles
Pinocytosis
“cellular drinking”
-nonselective
-cell captures surrounding fluid (takes in everything in the solution)
Exocytosis
substance to be expelled from cell is released by a vesicle
Bulk flow
transport of a large amount of molecules in/out of a cell, used in:
-receptor mediated cytosis
-phagocytosis (white blood cells)
-pinocytosis (fluid)
Metabolism
describes all chemical reactions that occur inside of cells including those that capture, store, or release energy
Metabolic passages
close chains of linked events that produce biomolecules in a cell (eg, amino acids, nucleotides)
ATP
adenosine triphosphate
-used to store energy and move it to other parts of the cell
-created by adding a phosphate group to ADP
-created in cellular respiration
anabolism
metabolic pathways that create complex molecules from smaller compounds
catabolism
metabolic pathways that release energy by breaking down complex compounds
ADP
adenosine diphosphate
-molecule leftover when phosphate at the end of ATP breaks off to release energy
Where does glycolysis occur?
the cytoplasm
Where does the Krebs Cycle occur?
mitochondrial matrix
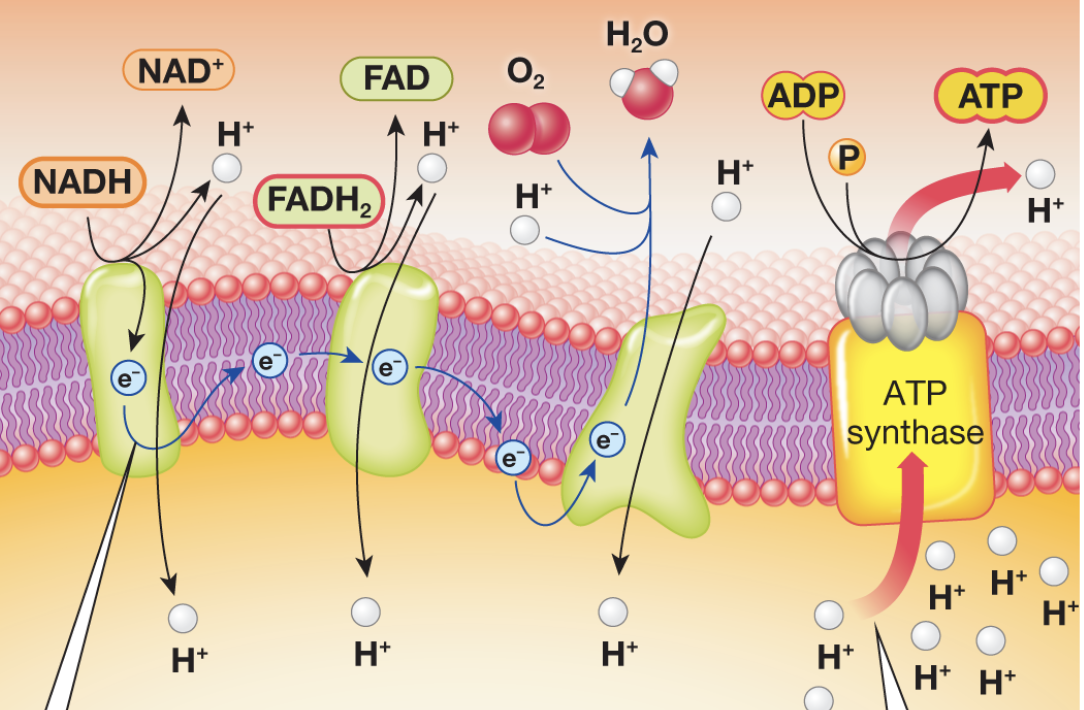
Where does Oxidative Phosphorylation occur?
the inner mitochondrial membrane
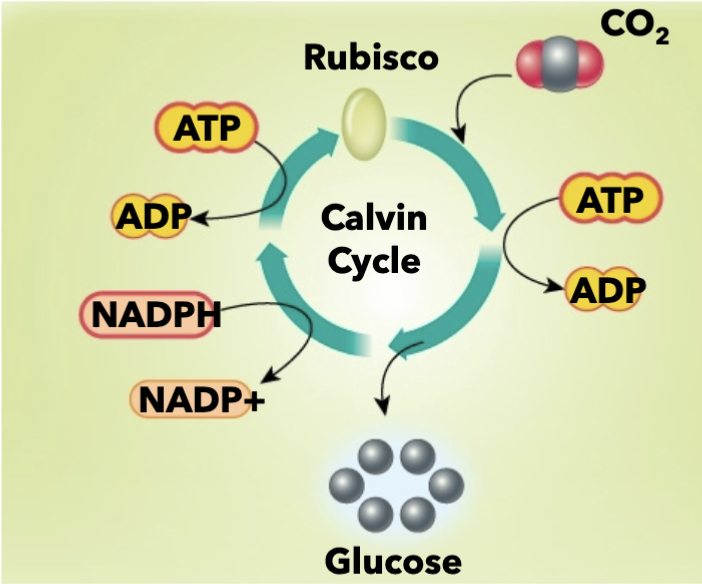
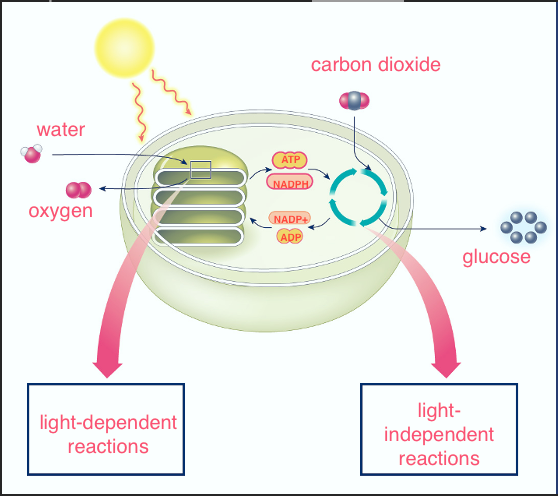
Inputs of Calvin cycle/light-independent reaction
CO2, ATP, NADPH (comes from light-dependent reaction)
Outputs of Calvin cycle/light independent reaction
NADP+, ADP, glucose
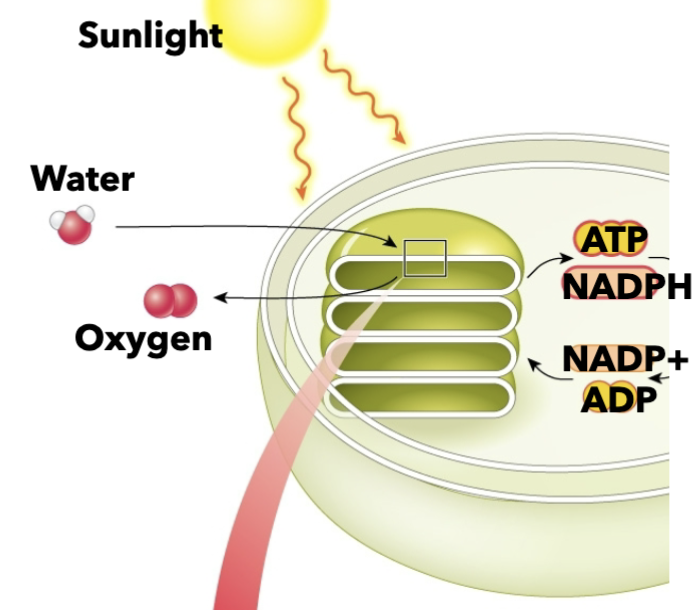
Inputs of the light-dependent reactoin
-ATP from sunlight
-water from the environment
NADP+ and ADP from the light independent reaction
Outputs of the light-dependent reaction
ATP, NADPH, oxygen
Competitive enzymes
changes shape of the enzyme/binds to active site so the substrate can’t bond
Role of electrons in photophosphorylation
provide energy for proton pump to move H+ ions
Non-competitive enzymes
binds to enzyme without filling the receptor site
Role of enzymes
lower activation energy → speeds interactions
Role of enzymes in the Thylakoid
helps create a hydrogen gradient and electron gradient
Label the photophosphorylation step in the light dependent reaction
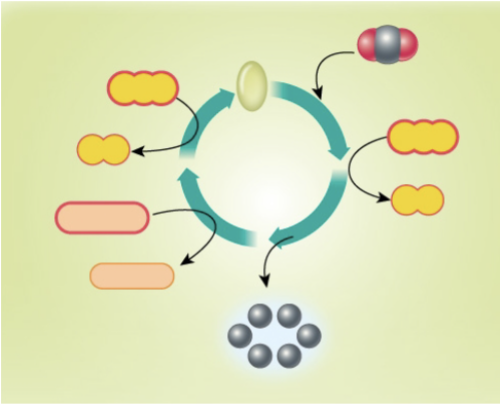
Label the Calvin Cycle
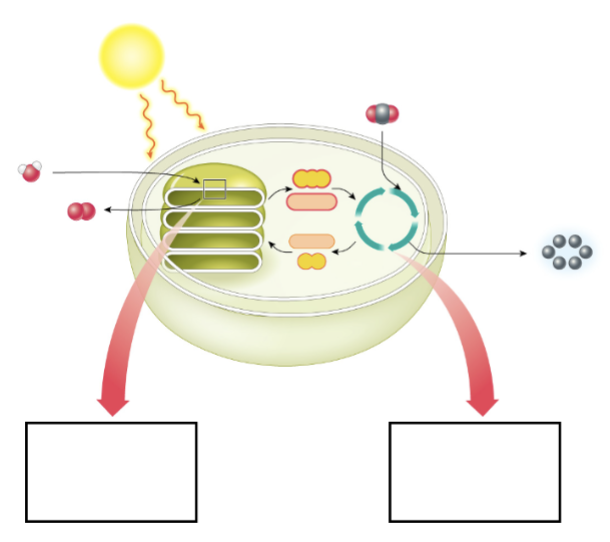
Label the photosynthesis diagram
Inputs of glycolysis
sugars (mostly glucose)
Outputs of glycolysis
-2 pyruvates
-ATP
-NADH
Outputs of the Krebs cycle
-CO2
-NADH
-FADH2
-ATP
Which step of cellular respiration happens twice per molecule?
Krebs cycle
Reaction of cellular respiration
C6H12O6 + 6O2 →6CO2 + 6H2O + 32 ATP + heat
Inputs of the Krebs cycle
-pyruvate
-NAD+
-FAD
-ADP
Inputs of oxidative phosphorylation
-NADH
-FADH2
-O2
-ADP
Why is heat an important output of oxidative phosphorylation?
maintains homeostasis through body temperature
Role of oxygen in oxidative phosphorylation
Final electron acceptor → allows you to produce metabolic water
What happens if there is no oxygen in cellular respiration?
Short term: anaerobic respiration (fermentation)
Long term: cells die
What does fermentation do?
turns pyruvate into lactic acid to regenerate NAD+
Which step(s) of cellular respiration can occur without oxygen?
only glycolysis
Terminal electron acceptor in photophosphorylation
NADP+
Terminal electron acceptor in oxidative phosphorylation
Oxygen
What provides electrons to the electron transport chain in photophosphorylation?
water
Oxidative phosphorylation
mitochondria use oxidative phosphorylation to transfer energy from food into chemical energy of ATP
Photophosphorylation
chloroplasts use photophosphorylation to transfer light energy into chemical energy of ATP
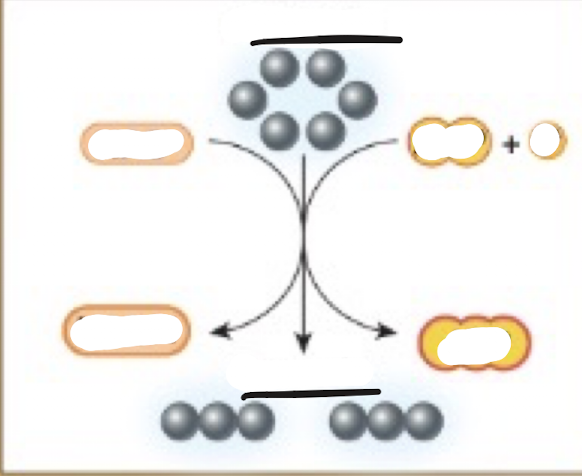
Complete the glycolysis diagram
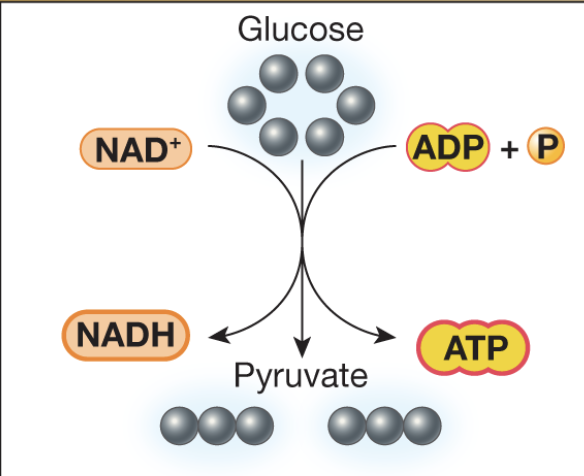
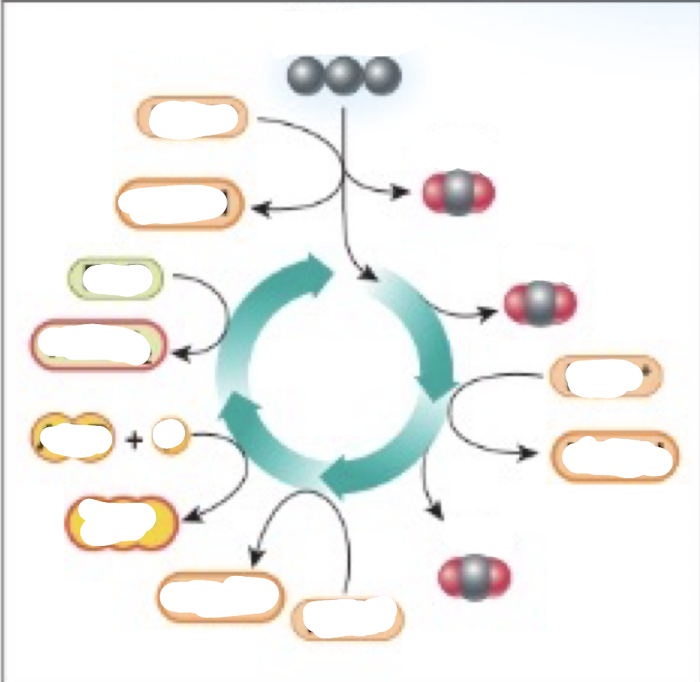
Complete the Krebs cycle diagram
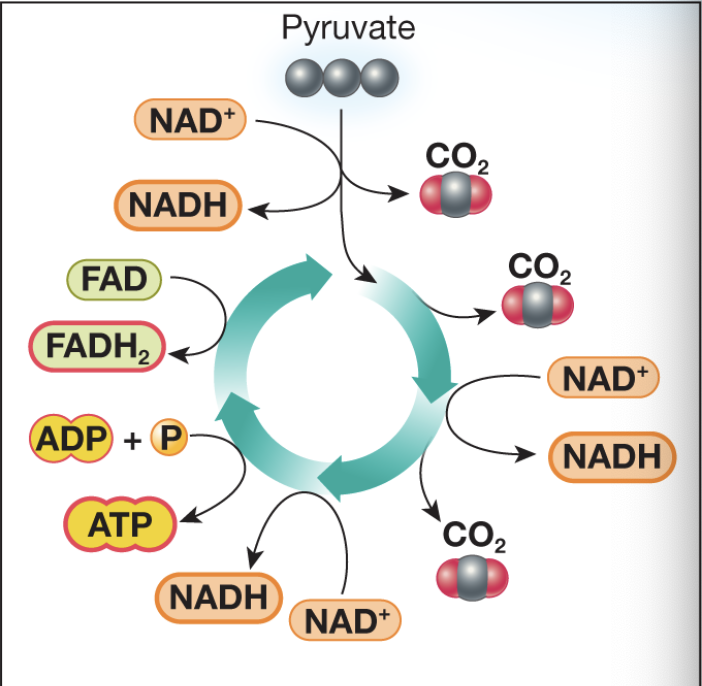
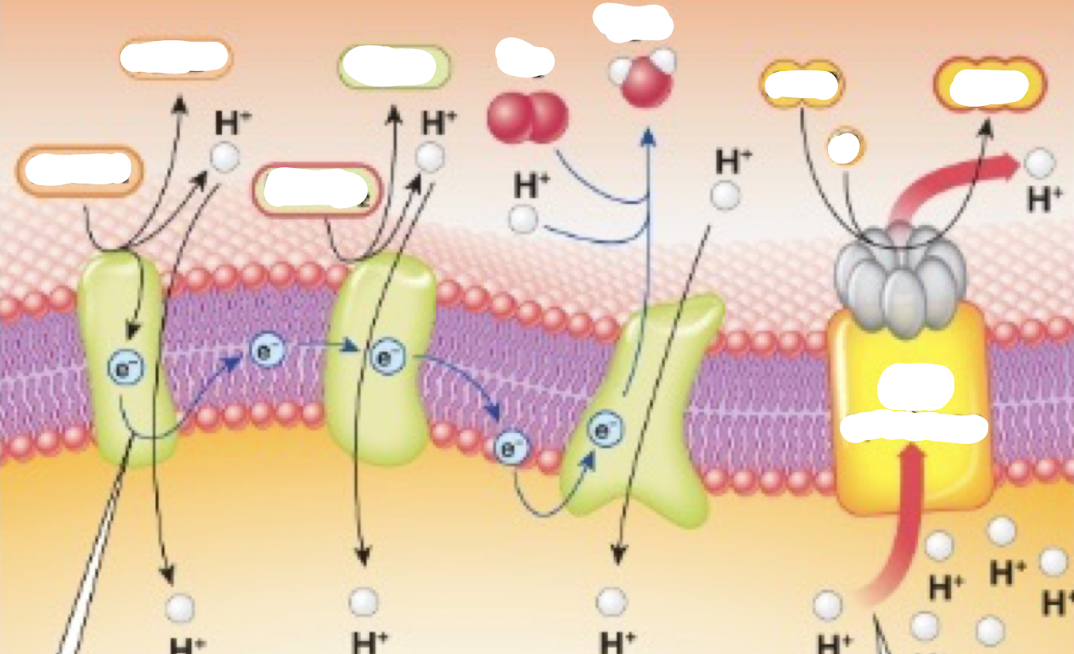
Complete the oxidative phosphorylation diagram
Induced fit
when an enzyme changes shape when molecules bind to the active site
Rubisco
enzyme that speeds up the Calvin cycle
Interphase
longest stage of the cell cycle
-cells spend most of their life in interphase (~90%)
-cells take in energy, manufacture proteins, grow larger, conduct specilaized functions
G1 phase
-first period of growth in size, protein content, and increased ATP production
-replicating organelles
-first checkpoint
G1 checkpoint
cell division pauses if cell is too small, nutrition is inadequate, and/or DNA is damaged
S phase
synthesis- DNA is replicaed
G2 phase
-last stage of interphase
-second period of growth (size/protein content)
-organelles replicate
-checkpoint before cell division
G2 checkpoint
cell division pauses if cell is too small, nutrition is inadequate, DNA is damaged, and/or if chromosome replication is incomplete
What happens if cells wrongfully pass a checkpoint?
damaged cells can replicate “runaway cell division”
-cancer
-organ dysfunction
-organs can form incorrectly
Asexual reproduction
the process by which parent cells generate clones (genetically identical)
Binary fission
the process by which a prokaryotic cell reproduces, essentially just splitting in half down the middle