Work and Energy
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
29 Terms
Energy
Property of a system that allows it to do work or cause change.
All forms of energy (kinetic, potential, thermal, etc.) are measured in joules (J) in the SI system.
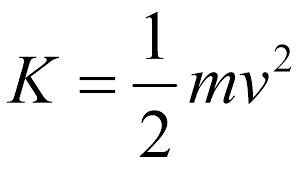
Kinetic Energy
Energy associated with the motion of an object.
Depends on mass and speed squared (scalar quantity; does not depend on direction).
Formula:
KE = 1/2mv2
Potential Energy
Energy stored in a system that can be converted into work or motion.
Types include:
Gravitational: U = mgh
Elastic: U = ½ kx2
Electrical
Chemical
Gravitational Potential Energy (GPE)
Energy an object has due to its position in a gravitational field.
Depends on:
Mass (m)
Height (h) above a reference point (datum)
Gravity (g)
Formula: U = mgh
Elastic Potential Energy
Energy stored in a stretched or compressed spring (or elastic object).
Depends on:
Spring constant (k) — stiffness of the spring
Displacement (x) — how much the spring is stretched or compressed
Formula: U = ½ kx2
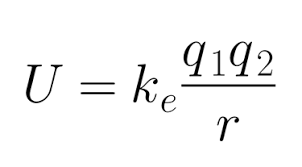
Electrical Potential Energy
Energy stored due to the position of charged particles relative to each other.
Depends on:
Magnitude of charges (q1,q2)
Distance between them (r)
Formula (Coulomb’s law form):
U = (ke) (q1q2/r)
Chemical Potential Energy
Energy stored in the chemical bonds of molecules or compounds.
Released or absorbed during chemical reactions.
Mechanical Energy:
Total energy of a system due to motion and position.
Formula:
Emechanical = KE + PE
KE = kinetic energy
PE = potential energy (gravitational, elastic, etc.)
Conservative Forces
Path independent: Work done depends only on initial and final positions, not the path taken.
Do not dissipate mechanical energy — total mechanical energy is conserved.
Conservation of Mechanical Energy
If only conservative forces act, the total mechanical energy (KE + PE) is constant.
Examples of conservative forces:
Gravity
Electrostatic forces
Elastic (spring) forces — nearly conservative in real situations.
Nonconservative Forces
Path dependent: Work done depends on the path taken.
Dissipate mechanical energy — convert it into other forms (e.g., heat, sound).
Effects of Nonconservative Forces
Total energy is still conserved, but some mechanical energy is transformed into thermal, chemical, or other forms.
Examples of nonconservative forces:
Friction
Air resistance
Viscous drag
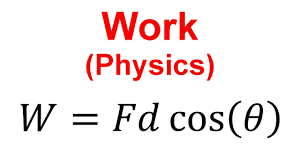
Work
Definition: A process by which energy is transferred from one system to another.
Work involves a force applied over a distance in the direction of the force.
Formula:
W = F⃗⋅d⃗ = Fd cosθ
Work in Thermodynamics
Definition: Energy transferred when a system expands or compresses against external pressure.
Formula:
W = Area under a Pressure–Volume (P–V) curve
Sign convention:
W>0 → work done by the system
W<0 → work done on the system
Power
Definition: The rate at which work is done or energy is transferred.
Formula:
P = W/t
W = work
t = time
SI unit: watt (W), where 1 W = 1 J/s

Work–Energy Theorem
Definition: When net work is done on or by a system, the system’s kinetic energy changes by the same amount:
Wnet = ΔKE
More generally, work can also be transferred into other forms of energy (potential, thermal, etc.).
Mechanical Advantage
Factor by which a simple machine multiplies the input force.
Formula:
Mechanical Advantage = Fout/Fin
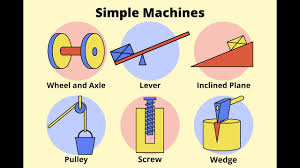
Six Simple Machines
Types:
Inclined plane
Wedge
Wheel and axle
Lever
Pulley
Screw
Purpose: Provide mechanical advantage, making work easier by multiplying force or changing its direction.
Mechanical Advantage and Work
Effect: Makes it easier to accomplish work by reducing the input force.
Trade-off: The distance over which the input force is applied increases by the same factor.
Assumption: 100% efficiency (no energy lost).
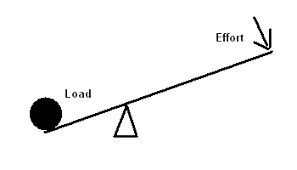
Load vs Effort in Simple Machines
Load: The output force of a machine. Acts over load distance → determines work output.
Effort: The input force applied to the machine. Acts over effort distance → determines work input.

Efficiency of a Machine
Definition: Ratio of work output to work input, accounting for nonconservative forces (like friction).
Formula:
Efficiency = Wout/Win ×100
Zeroth Law of Thermodynamics:
If two systems are each in thermal equilibrium with a third system, then they are in thermal equilibrium with each other.
If system A is in thermal equilibrium with system B, and system B is in thermal equilibrium with system C, then A and C are in equilibrium.
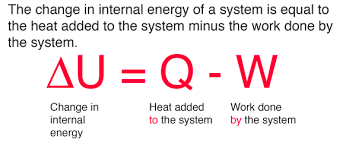
First Law of Thermodynamics:
Energy cannot be created or destroyed, only transformed.
ΔU = Q-W
ΔU: change in internal energy
Q: heat added to the system
W: work done by the system
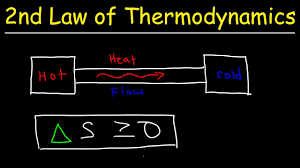
Second Law of Thermodynamics:
In any real (irreversible) process, the entropy of the universe increases.
ΔS universe ≥ 0
Heat flows spontaneously from hot to cold, not the other way around.
Third Law of Thermodynamics:
The entropy of a perfectly ordered crystalline substance approaches zero as temperature approaches absolute zero.
S→0 as T→0K
What is the condition for isothermal expansion of an ideal gas?
Temperature stays constant
(ΔT=0 ⇒ ΔU=0)
What is the condition for adiabatic expansion?
No heat exchange (Q=0).
What is the condition for an isobaric process?
Pressure is constant (P = constant).
What is the condition for an isochoric process?
Volume is constant (ΔV=0).