Ch 20 - Carboxylic Acids and their Derivatives
1/106
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
107 Terms
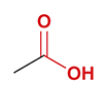
acetic acid
CH3COOH
methyl group attached to carboxylic acid group
a carboxylic acid responsible for the smell of vinegar
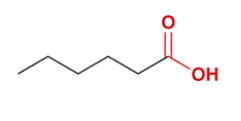
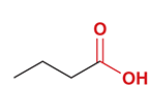
butanoic acid
CH3CH2CH2COOH
propyl group attached to carboxylic acid group (the C in the carboxylic acid group counts as the 4th C)
a carboxylic acid responsible for the rancid odor of sour butter

hexanoic acid
CH3CH2CH2CH2CH2COOH
pentyl group attached to carboxylic group (the C of the COOH makes the 6th C)
a carboxylic acid responsible for the odor of dirty socks
lactic acid
CH3CHOHCOOH
a carboxylic acid responsible for the taste of sour milk
nomenclature of monocarboxylic acids
name parent as the longest chain that includes the C of the carboxylic acid group (that C is assigned locant number 1)
suffix “-oic acid”
if the carboxylic acid is connected to a ring, the compound is named as an alkane carboxylic acid (e.g. cyclohexanecarboxylic acid)
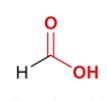
formic acid
common name for methanoic acid
acetic acid
common name for ethanoic acid
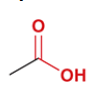
propionic acid
common name for propanoic acid
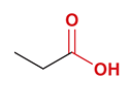
butyric acid
common name for butanoic acid
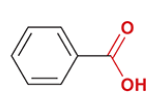
benzoic acid
common name for benzenecarboxylic acid
nomenclature for diacids
use suffix “dioic acid”
same rules apply as usual for choosing parent, assigning locants, and naming substituents
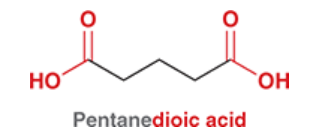
oxalic acid
common name for ethanedioic acid
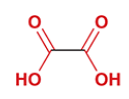
malonic acid
common name for propanedioic acid
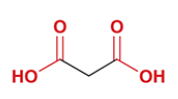
succinic acid
common name for butanedioic acid
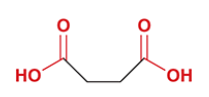
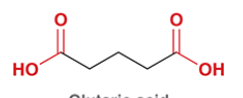
glutaric acid
common name for pentanedioic acid
structure and properties of carboxylic acids
sp2 hybridized C → trigonal planar geometry
can form 2 H-bonding interactions (between carbonyl O of one and H of the other); molecules can associate with each other in pairs
relatively high boiling point
mildly acidic; treatment with a strong bae (e.g. NaOh) yields a carboxylate salt
pKa usually between 4 and 5
very weak acids when compared to inorganic acids, but relatively acidic compared to most organic compounds
physiological pH
the pH of blood
~7.3
at this pH, carboxylate ion to carboxylic acid ratio is ~1000:1 (figured because of rearrangement of Henderson-Hasselbalch equation)
Henderson-Hasselbalch equation
pH = pKa + log([conjugate base]/[acid])
effect of substituents on acidity
pKa of acetic acid = 4.8
if 1 Cl added, pKa = 2.9 (at alpha position)
if 2 Cl added, pKa = 1.3 (at alpha position)
if 3 Cl added, 0.9 (at alpha position)
^because of inductive effects of Cl → stabilize the conjugate base
if 1 Cl added at beta position, pKa = 4.1
if 1 Cl added at gamma position, pKa = 4.5
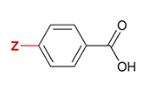
pKa of benzoic acid with para-substituted NO2
3.4
NO2 is substituted at the Z
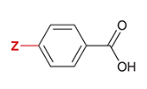
pKa of benzoic acid with para-substituted CHO
3.8
CHO is substituted at the Z
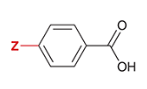
pKa of benzoic acid with para-substituted Cl
4.0
Cl is substituted at the Z
pKa of benzoic acid with para-substituted H
4.2
H is substituted at the Z
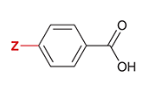
pKa of benzoic acid with para-substituted CH3
4.3
CH3 is substituted at the Z
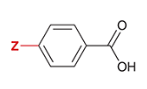
pKa of benzoic acid with para-substituted OH
4.5
OH is substituted at the Z
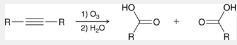
oxidative cleavage of alkynes
a way to prepare carboxylic acids
alkyne gets treated with 1) O3 and 2) H2O
oxidation of primary alcohols
a way to prepare a carboxylic acid
primary alcohol is treated with Na2Cr2O7, H2SO4, H2O
yields 1 carboxylic acid
various strong oxidizing agents can be used
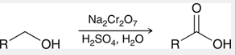
oxidation of alkylbenzenes
an alkylbenzene is treated with Na2Cr2O7, H2SO4, H2O
alkyl group is completely oxidized, as long as the benzylic position is not quaternary
yields 1 carboxylic acid
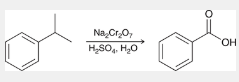
hydrolysis of nitriles
a way to prepare a carboxylic acid
a nitrile is treated with aqueous acid
produces 1 carboxylic acid
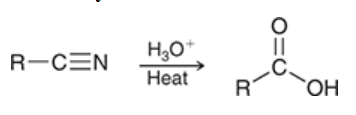
carboxylation of Grignard reagents
a way to prepare a carboxylic acid
Grignard reagent is treated with carbon dioxide and acid
yields 1 carboxylic acid
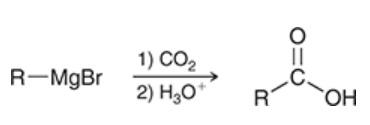
process of carboxylation of Grignard reagents
Grignard reagent (Nuc) attacks electrophilic center of CO2 → generates carboxylate ion
carboxylate gets protonated to yield carboxylic acid
2 steps occur separately
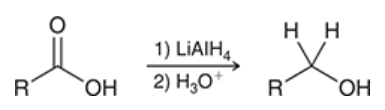
reduction of carboxylic acid with LAH
carboxylic acid gets treated with 1) LiAlH4 and 2) H3O+
produces alcohol
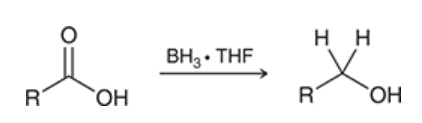
reduction of carboxylic acid with borane
carboxylic acid is treated with BH3 and THF
produces alcohol
reduction with LAH vs BH3
reduction with borane is often preferred over with LAH
borane reacts selectively with carboxylic acid group in the presence of another carbonyl group; LAH is so strong it reduces both carbonyls
carboxylic acid derivatives
compounds that are similar in structure to a carboxylic acid (RCOOH) but the OH has been replaced with a different group, Z
Z is a heteroatom, e.g. Cl, O, N, etc.
nitriles (R-CN) are also derivatives because they have the same oxidation state as carboxylic acids
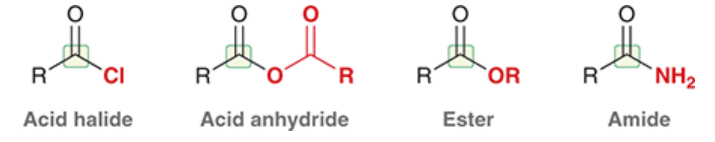
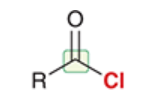
acid halide
a carboxylic acid derivative in which the OH of RCOOH has been replaced by a halogen (e.g. Cl)
full structure is RCOX (X = halogen)
highly reactive; not very common in nature
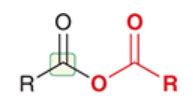
acid anhydride
a carboxylic acid derivative in which the OH of RCOOH has been replaced by OCOR
full structure is RCOOCOR
highly reactive; not very common in nature
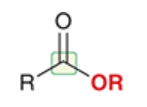
ester
a carboxylic acid derivative in which the OH of RCOOH has been replaced by OR
full structure is RCOOR
more stable than acid halides and acid anhydrides
abundant in nature; often have pleasant odors and contribute to the aromas of fruits and flowers
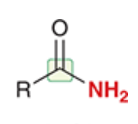
amide
a carboxylic acid derivative in which the OH of RCOOH has been replaced by NH2
full structure is RCONH2
abundant in living organisms
e.g. repeating linkages make up proteins
nitrile
a carboxylic acid derivative with the same oxidation state as a carboxylic acid; C has three bonds to a heteroatom (N)
conversion of nitrile to carboxylic acid, or vice versa, is neither reduction or oxidation
structure is RCN
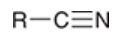
nomenclature of acid halides
replace suffix “ic acid” with “yl halide”
e.g. acetic acid → acetyl bromide
e.g. benzoic acid → benzoyl chloride
e.g. cyclohexanecarboxylic acid → cyclohexanecarbonyl chloride
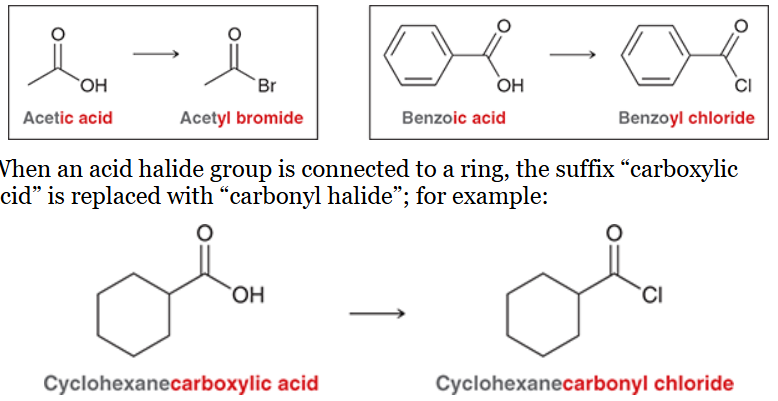
nomenclature of acid anhydrides
replace suffix “acid” with “anhydride”
unsymmetrical anhydrides are prepared from two different carboxylic acids; named by indicating both acids alphabetically followed by suffix “anhydride”
e.g. acetic acid → acetic anhydride
e.g. succinic acid → succinic anhydride
e.g. acetic benzoic anhydride
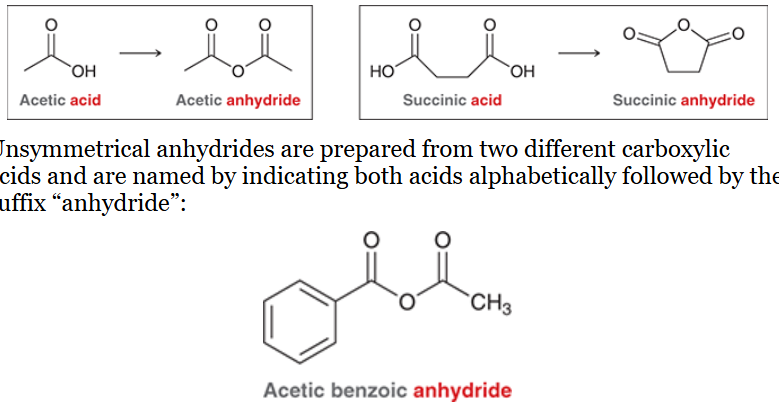
nomenclature of esters
indicate the alkyl group attached to the oxygen first
follow with carboxylic acid, and replace suffix “ic acid” with “ate”
see picture for examples
nomenclature of amides
replace suffix “ic acid” or “oic acid” with “amide”
e.g. acetic acid → acetamide
e.g. benzoic acid → benzamide
if connected to a ring, suffix “carboxylic acid” is replaced with “carboxamide”
if N atom bears alkyl groups, the groups are connected at the beginning of the name and the letter “N'“ is used as a locant instead of a number
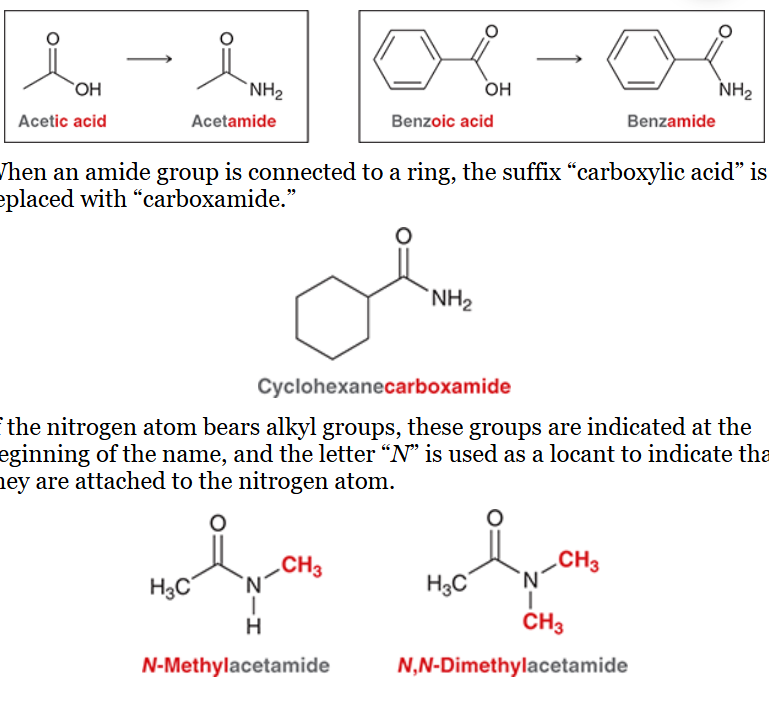
nomenclature of nitriles
replace suffix “ic acid” or “oic acid” with “onitrile”
e.g. acetic acid → acetonitrile
e.g. benzoic acid → benzonitrile
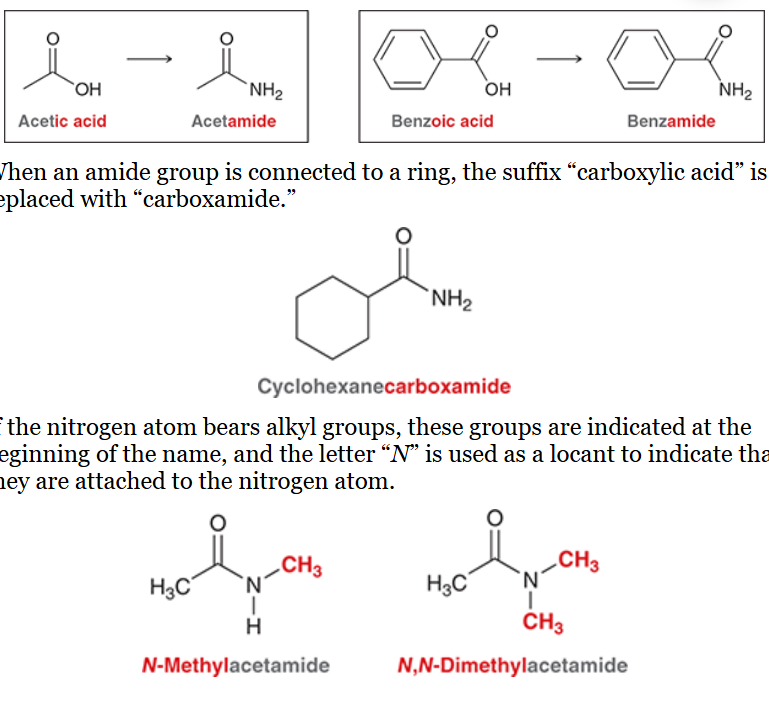
relative order of reactivity of carboxylic acid derivatives
listed from least to most reactive
amide, ester, acid anhydride, acid halide
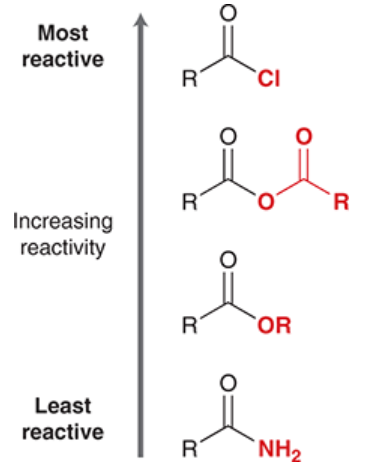
reactivity of carboxylic acid derivatives (in general)
similar to reactivity of aldehydes and ketones
carbonyl group is electrophilic; subject to nucleophilic attack
heteroatom can function as a leaving group (unlike ketones and aldehydes)
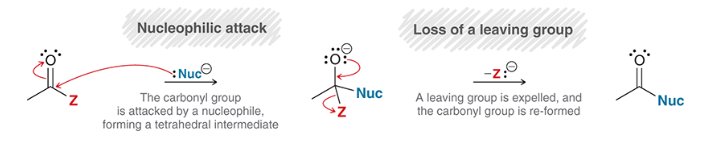
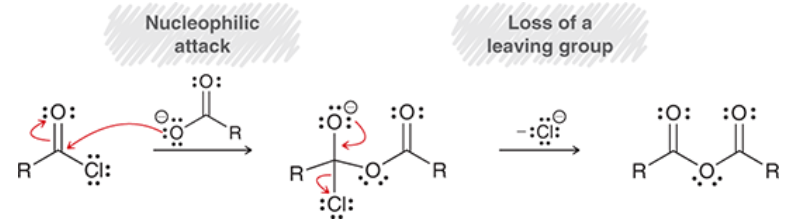
nucleophilic acyl substitution
a reaction in which a nucleophile attacks a carboxylic derivative
nucleophile replaces the Z leaving group
note: most reactions in this chapter are variants of this type of reaction
mechanism for nucleophilic acyl substitution
nucleophilic attack: forms tetrahedral intermediate
loss of leaving group and carbonyl reformation
note: even poor leaving groups (e.g. RO-) can be expelled
NOT Sn2 (not concerted) because Sn2 does not happen readily at sp2-hybridized centers
proton transfer may happen either 1) before nucleophilic attack, 2) before loss of leaving group, or 3) at the end of the mechanism. May happen at all three steps, may only happen at one. Depends on conditions
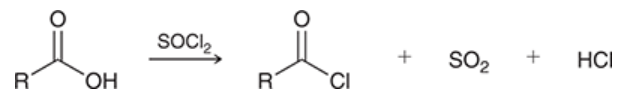
preparation of acid chlorides
carboxylic acid is treated with thionyl chloride (SOCl2)
yields an acid chloride + SO2 + HCl
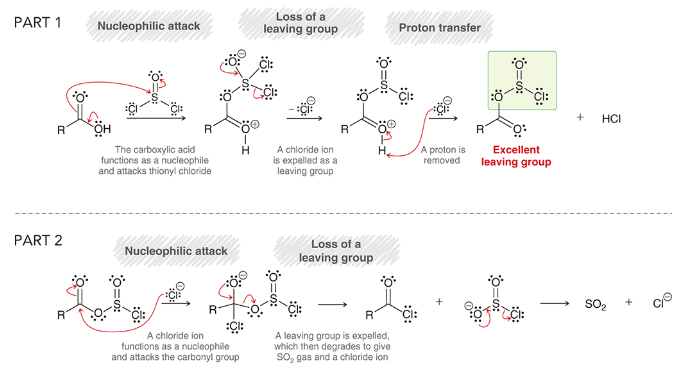
mechanism for the preparation of acid chloride via thionyl chloride
part 1:
nucleophilic attack: carboxylic acid functions as Nuc
loss of leaving group: Cl- is expelled
proton transfer: deprotonation
results in a molecule with OSOCl, an excellent leaving group
part 2:
nucleophilic attack: Cl- is Nuc, attacks carbonyl
loss of leaving group
rearrangement of product to make SO2 and Cl-
hydrolysis of acid chloride
acid chloride is treated with water
yields carboxylic acid
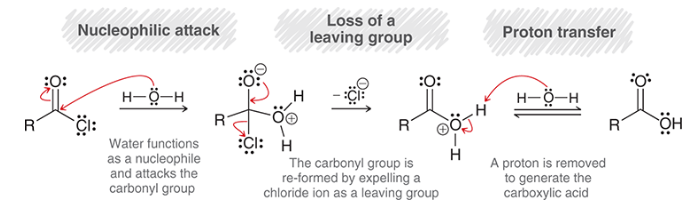
mechanism for hydrolysis of an acid chloride
nucleophilic attack: water is Nuc
loss of leaving group: carbonyl reforms, Cl- is expelled
proton transfer: deprotonation
pyridine may be used to remove HCl as it is produced, because HCl can often react with other functional groups in the compound (unwanted)
alcoholysis of acid chlorides
acid chloride is treated with alcohol
converts acid chloride into ester
mechanism is directly analogous to mechanism for hydrolysis of an acid chloride
pyridine is used, again
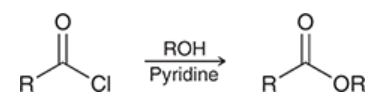
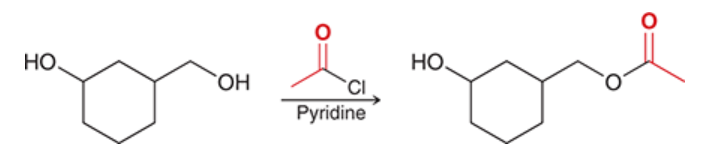
acylation of an alcohol
alcohol is treated with acyl chloride and pyridine
yields ester
selective for primary alcohols in the presence of secondary alcohols
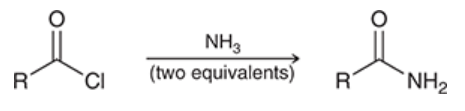
aminolysis of acid chlorides
acid chloride is treated with 2 equivalents of ammonia
produces an amide
pyridine is not used because ammonia is a strong enough base to neutralize HCl as it is produced (this is why a second equivalent is needed)
analogous mechanism to hydrolysis; nucleophilic attack, loss of leaving group, proton transfer
aminolysis of acid chlorides with amines
primary or secondary amines can be used instead of ammonia
N-substituted amides are produced
same process as regular aminolysis
reduction of acid chlorides
acid chloride is treated with LiAlH4
reduced to give alcohol
2 steps are required (acidic workup is needed)
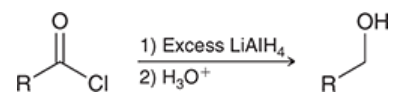
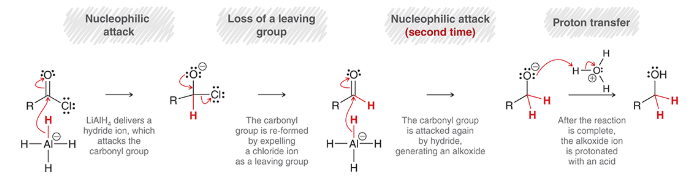
mechanism for reduction of an acid chloride with LAH
nucleophilic attack: LiAlH4 delivers a hydride ion nucleophile
loss of leaving group: Cl- is expelled
second nucleophilic attack: again, by a hydride
proton transfer: protonation
conversion of acid chloride into aldehyde
acid chloride is treated with 1) LiAl(OR)3H and 2) H3O+ to produce aldehyde
reacts with acid chloride rapidly and with aldehyde more slowly
allows aldehyde to be isolated
addresses the issue of using one equivalent of LiAlH4
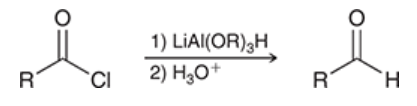
reaction between acid chloride and organometallic reagents
acid chloride is treated with a Grignard reagent (in excess)
converts acid chloride into alcohol; two alkyl groups are introduced
2 steps are needed
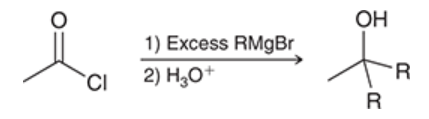
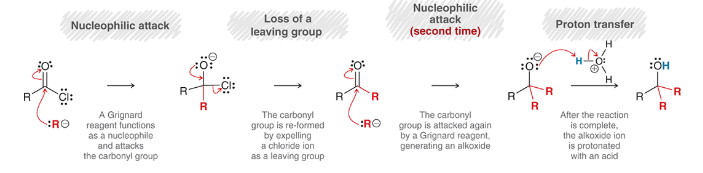
mechanism for reaction between an acid chloride and a Grignard reagent
nucleophilic attack: Grignard reagent is Nuc
loss of leaving group: Cl- is expelled (produces a ketone)
second nucleophilic attack: another Grignard reagent attacks carbonyl (produces an alkoxide ion)
proton transfer: protonation
acid chloride conversion to ketone
Grignard reagent cannot be used; not selective enough to make a ketone
more selective reagent, like Gilman reagent, is needed
Gilman reagent
a lithium dialkyl cuprate (R2CuLi)
used to convert an acid chloride into a ketone
similar to a Grignard reagent, but more selective
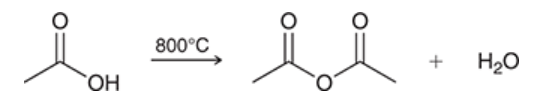
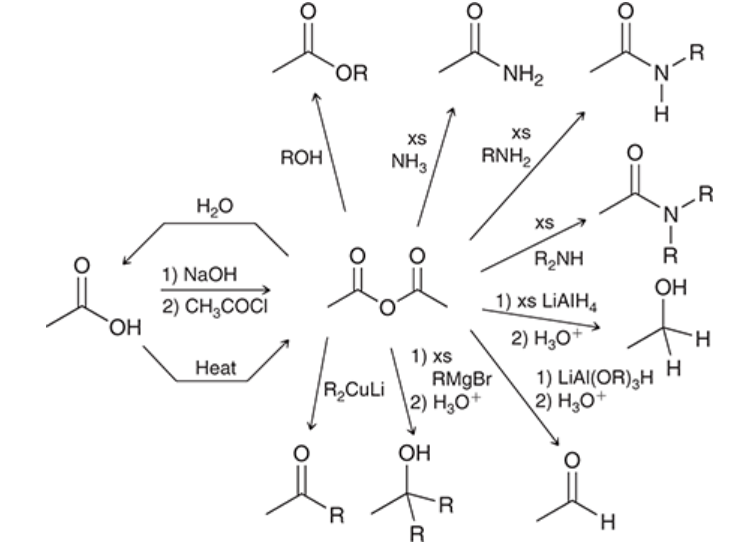
conversion of acetic acid into an acid anhydride
acetic acid is excessively heated (800*C) to convert into an acid anhydride and water
only practical for acetic acid because most other acids cannot survive the heat
conversion of carboxylic acids into acid anhydrides (not excessive heating)
acid chloride is treated with carboxylate ion
carboxylate functions as a nucleophile
prepares an acid anhydride
can produce symmetrical or unsymmetrical anhydrides
mechanism of acid chloride to acid anhydride conversion
nucleophilic attack
loss of leaving group: Cl- is expelled
reactions of acid anhydrides
reaction mechanisms are analogous to reactions of acid chlorides, except the leaving group is different
leaving group is a carboxylate ion
pyridine is not needed, because HCl is not produced
not as efficient as starting materials (when compared to acid chlorides) because of poor atom anatomy (half of the starting material is lost)
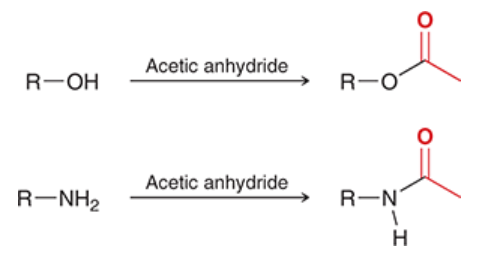
acetylation with acetic anhydride
an alcohol or amine is often acetylated with acetic anhydride
these reactions are used in preparation of aspirin and tylenol
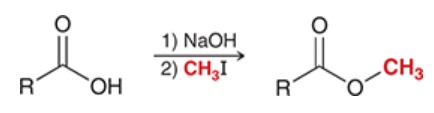
preparation of esters
carboxylic acid is treated with 1) strong base and 2) alkyl halide
carboxylic acid is deprotonated → yields carboxylate ion, which functions as nucleophile and attacks alkyl halide
Sn2 process
tertiary alkyl halides cannot be used
Fischer esterification
carboxylic acid is treated with alcohol in the presence of an acid catalyst
produces an ester
reversible
ester formation can be favored by either using excess of alcohol (use alcohol as solvent) or by removing water from reaction mixture as it is formed
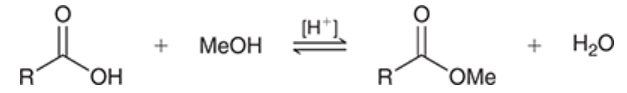
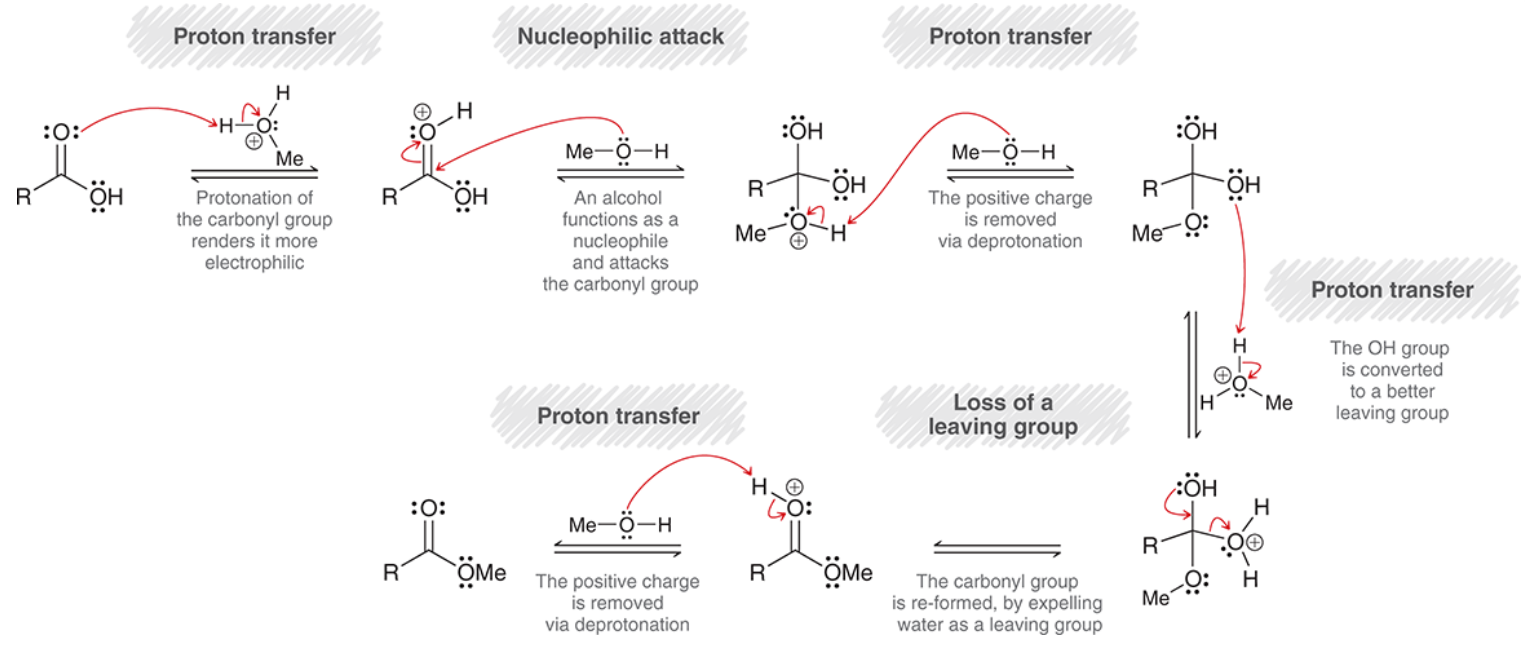
mechanism for Fischer esterification
proton transfer: carbonyl is protonated (makes it more electrophilic)
nucleophilic attack: alcohol is Nuc
proton transfer: deprotonation
proton transfer: OH is protonated
loss of leaving group: H2O is expelled
proton transfer: carbonyl is deprotonated
ester preparation via acid chloride
acid chloride is treated with alcohol
prepares an ester
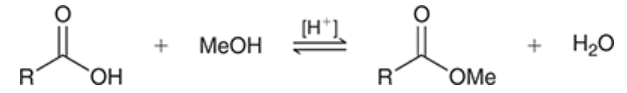
saponification
base-catalyzed hydrolysis of an ester
ester is treated with 1) NaOH and 2) an acid
converts ester into carboxylic acid
this process is used to make soap
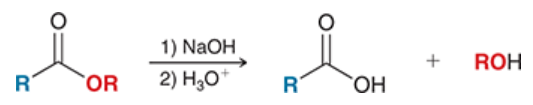
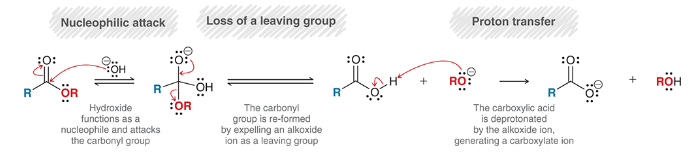
mechanism for saponification
nucleophilic attach: OH- is nucleophile
loss of leaving group: OR- is expelled, carbonyl reforms
proton transfer: deprotonation
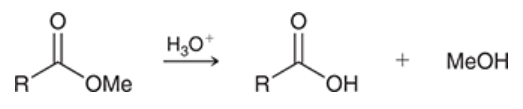
acid-catalyzed hydrolysis of esters
ester is treated with acid
produces carboxylic acid
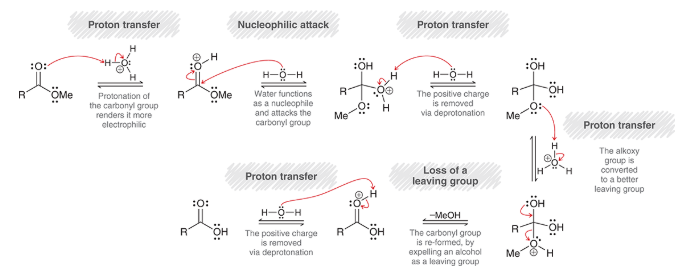
mechanism for acid-catalyzed hydrolysis of esters
proton transfer: protonation of carbonyl
nucleophilic attack: water is nucleophile
proton transfer: deprotonation of H2O
proton transfer: protonation of alkoxy group
loss of leaving group: MeOH is expelled
proton transfer: deprotonation
aminolysis of esters
ester is treated with amine, reacts slowly
yields amides
little practical utility, because amide preparation is achieved more efficiently from reaction between acid chlorides and ammonia (or 1* or 2* amines)
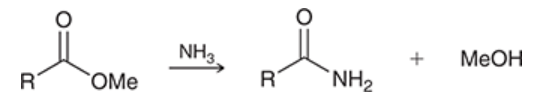
ester reduction with hydride-reducing agents
ester is treated with LAH
yields alcohol
mechanism for ester reduction with LAH
nucleophilic attack: LiAlH4 delivers hydride ion (Nuc)
loss of leaving group: OMe is expelled
second nucleophilic attack: another hydride attacks
proton transfer: protonation
ester reduction with DIBAH
ester is treated with 1) DIBAH and 2) HO+
yields aldehyde
performed at low temperature to prevent further reduction of the aldehyde
used to prevent further reduction that would happen if LAH was used
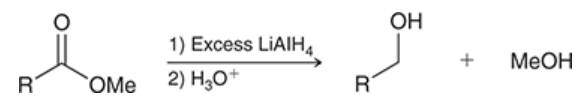
reaction between ester and Grignard reagents
ester is treated with 1) excess RMgBr and 2) H3O+
yields alcohol with two alkyl groups introduced
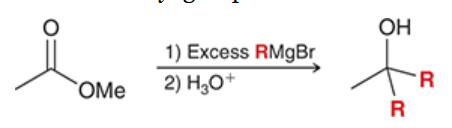
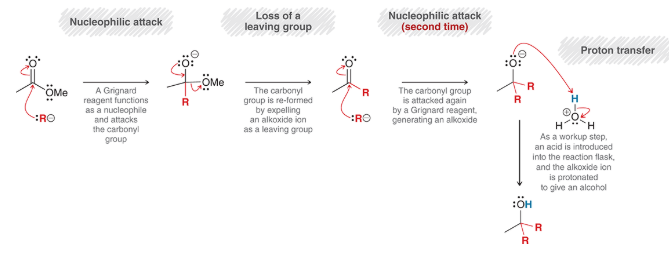
mechanism for reaction between an ester and Grignard reagent
nucleophilic attack: Grignard reagent is Nuc
loss of leaving group: OMe is expelled
second nucleophilic attack: another Grignard reagent attacks
proton transfer: protonation
amide preparation
amides can be made from any carboxylic acid (except nitriles)
most efficient preparation is from acid chloride, treated with 2 equivalents of NH3
(acid halides are best starting material because acid halides are the most reactive derivative)
acid-catalyzed hydrolysis of amides
amide is hydrolyzed in the presence of aqueous acid
gives carboxylic acid
process is slow and requires heating
ammonium ion (NH4+) is formed as a byproduct; much weaker than H3O+ (pKa 9.2 vs -1.7)
equilibrium greatly favors product formation → process is effectively irreversible
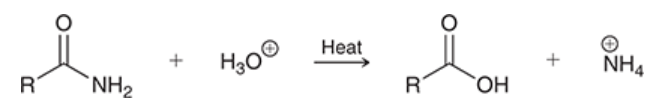
mechanism for acid-catalyzed hydrolysis of an amide
proton transfer: protonation of carbonyl
nucleophilic attack: water is Nuc
proton transfer: deprotonation of H2O
proton transfer: protonation of amino group
loss of leaving group: NH3 is expelled
proton transfer: deprotonation
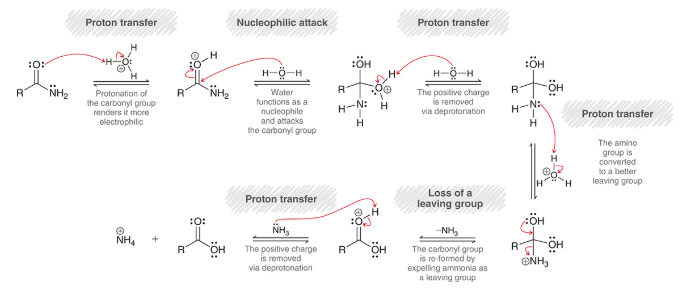
amide hydrolysis under basic conditions
amide is treated with 1) NaOH, heat and 2) H3O+
process is very slow; analogous to mechanism for ester saponification
mechanism for hydrolysis of amides under basic conditions
nucleophilic attack: OH- is Nuc
loss of leaving group: NH2 is expelled
proton transfer: deprotonation
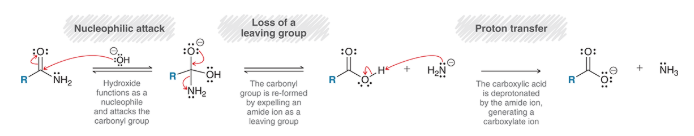
amide reduction
amide is treated with excess LAH and followed by aqueous workup
treated with 1) xs LiAlH4 and 2) H2O
different from most other reactions: carbonyl is completely removed
workup is different; H2O is used instead of H3O+ because amines get protonated in the presence of H3O+ to give RNH3+ ions (protonating the amine should be avoided)
nitrile preparation via Sn2
alkyl halide is treated with cyanide ion (NaCN)
yields nitrile
Sn2 mechanism
tertiary alkyl halides cannot be used
dehydration of amides
prepares nitrile from amide
amide is treated by SOCl2 or other reagents
benefit: can prepare tertiary nitriles, which cannot be prepared via Sn2
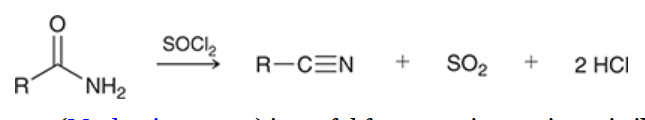
mechanism for dehydration of amides
nucleophilic attack: amide is Nuc
loss of leaving group: Cl- is expelled
proton transfer: positive charge on N is removed via deprotonation by base
proton transfer: eliminates proton and a leaving group
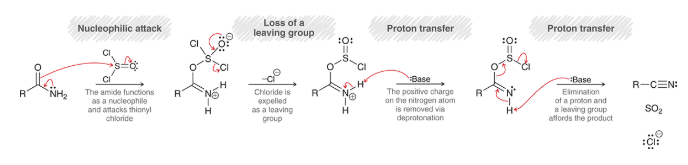
hydrolysis of nitriles
nitrile is hydrolyzed in aqueous acidic condition
yields amide
further hydrolysis yields carboxylic acid
mechanism for acid-catalyzed hydrolysis of nitriles
proton transfer: nitrile is protonated
nucleophilic attack: water is nucleophile
proton transfer: deprotonation removes positive charge
proton transfer: protonation to make intermediate
resonance rearrangement of intermediate
proton transfer: deprotonation
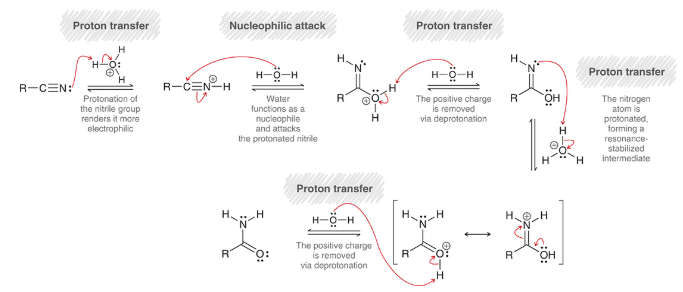
base-catalyzed hydrolysis of nitrile
nitrile is treated with 1) NaOH, H2O and 2) H3O+
nitrile is hydrolyzed in aqueous base; converted to amide then carboxylic acid
mechanism for base-catalyzed hydrolysis of nitriles
nucleophilic attack: OH- is nucleophile
proton transfer: protonation
proton transfer: deprotonation
resonance rearrangement
proton transfer: protonation
reaction between nitrile and Grignard reagent
Grignard reagent attacks nitrile
resulting anion is treated with aqueous acid → gives imine
imine is hydrolyzed to ketone under acidic conditions
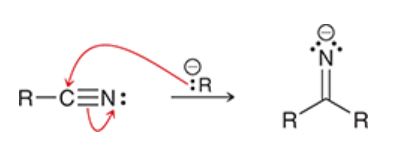
reduction of nitriles
nitrile is treated with 1) xs LiAlH4 and 2) H2O
yields amine
important to use H2O for workup instead of H3O+ to avoid protonation of amine
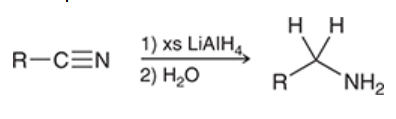
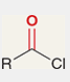
IR signal for an acid chloride carbonyl
~1790 cm-1
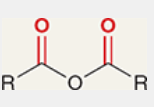
IR signals for an acid anhydride carbonyls
1760 and 1820 cm-1
two signals