Stage 2 Chemistry, Unit 1.4- Monitoring the Environment-Chromatography
1/19
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
What is Chromatography?
a technique used to isolate and identify individual components of a mixture
What does Chromatography usually contain?
A Stationary and mobile phase
What is a stationary phase?
A solid or liquid medium that components are adsorbed to.
What is a mobile phase?
A liquid or gas that flows through the stationary phase, taking components with it
What determines the seperation of components in Chromatography?
there is a difference in polarity between the mobile and stationary phase, so chemicals with differing polarity are attracted to their likes.
the effectiveness of separation is dependent on the strength of interaction and attraction of substances between the two phases.
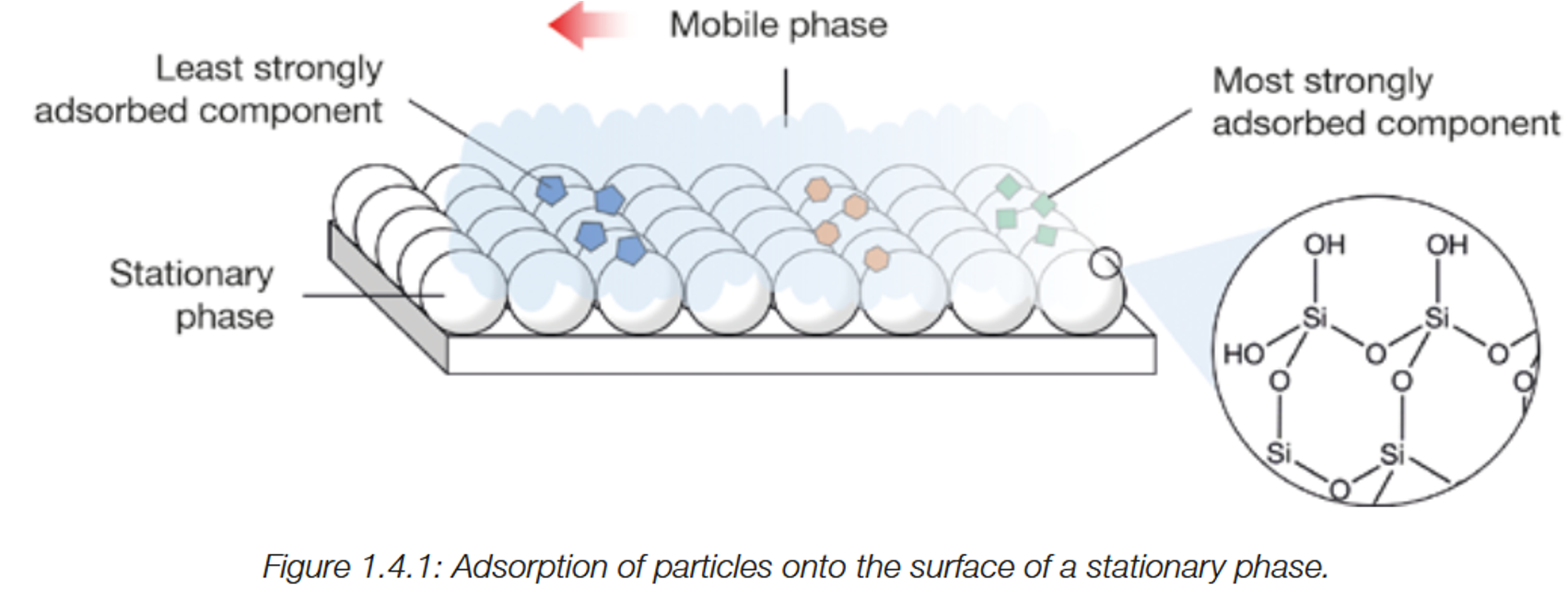
If the stationary phase is polar, and there is a slow moving substance, this means the substance is
highly polar
If the stationary phase is polar, and there is a fast moving substance, this means the substance is
less polar (or non polar)
If Strongly adsorbed to the stationary phase,
a component will travel slower
If less strongly adsorbed to the stationary phase
the component will travel faster
What is Adsorption?
the attraction of molecules to the surface of a solid).
Thin Layer Chromatography (TLC)
Has a thin layer of adsorbent material (alumina or silica) material as its stationary phase. The sample is placed on this stationary phase.
The mobile phase is a solvent which moves up the stationary phase, moving individual compounds.
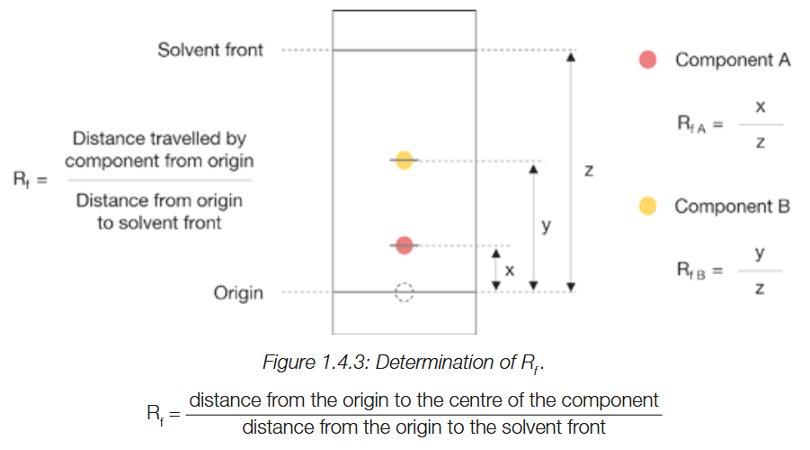
How is Rf determined?
RF is the distance that each component has travelled from its origin, divided by the distance from the origin to solvent front(when the TL was removed from the solvent).
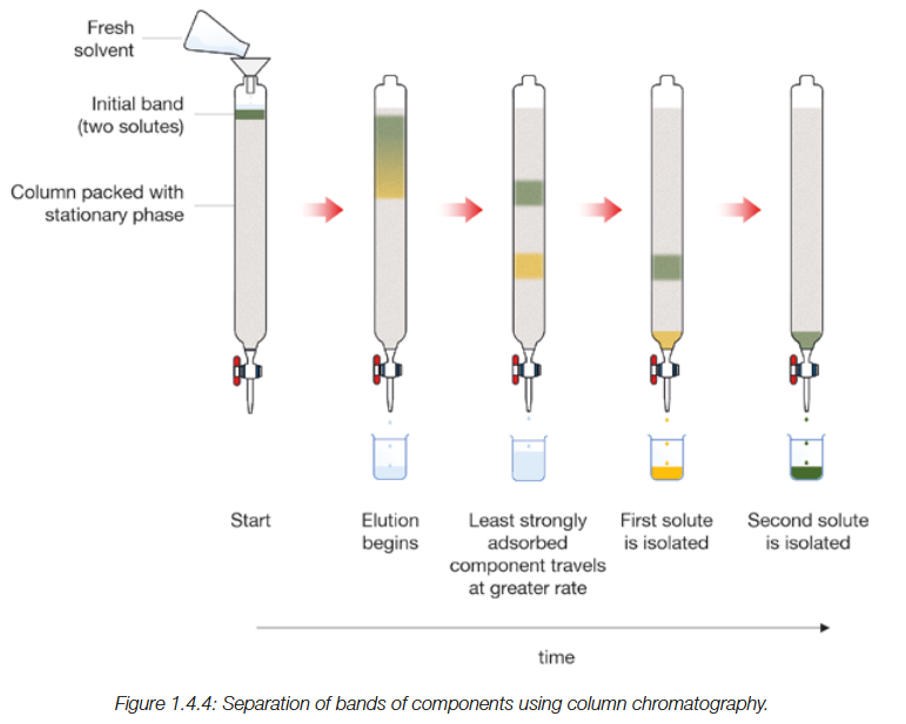
Column Chomatography
A collumn is packed with a solid adsorbent (stationary phase), while a eluent mobile phase passes through it due to gravity. The components can travel at seperate speeds, with strongly adsorbed traveling slowly. This allows isolation andseperation of the two components.
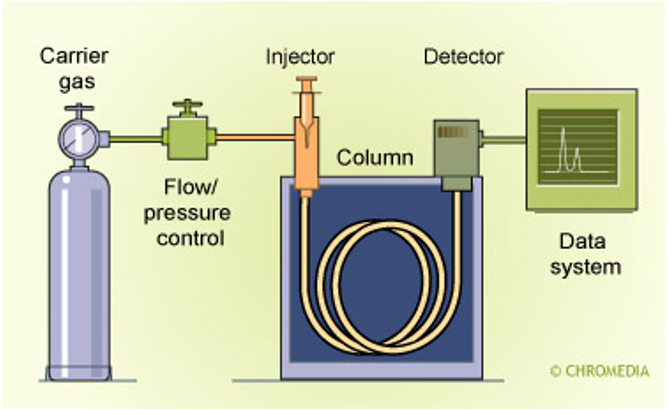
Gas Chromatography
Allows for the separation of small quantities of gases.
The mobile phase is an inert carrier gas,
A capillary column has the stationary phase coated to it.
components are retained in the column depending on the absorbency, eluting later if strongly adsorbed
Gas Chromatography rentention time
Allows the concentration of each chemical, and how well it retained to the stationary phase to be viewed
High- Performance Liquid Chromatography
The stationary phase is short, with high surface area.
A liquid solvent is the mobile phase, which transports the components through the HPLC.
Components move slower if strongly adsorbed to the stationary phase, causing substances to elute at different times.
How is HPLC a better alternative to gas chromatography?
HPLC is best for mixtures that may deteriorate in the high temperatures of GC.
HPLC is more effective than gas chromatography due to smaller particles in the shorter stationary phase, leading to a larger surface area.
Ion Chromatography
the mobile phase carries the sample through the stationary phase.
Stationary phase is a solid ion exchange resin (either positively or negatively charged) before use, the stationary phase is coated in ions of different charge (different than the charge of the phase itself but same as the sample.)These exchangeable ions take up the binding sites.
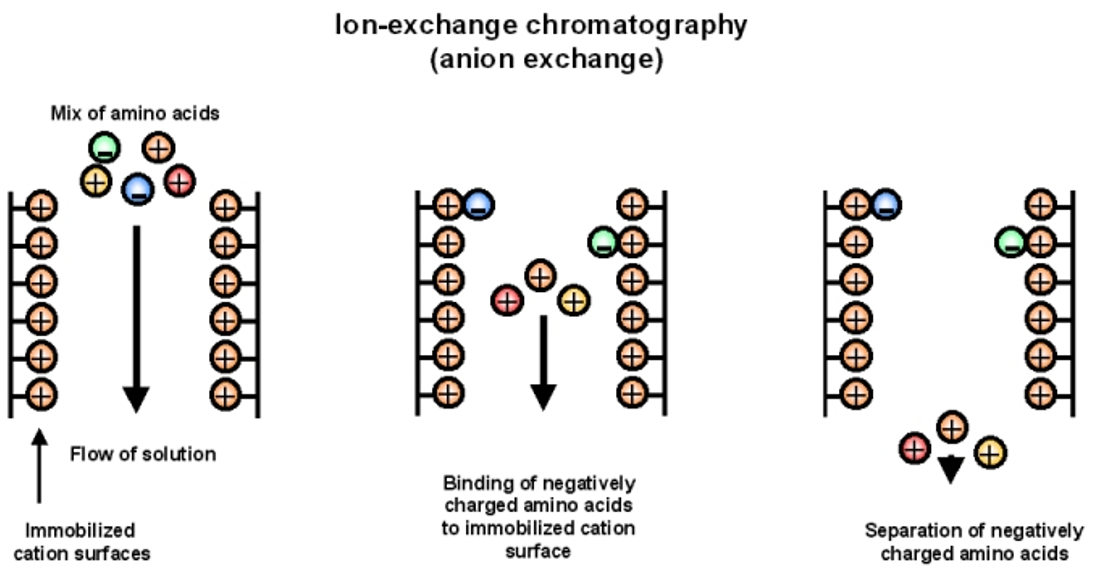
How does Ion Chromatography work?
Chromatography that occurs due to ion exchange occurring in a equilibrium system.
If ions that have a higher electrostatic force of attraction are present, they will exchange with those binded to the stationary phase. Only ions with strong electrostatic forces will remain after the elution of the solvent.
How is Ion chromatography reset?
the ion concentration of the resin is altered, by flooding the system with a high concentration of the original coating, placing stress on the equilibrium forcing the reserve reaction
Resin-(s)Na+ + NH+4(aq) ⇌ Resin-(s)NH+4 + Na+(aq)