Glucose & Hormone Regulation
1/312
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
313 Terms
What are the endocrine glands in the body?
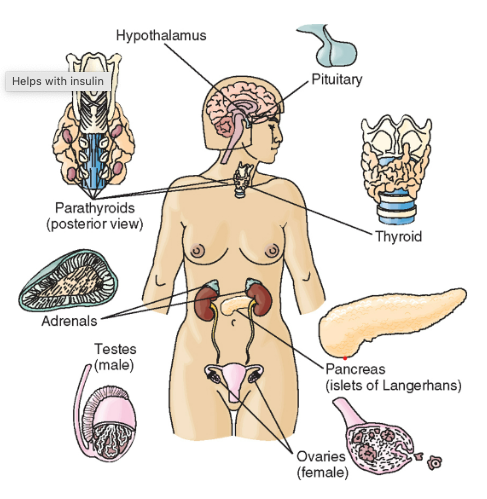
What is the pathophysiology of hormone secretion?

What is glucoses needed for?
Energy in order to function
What happens when we consume glucose?
Consuming glucose causes blood sugar levels to rise, which signals the pancreas to release insulin
What role does insulin play in glucose metabolism?
Insulin is required for glucose to enter muscle, liver, and fat cells
What role does insulin play in glucose metabolism?
Insulin is required for glucose to enter muscle, liver, and fat cells
How is glucose used by the body after entering cells?
Glucose is either transported and used by the body for energy or stored in the liver as glycogen
Besides glucose metabolism, what other roles does insulin play?
Insulin also plays a role in protein synthesis and fat deposition (storage)
What triggers the pancreas to stop releasing insulin and release glucagon?
A decrease in blood sugar levels (e.g., between meals) triggers the pancreas to stop releasing insulin and release glucagon
What does glucagon do when blood sugar levels are low?
Glucagon stimulates the liver to convert glycogen back into glucose (glycogenolysis), raising blood sugar levels
How does glucagon help raise blood sugar levels?
Glucagon causes protein to be released from muscle cells and fat from adipose tissue, which are transformed into glucose (gluconeogenesis) in the liver
What does a decrease in blood sugar signal the adrenal glands to do?
A decrease in blood sugar signals the adrenal glands to release epinephrine
What are the effects of epinephrine on blood sugar regulation?
Epinephrine increases glycogenolysis and gluconeogenesis in the liver, reduces insulin secretion while increasing glucagon release, reduces glucose uptake and utilization, increases glycolysis in muscle, and increases lipolysis in adipose tissue
How does epinephrine contribute to hypoglycemia symptoms?
An increase in epinephrine leads to the initial signs and symptoms of hypoglycemia
What medication is used to treat T2DM in conjuction with metformin?
1. Sulfonylurea (class)/ gliclazide (drug): Gliclazide is another common medication used to treat T2DM and is often used in conjunction with metformin.
Explain the lock and key of the mechanism of hormone regulation
Explanation Using Insulin as an Example:
Step 1: Hormone Release
After you eat a meal, your blood sugar (glucose) levels rise.
The pancreas detects this increase and secretes insulin into the bloodstream.
Insulin (represented as blue shapes) travels through the blood vessels (capillaries), reaching different parts of the body.
Step 2: Target Cells vs. Nontarget Cells
Insulin does not affect all cells—only the ones that have specific insulin receptors.
Target Cells:
These are cells in organs like the muscles, liver, and fat tissue.
They have special purple receptors that match the shape of insulin, allowing it to bind.
Nontarget Cells:
These cells do not have insulin receptors.
Since insulin cannot bind to them, these cells are not affected by insulin.
Step 3: Lock-and-Key Mechanism
Insulin works like a key, and its receptor on the target cell is like a lock.
When insulin binds to its receptor:
It sends a signal inside the cell.
This signal activates glucose transporters (proteins in the cell membrane). = opens up chambers to tissues so that insulin can be utilized
These transporters pull glucose from the bloodstream into the cell.
Step 4: No Binding = No Effect
If a cell does not have the insulin receptor, insulin cannot bind to it.
Since insulin does not attach, there is no effect on that cell.
This ensures that insulin only influences cells that need to take up glucose.
What are the 2 main organs that maintain a normal blood glucose level?
Pancreas and the liver
Where is the pancreas located in and plays both what roles?
Located behind the stomach
Plays both exocrine and endocrine roles
What percentage of the pancreas does the exocrine function?
98%
Most of the pancreas is involved in. a digestive enzyme secretion (through the ductal system)
What percentage of the pancreas does the endocrine function?
Islets of Langerhans (tiny clusters of hormone producing cells) make up about 2% of the pancreas gland but plays a major role in blood sugar regulation
What are the different cell types in the Islets of Langerhans?
Alpha cells
Beta cells
Delta cells
PP cells
Capillaries
What do beta cells (β-cells) produce and secrete?
Insulin
Make up 50% of Islets
Lowering blood sugar by helping cells absorb glucose
What do alpha cells (α-cells) produce and secrete?
Glucagon
Raising blood sugar by triggering the liver to release stored glucose
What do delta cells produce?
Somatostatin
Regulates other hormones which slows down insulin and glucagon release
What do PP cells release?
Pancreatic polypeptide
Helps regulate digestion and appetite
What do capillaries do in the Islets of Langerhans?
Surrounds the Islets, allowing hormones to enter the bloodstream and reach target organs
How is insulin released into the body (+after eating)?
Continuously released (basalrate) into the body, with increased amount released after eating (bolus rate)
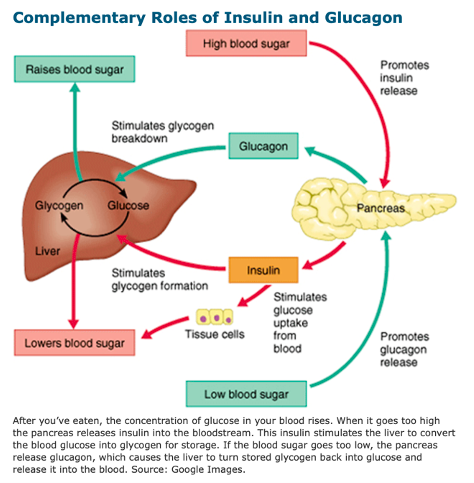
What does insulin regulate?
Metabolism ad storage of ingested carbohydrates, fats, and proteins
What does insulin transport?
Glucose (+ potassium) across cell membranes
Why do some cells not require insulin to transport glucose (+ examples)?
Some cells do not require insulin to transport glucose because they use insulin-independent glucose transporters (GLUTs) that allow glucose to enter the cell without insulin's help. These cells need a constant supply of glucose, even during fasting or low insulin levels, to maintain essential functions.
Brain:
Why? The brain relies on a constant glucose supply for energy.
Transporter Used? Insulin-Independent
Nerve Cells:
Why? They do not need insulin to transport glucose because they rely on a continuous and uninterrupted energy supply to function properly
The brain is one of the most energy-demanding organs, consuming about 20% of the body's glucose even at rest.
If neurons depended on insulin, any drop in insulin levels (like during fasting, stress, or diabetes) could starve the brain of glucose, leading to dysfunction or even coma.
When is glucagon released?
In response to low levels of blood glucose, protein ingestion, and exercise
How and why is glucagon counter regulatory to insulin?
While insulin lowers blood sugar, glucagon raises it when levels drop too low. This counter-regulation prevents extreme highs (hyperglycemia) or lows (hypoglycemia), keeping glucose levels stable for normal body function.
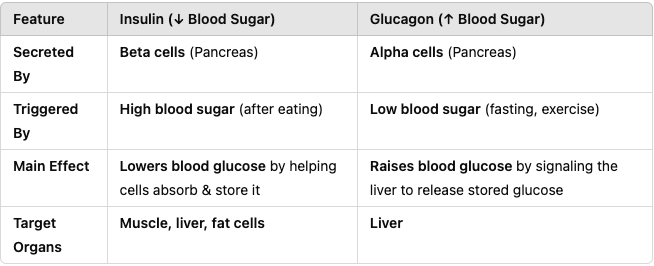
How does the body regulate blood glucose?
The body maintains blood glucose balance through insulin and glucagon, two counter-regulatory hormones secreted by the pancreas. This ensures that glucose levels remain within the normal range (70-99 mg/dL fasting, <140 mg/dL after meals).
1. Normal Blood Sugar Ranges
Fasting blood sugar (before meals): 70–99 mg/dL (3.9–5.5 mmol/L)
Postprandial blood sugar (1-2 hours after eating): Less than 140 mg/dL (7.8 mmol/L)
HbA1c (average over 3 months): Below 5.7%
For people with diabetes, target ranges may be slightly different based on individual health conditions
When Blood Sugar is High (After Eating) → Insulin is Released Lowering Glucose
Triggered by: Eating a meal, leading to increased glucose in the blood
Pancreas Response: Releases insulin (from beta cells)
Insulin Actions:
Stimulates cells (muscles, liver, fat) to absorb glucose.
Promotes glucose storage as glycogen in the liver (glycogenesis).
Inhibits glucagon release (so the liver stops making glucose).
→ Outcome: Blood sugar decreases to normal levelsGoal: Prevents hyperglycemia (high blood sugar) and stores energy for later.
When Blood Sugar is Low (Fasting, Exercise) → Glucagon is Released
Triggered by: Fasting, exercise, or low blood sugar
Pancreas Response: Alpha cells in the pancreas release glucagon
Glucagon Actions:
Stimulates the liver to break down glycogen into glucose (glycogenolysis)
Signals the liver to create new glucose from proteins & fats (gluconeogenesis)
Releases glucose into the bloodstream to increase blood sugar
→ Outcome: Blood sugar rises back to normal
Goal: Prevents hypoglycemia (low blood sugar) and ensures continuous energy supply
What are the major pancreatic hormones?
Insulin
Glucagon
What does insulin do to blood sugar?
Lowers blood sugar
What is insulin required for? + why?
Glucose uptake in skeletal, fat, and heart muscle
Stimulates the movement of glucose transporter proteins to the cell membrane. These transporters allow glucose to enter the cells for energy production or storage. Without insulin, these transporters are less active, making it difficult for cells to take up glucose efficiently.
What does insulin suppress? + why?
Suppress liver production of glucose and promotes glycogenesis (Stimulates the liver to break down glycogen into glucose)
Insulin suppresses liver production of glucose (gluconeogenesis) and promotes glycogenesis (the formation of glycogen) because when blood glucose levels are high, the body focuses on storing energy rather than producing more. Insulin signals the liver to store glucose as glycogen, preventing excess glucose production and helping to regulate blood sugar levels.
What does insulin increase? + why?
Protein and Lipid synthesis
Insulin is a storage & growth hormone that promotes nutrient uptake and energy storage
Protein Synthesis (Muscle Growth):
Increases amino acid uptake into muscle cells
Activates protein synthesis & prevents protein breakdown
Lipid Synthesis (Fat Storage):
Increases glucose uptake in fat cells
Stimulates lipogenesis (glucose → fatty acids → triglycerides)
Inhibits lipolysis (fat breakdown)
Purpose: Stores energy for future use—carbs as glycogen, proteins for growth, fats as triglycerides!
What does insulin inhibit? + why?
Ketogenesis (fat to glucose) and gluconeogenesis (protein to glucose)
Insulin inhibits ketogenesis and gluconeogenesis because when insulin levels are high, glucose is readily available for energy. The body prioritizes storing energy rather than producing more. Insulin promotes fat storage, inhibits fat breakdown (lipolysis), and reduces the need for protein breakdown into glucose, as the body has sufficient glucose from meals.
What does insulin convert? + why?
Excess glucose to free fatty acids
Insulin converts excess glucose to free fatty acids because when glucose levels are high (such as after eating), insulin promotes the storage of energy. It stimulates the conversion of glucose into fat (lipogenesis) to store it for later use, preventing excessive blood sugar levels and ensuring energy availability for future needs.
What does glucagon do to blood glucose?
Raises blood glucose
What is the relationship between glucagon and insulin?
Counter regulatory
What does glucagon stimulate?
Glycogenolysis
(The breakdown of glycogen into glucose) to increase blood sugar levels when they are low, such as during fasting or between meals. It activates enzymes in the liver and muscles that break down stored glycogen into glucose, which is then released into the bloodstream to provide energy for the body.
Gluconeogenesis
(The production of glucose from non-carbohydrate sources like amino acids and glycerol) to increase blood glucose levels during periods of fasting or low blood sugar. This ensures a continuous supply of glucose for vital functions, especially for the brain, by promoting the synthesis of glucose in the liver.
→ Stimulates glucose to be released from the liver
What does endogenous secretion mean?
Endogenous insulin secretion refers to the natural production and release of insulin by the pancreas, specifically the beta cells in the islets of Langerhans, into the bloodstream. This process occurs in response to changes in blood glucose levels.
"Endogenous" means originating from within the body, so endogenous insulin secretion refers to the insulin your body produces naturally to regulate blood sugar levels
It contrasts with exogenous insulin, which refers to insulin that is injected or administered from an outside source (like insulin injections for people with diabetes).
Endogenous insulin secretion describes how the body naturally increases or decreases insulin levels based on factors like food intake, carbohydrate absorption, and the body's needs for glucose regulation.
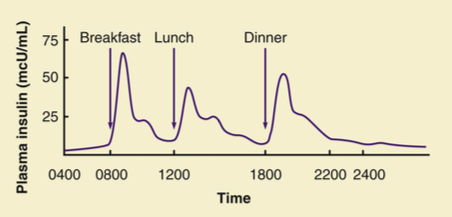
What is the pattern of normal endogenous insulin secretion after meals?
After meals, insulin concentrations rise rapidly in the first hour or two and peak at about 1 hour.
As carbohydrate absorption from the gastrointestinal tract declines, insulin concentrations promptly decrease toward preprandial values.
After carbohydrate absorption from the gastrointestinal tract is complete and during the night, insulin concentrations are low and constant, with a slight increase at dawn.
What are the values for hypoglycemia?
<4 mmol/L
What are the values for euglycemia?
4 to 6 mmol/L
What are the values for hyperglycemia?
>6 mmol/L
What does Diabetes Mellitus mean in latin?
Diabetes Mellitus: (Diabetes = fountain; Mellitus = honey)
What is Diabetes Mellitus?
Multisystem disease related to abnormal insulin production, impaired insulin utilization, or both
Metabolism disorder impacting carbohydrates, fats, and proteins
What are the types of diabetes mellitus?
Type1
Type 2 (prediabetes)
Gestational
Other (secondary diabetes (pancreatic cancer))
How many adults are living with diabetes?
830 million
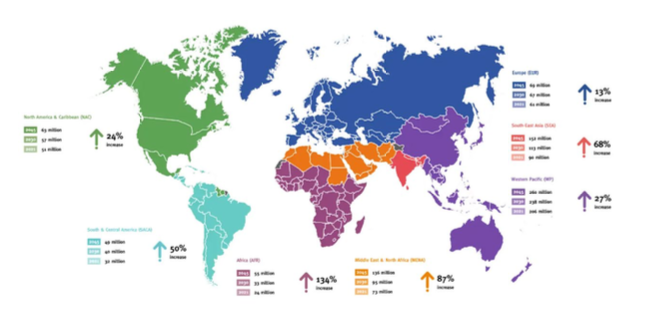
How many in out of 4 adults live with diabetes in low and middle income countries?
3 out of 4
How many Canadians live with diabetes or prediabetes?
11.7 million
Among Manitobans, what is the % of people living with diabetes or prediabetes?
30%
Among Manitobans, what is the % of people living with diagnosed diabetes (Type 1 and Type 2)?
11%
What can diabetes mellitus complications lead to?
Skin Problems (poor wound healing), Eye Problems (blindness, cataracts, glaucoma), Foot and Leg Amputations (poor circulation), Premature Death
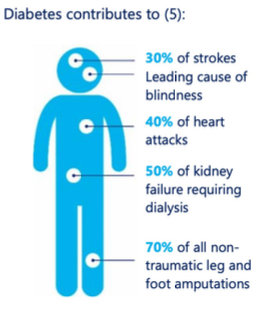
How many years does diabetes reduce the lifespan by?
5 to 15 years
What is the cost of diabetes mellitus to the province?
$145 million
What are the determinants of health-diabetes?
Income and social status:
Factors: in Western world
Living longer
obesity
Sedentary lifestyle
Biology and genetics:
Genetic factor (Indigenous population, hispanic, asian, southeasian, african)
Personal health practices and coping skills:
We can prevent it by weight loss and weight control; which may be harder to implement in northern communities (have to be mindful of that situation)
What is the pathophysiology of diabetes mellitus?
Inadequate/Absent production of insulin
Insulin Resistance
Elevated Stress (Cortisol)
What is the outcome of an inadequate/absent production of insulin (diabetes mellitus)?
Destruction of beta(ß) cells
Genetics, viruses, pancreatic cancer, pancreatitis, idiopathic (unknown)
Over production of insulin – destroyed over time
What is the outcome of insulin resistance (diabetes mellitus)?
Insulin resistance: target cells are less able to metabolize the glucose available to them => hyperglycemia => hyperinsulinemia
Reduced binding of insulin to its receptors
Reduced receptor numbers
Reduced receptor responsiveness
What is the outcome of elevated stress (cortisol)(diabetes mellitus)?
Pro-inflammatory state
Stimulated glycogenolysis and gluconeogenesis
E.g., trauma, living conditions, societal, infection
What is the peak onset of diabetes mellitus?
11 - 13 yrs; can extend to 35 yrs
What is the cause of diabetes mellitus?
Autoimmune destruction of pancreatic beta cells
80-90% of beta (ß) cells are destroyed before hyperglycemia
and other manifestations occur
Long pre-clinical period – patient becomes symptomatic when they can no longer produce insulin which leads to rapid onset of symptoms
Treatment requires exogenous insulin
What causes the autoimmune destruction of the beta (ß) cells?
Genetic predisposition, virus, idiopathic (unknown etiologies)
What are the clinical manifestations of type 1 diabetes mellitus?
Typically, abrupt onset of symptoms:
Glucosuria (glucose in the urine)
Polyuria (frequent urination)
Polydipsia (excessive thirst)
Polyphagia (excessive hunger)
Weight loss (underweight)
What is the most prevalent type of DM?
> 90% of patients with DM
What are the risk factors of type 2 diabetes mellitus?
Obesity (central adiposity), family pre- disposition, low income (food insecurity), high-risk populations, HTN, dyslipidemia, antipsychotic medications (used to treat schizophrenia)
central adiposition = fat in midriff
→ cuts down to 58% by exercising and losing weight
What is the etiology and pathophysiology of type 2 diabetes mellitus?
Pancreascontinuestoproducesomeendogenousinsulin
Insulin resistance: target cells are less able to metabolize the glucose available to them => hyperglycemia => hyperinsulinemia
Reduced binding of insulin to its receptors
Reduced receptor numbers
Reduced receptor responsiveness
Hyperinsulinemia leads to increased glucagon release from pancreas leading to increased glucose production by the liver
Impaired insulin secretion: insulin release no longer tightly coupled to plasma glucose content
Gradual loss of Islet of Langerhans function– beta cells (β) fatigued or beta cell mass is lost
What are the 4 factors of diabetes?
Insulin resistance
Marked decrease ability of pancreas to make insulin
Liver inappropriately secretes glucose (not as common)
Production of hormones and cytokines by adipose tissues can play a role insulin sensitivity
adeponeptin and lepetin
What are the clinical manifestations of type 2 diabetes mellitus?
May experience some of the clinical manifestations of Type 1 DM
Typically, a gradual onset
Patients may present with chronic complications of hyperglycemia:
Fatigue, recurrent infections, prolonged wound healing, visual acuity changes, and painful peripheral neuropathy in the feet
Lots of sugar in body = sugar is sticky (ends up producing anthereloscresos or plaque); issues to perfusion
→ Numbness in the feet/pain (assess for PVD and injury)

What are the four methods of diagnosis (for both Type 1 and Type 2)?
must be confirmed w/ other two positive tests
HemoglobinA1C > 6.5%
Determining glycemic control over time- shows the amount of glucose that has been attached to Hgb molecules over the lifespan of the blood cell (120 days)
Overall glucose control ‘average’ over last 90-120 days.
Fasting blood glucose >7mmol/L (fast for 8 hours, water intake is ok)
Random plasma blood glucose >11.1, plus classic symptom of DM
2 hour - Oral glucose tolerance test:
Patient drinks 75g of glucose, blood/capillary blood glucose is measured at 30, 60, and 120 min.
Normal: <11.1 mmol/L at 30 and 60 min; < 7.8 mmol/L at 120 min
What is the screening for type 2 diabetes in adults?
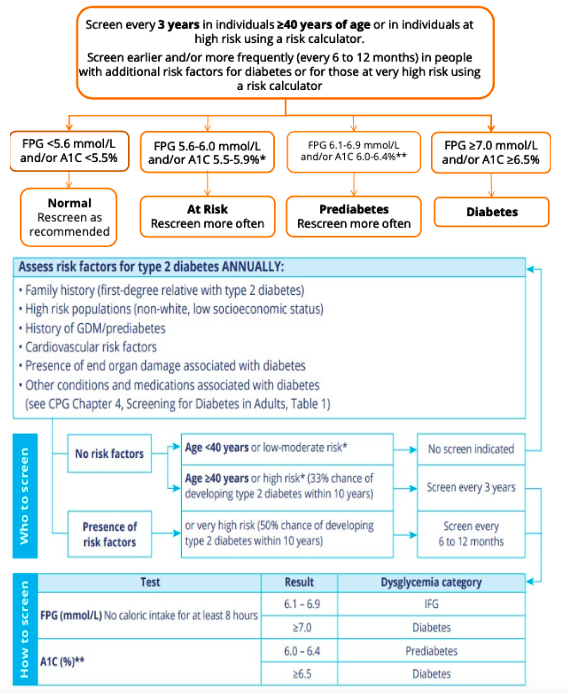
How frequently should a patient’s blood sugar be monitored?
When would a nurse perform a blood glucose test?
How does a nurse perform a bedside blood glucose test?
What is the normal blood glucose level?
Monitoring frequency depends on diabetes type and treatment:
Type 1 Diabetes: 4+ times daily (before meals, before bed, and as needed)
Type 2 Diabetes (on insulin): At least 2–4 times daily, or as directed
Type 2 Diabetes (not on insulin): Varies—may check daily or a few times per week, based on treatment and glycemic control
Continuous Glucose Monitoring (CGM): Provides real-time data for better management, especially for insulin-dependent patients
When would a nurse perform a blood glucose test?
A nurse would perform a blood glucose test in the following situations:
Before meals and bedtime (for diabetic patients on insulin)
Before administering insulin or other glucose-lowering medications
If a patient shows signs of hypoglycemia (shakiness, sweating, confusion) or hyperglycemia (excessive thirst, frequent urination, fatigue)
During acute illness, infection, or stress, which can affect blood sugar levels
Before and after surgery or other medical procedures
For pregnant patients with gestational diabetes
As part of routine monitoring in hospitalized patients with diabetes
How does a nurse perform a bedside blood glucose test?
Steps to perform a bedside blood glucose test:
Gather supplies – Glucometer, test strip, lancet, alcohol swab, gloves, and gauze.
Verify patient identity and explain the procedure.
Perform hand hygiene and put on gloves.
Prepare the glucometer – Insert a test strip into the device.
Clean the patient’s fingertip with an alcohol swab and let it dry.
Use the lancet to prick the side of the fingertip (avoids nerve endings in the center).
Gently squeeze the finger to obtain a drop of blood.
Touch the blood drop to the test strip and allow the glucometer to read.
Wait for the result, then document the reading.
Apply gauze if needed and dispose of used materials properly.
Interpret and act on the result (e.g., administer insulin if needed, notify provider if abnormal).
Perform hand hygiene and ensure patient comfort.
What is the normal blood glucose level?
Euglycemia: 4 to 6 mmol/L
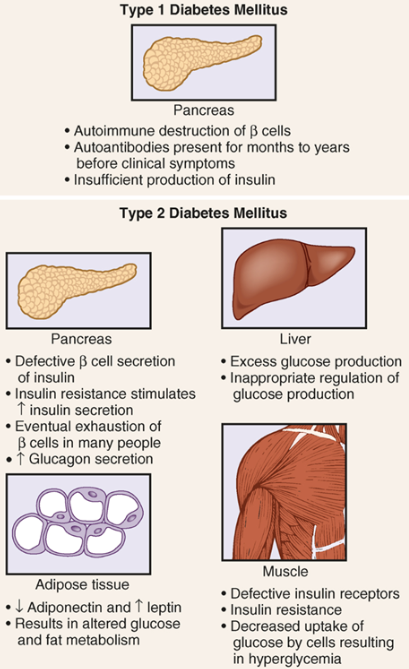
🩸 Type 1 Diabetes Mellitus (T1DM)
Cause: Autoimmune destruction of β (beta) cells in the pancreas.
Key Features:
The body attacks its own insulin-producing cells.
Autoantibodies can be detected months to years before symptoms appear.
No insulin production, leading to high blood glucose (hyperglycemia).
Treatment: Requires exogenous insulin (injections or pump) for survival.
🩸 Type 2 Diabetes Mellitus (T2DM)
Cause: Insulin resistance and eventual β-cell dysfunction.
Key Features:
Pancreas: Initially, β cells work harder to produce more insulin, but over time, they become exhausted.
Liver: Produces excess glucose, worsening hyperglycemia.
Muscle: Insulin resistance—muscles can't effectively take up glucose.
Adipose tissue: Altered metabolism due to low adiponectin and high leptin, contributing to insulin resistance.
Treatment: Lifestyle changes, oral medications, and possibly insulin therapy in later stages.
Comparison Chart
Feature | Type 1 Diabetes Mellitus | Type 2 Diabetes Mellitus |
|---|---|---|
Cause | Autoimmune β-cell destruction | Insulin resistance & β-cell dysfunction |
Insulin production | None or very little | Initially high, but decreases over time |
Onset | Abrupt, usually in childhood or young adulthood | Gradual, often in adults (but increasing in youth) |
Pancreas | Destroys β cells → No insulin | Insulin resistance → Overworked β cells → Failure |
Liver | Normal | Excess glucose production, worsening hyperglycemia |
Muscle | Normal | Defective insulin receptors, ↓ glucose uptake |
Fat metabolism | Normal | Altered by ↓ adiponectin & ↑ leptin, affecting glucose and fat metabolism |
Glucagon levels | Normal | Increased, leading to even higher glucose levels |
Treatment | Insulin required for survival | Lifestyle, oral meds, insulin (if needed later) |
What are the clinical manifestations of hyperglycemia?
Polyuria: Frequent urination
Polydipsia: Excessive thirst
Cellular dehydration: Cellular dehydration; occurs due to increased osmotic diuresis and increased serum osmolality caused by high blood glucose levels
Blurred vision: Can occur when high blood glucose levels lead to cellular dehydration and increased serum osmolality, affecting the eyes
Hyperglycemia causes increased osmotic diuresis and increased serum osmolality
Polyphagia
Elevated blood glucose
Increase in appetite followed by lack of appetite
Weakness, fatigue
Headache
Nausea and vomiting
Abdominal cramps
From undiagnosed DM, untreated/under- treated diabetes, inactivity, stress, acute illness, infection, surgery, medications (e.g. corticosteroids)
How does hyperglycemia progressed to DKA or HHS?
DKA (Diabetic Ketoacidosis)
Primarily in Type 1 DM due to absolute or near-absolute insulin deficiency.
Lack of insulin leads to the body breaking down fats for energy, producing ketones.
Excessive ketones cause metabolic acidosis.
Hyperglycemia persists as glucose cannot enter cells.
Kidneys excrete excess glucose (glucosuria), leading to osmotic diuresis, causing dehydration and electrolyte imbalances.
Precipitated by illness, infection, inadequate insulin, or undiagnosed Type 1 DM.
HHS (Hyperosmolar Hyperglycemic State)
Commonly seen in Type 2 DM with some endogenous insulin.
Extreme hyperglycemia (blood glucose > 34 mmol/L) and increased serum osmolality.
Relative insulin deficiency prevents extreme hyperglycemia complications but is insufficient to manage glucose.
Leads to severe dehydration due to osmotic diuresis.
Often occurs when hyperglycemia is not recognized or adequately treated, frequently with illness or infection.
Less ketone production compared to DKA.
What are signs of insulin resistance?
Acanthosis Nigricans
Skin tags
What are acute complications of diabetes mellitus?
Diabetic Ketoacidosis (DKA), HHS
How does diabetic ketoacidosis occur?
In the absence of insulin, fats are metabolized instead of glucose
Where is DKA mostly seen?
Type 1 DM
What is DKA characterizied?
Hyperglycemia, ketosis, metabolic acidosis, vomiting, and dehydration (osmotic diuresis leading to hypokalemia)
Ketones are an acidic byproduct of fat metabolism
Ketonuria – ketones secreted in the urine
What are clinical manifestations of DKA?
Hypovolemia, hypokalemia, Kussmaul’s respirations, fruity breath (bc of ketones)
What does insulin deficiency stimulate (DKA)?
Stimulates the production of glucose from amino acids in the liver further increasing the hyperglycemia
What are the preciptiating factors of DKA?
Illness, infection, inadequate insulin dosage, insulin omission, undiagnosed Type 1 DM, poor self-management
Where is Hyperosmolar hyperglycemic state (HHS) commonly seen?
People with Type 2 DM who have ability to produce some endogenous insulin
→ can be extreme before HHS is recognized
What are the laboratory values of HHS (diabetes mellitus)?
Blood glucose greater than (>) 34mmol/L, increased serum osmolality
What are the treatment of HHS (diabetes mellitus)?
Fluid replacement (0.9% or 0.45% NS)
What causes blood sugar (BS) to drop below 4 mmol/L?
Too much insulin in relation to available glucose leads to hypoglycemia
How does the body respond to hypoglycemia?
It triggers the release of neuroendocrine hormones (e.g., epinephrine), activating the autonomic nervous system and causing neurogenic symptoms
What happens if blood sugar continues to drop?
The brain lacks glucose (neuroglycopenia), leading to severe symptoms like confusion, loss of consciousness, seizures, coma, and even death if untreated
What is hypoglycemic unawareness?
A condition in diabetes mellitus where autonomic neuropathy prevents the secretion of counter-regulatory hormones, making it difficult to detect and respond to low blood sugar